DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
CTM Castellum Inc

In early December 2025 Castellum’s CEO Glen Ives released a year-end shareholder letter summarizing a step-change year: record Q3 revenue, the company’s first GAAP net profit, significant debt reduction, and a contract backlog that more than doubled year-on-year. The letter also highlights a 103.3 million dollar NAVAIR Special Missions contract (via GTMR), a 66.2 million dollar NAWCAD Lakehurst prime contract for the SSI subsidiary, and fresh awards under the Missile Defense Agency’s SHIELD IDIQ program.
UNCY Unicycive Therapeutics Inc

On 18 December 2025, law firm Bragar Eagel & Squire announced a new investigation into certain officers and directors of Unicycive Therapeutics. The investigation follows the securities class action already filed in August 2025 over the company’s public statements around the OLC New Drug Application and the June 2025 FDA Complete Response Letter.
In practical terms this does not change day-to-day operations, but it reinforces a clear message: the OLC NDA path and related communication are now under legal as well as regulatory scrutiny.
VNDA Vanda Pharmaceuticals Inc

Tradipitant (motion sickness) is under NDA review with an unchanged PDUFA action date on December 30, 2025. FDA has started formal labeling discussions.
The FDA has lifted the partial clinical hold on long-term tradipitant studies, accepting that motion sickness is acute, so a 6-month dog tox study is no longer required.
SGMO Sangamo Therapeutics Inc

Full dossier on Sangamo Therapeutics (NASDAQ: SGMO) as of 18 December 2025: Fabry gene therapy ST-920, neurology pivot, cash and dilution, Nasdaq listing risk, analyst and retail sentiment, and scenario analysis including a hypothetical bullish path if the Fabry BLA is accepted for review.
RunUp Biotech 2026
This guide is the core pillar of the Run-Up Biotech 2026 framework. It is designed as a reference map for event-driven traders who want to follow biotech catalysts in a more structured way: from calendar building and candidate selection to timing, risk management and post-event review.
NRXP NRX Pharmaceuticals Inc

NRx has announced the conversion of the remaining 5.4M USD owed to Anson Funds into common equity, with no additional warrants or repricing features disclosed. Management states that, following this transaction, the year-end 2025 balance sheet is expected to show no remaining convertible debt and a “debt-free” capital structure.
OMER Omeros Corp Yartemlya (narsoplimab)

This article is built around one simple idea: before the label is public, you can only discuss “expectations” seriously if you anchor them to official sources, peer-reviewed evidence, and observable regulatory patterns. This is not a “tip.” It is a disciplined reading exercise.
OMER Omeros: The Finish Line Approaches

Omeros Corporation is approaching a binary regulatory event: the U.S. Food and Drug Administration (FDA) is scheduled to decide on the Biologics License Application (BLA) for narsoplimab (to be marketed as Yartemlya) in hematopoietic stem cell transplant-associated thrombotic microangiopathy (TA-TMA) on December 26, 2025. This is the company’s most important near-term catalyst and one of the most closely watched PDUFAs into year-end.
RXRX Recursion Pharmaceuticals Inc

J.P. Morgan upgraded Recursion Pharmaceuticals (RXRX) from Neutral to Overweight and increased its price target from 10 to 11 USD after reviewing new Phase 1b/2 data for REC-4881 in familial adenomatous polyposis (FAP). At a spot price around 4.20 USD, the new target implies more than 160 percent upside versus the current quote
PMGC’s subsidiary NorthStrive Biosciences

On December 17, 2025, PMGC’s subsidiary NorthStrive Biosciences announced the initiation of Phase III of its AI-driven drug discovery program in partnership with Yuva Biosciences:contentReference[oaicite:5]{index=5}. This stage is intended to experimentally validate multiple candidate compounds prioritized by YuvaBio’s MitoNova™ AI platform during Phase II, focusing on enhancing mitochondrial function and preserving muscle mass in obese patients
ADTX Sparked Aditxt

Deep dive on Aditxt Inc. (ADTX), a highly distressed micro-cap trying to build an ecosystem around immune tolerance, precision diagnostics and women’s health, while struggling with going concern warnings, heavy dilution and a very fragile balance sheet.
ALDX Aldeyra Therapeutics Inc Update dec 16

On December 16, 2025, the FDA formally extended the PDUFA target action date for reproxalap in dry eye disease from December 16, 2025 to March 16, 2026, after Aldeyra submitted the Clinical Study Report (CSR) for its dry-eye field trial to the NDA at the FDA’s request.
CUE Emergency Deep Dive

Cue Biopharma is a clinical-stage biopharmaceutical company developing a novel class of injectable biologics designed to selectively engage and modulate disease-specific T cells using its Immuno-STAT platform, targeting both autoimmune disease and cancer.
FBIO Fortress Biotech

Fortress Biotech has evolved into a hybrid model: part commercial dermatology company (via Journey Medical), part royalty/affiliate platform with exposure to multiple late-stage assets developed by partners and subsidiaries.
RCKT Rocket Pharmaceuticals Inc

Rocket Pharmaceuticals is a gene therapy-focused company with disclosed programs across lentiviral (hematology) and AAV (cardiovascular) platforms, as described in SEC-filed materials.
GRCE Grace Therapeutics

Grace Therapeutics has disclosed that it submitted an NDA for GTx-104 (intravenous nimodipine) on June 25, 2025, and that the FDA accepted the application for review with a PDUFA target date of April 23, 2026.
Biotech Weekly Recap

This week delivered a clear “biotech reality check” mix: multiple FDA decisions (including a first-in-class gene therapy), two new oral options for gonorrhea, and several high-visibility clinical readouts across obesity/metabolic and rare disease. The flow of headlines was “event-driven”, with market attention concentrating on hard endpoints, safety/tolerability, and execution quality.
MARKET REPORT – WEEK 15–19 DECEMBER 2025
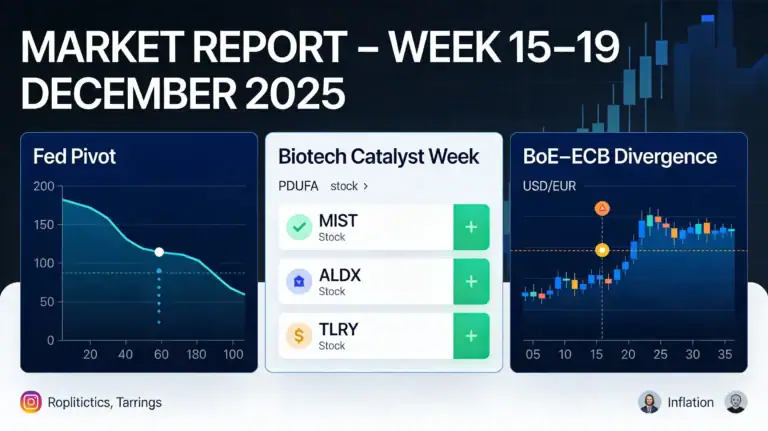
The coming week looks like a genuine pivot week for global markets, with three macro catalysts in sequence that will reshape expectations on rates, growth and inflation. The backdrop is a rotation into small caps and cyclicals, supported by a Fed that has shifted into a less aggressive “pause” mode and by a potential divergence between central banks: the BoE is expected to cut (18 Dec), the ECB is likely to stay on hold, and US CPI (18 Dec) together with labour data will refine the timing of 2026 rate cuts.
TENX Tenax Therapeutics

Tenax Therapeutics is advancing oral levosimendan for pulmonary hypertension associated with HFpEF (PH-HFpEF), with a registrational path built around the LEVEL Phase 3 program and a planned second global Phase 3 study (LEVEL-2) accepted by FDA in 2025.
SANA Biotechnology
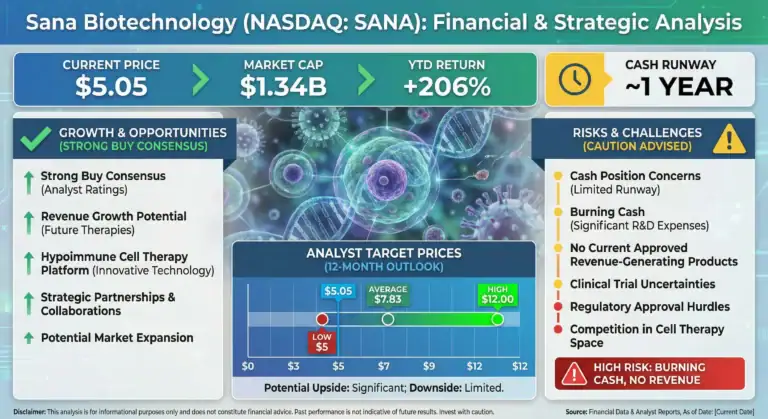
SANA Biotechnology is often framed as a high-risk / high-reward cellular engineering story. The core thesis revolves around its proprietary hypoimmune (HIP) platform: engineered cells designed to evade immune rejection, with the long-term goal of enabling allogeneic cell therapies without chronic immunosuppression.