DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
QNTM Quantum BioPharma Ltd

QNTM Quantum BioPharma – Comprehensive Report EN IT QNTM – Quantum BioPharma Ltd NASDAQ: QNTM | CSE: QNTM | FSE: 0K91 Sector: Biotech / Pharmaceuticals Report Date: December 5, 2025 ⚠️ SPECULATIVE – HIGH RISK Current Price (Dec 5) $9.755…
QCLS Q/C Technologies
Q/C Technologies (QCLS) – Photonic Hype, Shkreli Factor and a Very Thin Runway | Q/C Technologies (QCLS) – Photonics, Shkreli factor e runway molto corta English Italiano Q/C Technologies (QCLS) NASDAQ: QCLS – Photonic computing pivot and Shkreli-fueled squeeze story…
AKBA Akebia Therapeutics Inc

Akebia Therapeutics is a Cambridge, MA-based biopharmaceutical company focused on kidney disease treatment. Leading the market with innovative therapies for anemia and renal dysfunction.
PDUFA dates december 2025
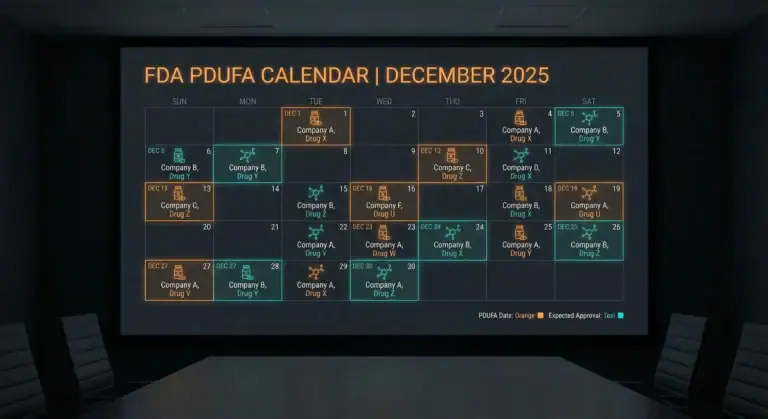
December 2025 is the densest FDA decision month of the year. In only 26 days the agency is expected to rule on fourteen key dossiers across oncology (Breyanzi), cardiology (Aficamten), ophthalmology (EYLEA HD, LYTENAVA), rare diseases (Narsoplimab, Mitapivat), endocrine disorders (Relacorilant), infectious diseases (Zoliflodacin) and more.
PDSB PDS Biotechnology Corporation

On December 3, 2025, PDS Biotech announced via an official 8-K filing that the FDA has granted a Type C Meeting to discuss a potential accelerated approval pathway based on PFS for PDS0101 + pembrolizumab in first-line HPV16+ HNSCC.
The market reaction was immediate: the stock gained roughly +18% on Dec 3 and closed at $0.899 on Dec 4, about +12% across 48 hours.
HOTH Hoth Therapeutics Inc

Hoth Therapeutics is a micro-cap, clinical-stage biotech focused on oncology-supportive care, mast-cell–driven diseases, neurodegeneration and metabolic/obesity indications. The company just issued a comprehensive pipeline update covering four core programs: HT-001 (topical therapy for EGFR-inhibitor rash), HT-KIT (KIT-driven mast-cell diseases, Orphan Drug Designation), HT-ALZ (Alzheimer’s / CNS inflammation) and a newly launched GDNF-based metabolic program in collaboration with the Atlanta VA Medical Cente
ONDS Ondas Holdings Update dec 3
2025, Ondas Holdings transformed from a marginal wireless communications company into an emerging leader in autonomous defense, driven by strategic acquisitions, record government contracts, and a rapidly expanding global customer pipeline. With Q3 2025 revenue of $10.1 million, a record backlog of $23.3 million, and an ultra-strong cash position of $840 million, Ondas is exceptionally positioned to capitalize on the boom in the counter-UAS market.
PLRZ Polyrizon Ltd

Polyrizon Ltd is an early-stage biotech leveraging proprietary Capture & Contain (C&C) hydrogel technology for nasal barriers against allergens/viruses and Trap & Target (T&T) for enhanced intranasal drug delivery
MREO Mereo BioPharma

LONDON, Nov. 10, 2025 (GLOBE NEWSWIRE) -- Mereo BioPharma Group plc (NASDAQ:MREO) ("Mereo" or the "Company"), a clinical-stage biopharmaceutical company focused on rare diseases, today announced its financial results for the third quarter ended September 30, 2025, and provided recent corporate highlights.
ALT Altimmune Update dec 3
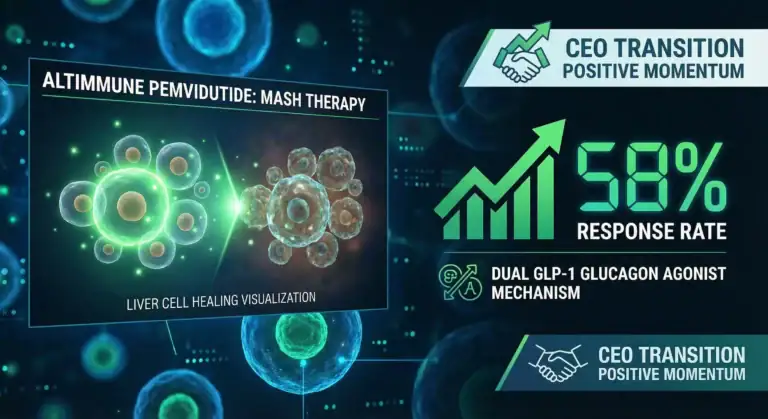
Altimmune is entering a pivotal phase with several bullish drivers converging through Q4 2025 and into 2026
ADCT ADC Therapeutics
ADC Therapeutics announced updated LOTIS-7 Phase 1b trial results on December 3, 2025, showing highly encouraging efficacy and manageable safety for ZYNLONTA + glofitamab combination in relapsed/refractory diffuse large B-cell lymphoma (DLBCL)
JANX

Janux Therapeutics has gone from market darling to high-risk story stock in 2025:
52-Week High (early 2025): around US$71–72 per share (after strong early JANX007 data and sector momentum).
Price after December 2–3, 2025 update: mid-teens (≈US$16–17), following a two-day collapse of ~50% after the latest Phase 1 JANX007 update.
CAPR Capricor Therapeutics (CAPR)

Positive Phase 3 HOPE-3 topline results in Duchenne muscular dystrophy cardiomyopathy with statistically significant skeletal and cardiac benefit, reopening the regulatory path after the 2025 CRL.
GUTS Fractyl Health Inc

Fractyl Health (NASDAQ: GUTS) is emerging as one of the most interesting names in the metabolic space thanks to its flagship device Revita®, a duodenal mucosal resurfacing (DMR) procedure designed to maintain weight loss after stopping GLP-1 drugs.
VERA Vera therapeutics

VERA Therapeutics is a biopharmaceutical company in advanced clinical stage focused on serious immunological diseases. The company achieved its most important milestone in November 2025 with the submission of a Biologics License Application (BLA) to the FDA for atacicept, its lead candidate for the treatment of IgA Nephropathy (IgAN).
VSTM Verastem Inc

Verastem Oncology is transitioning from a late-stage developer into a commercial oncology company focused on RAS/MAPK pathway-driven cancers.
VERI Veritone
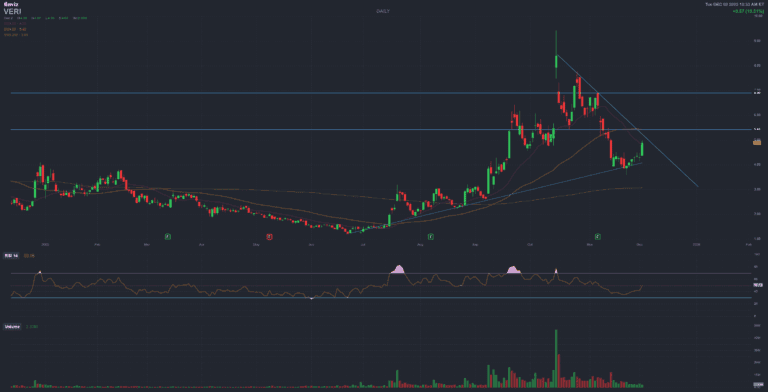
Veritone is a small-cap AI/software company that has struggled for several years with dilution, weak fundamentals, and high volatility. Today’s ~12% move appears more like a speculative rebound than a structural change
PMCB PharmaCyte Biotech
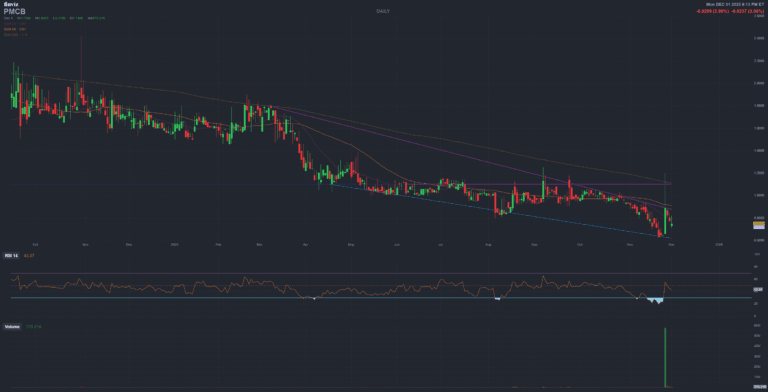
PharmaCyte has successfully monetized its equity stake in Femasys Inc. (FEMY), increasing its cash and marketable securities to roughly ~20 million USD, up from about 13.3 M USD as of July 31, 2025, according to the company’s press release and BusinessWire distribution.
CTOR Citius Oncology Inc (ctxr)
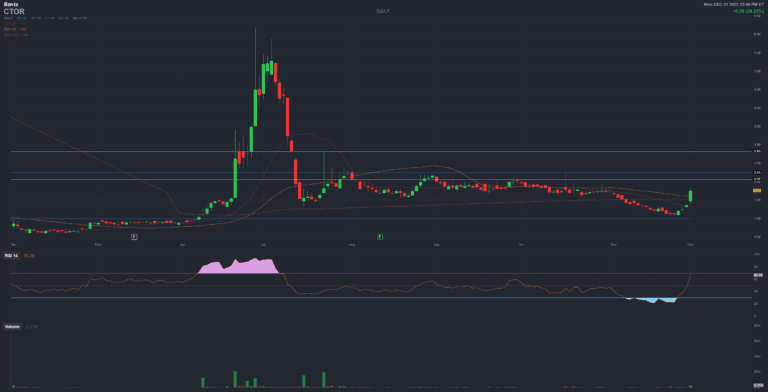
FDA approval secured June 2024 with orphan drug exclusivity through 2031
$8.27M inventory ready for immediate commercialization
Strong clinical efficacy: 36.2% ORR, 84.4% tumor burden reduction
HOLO MicroCloud Hologram Inc.

MicroCloud Hologram Inc. (HOLO) is a Chinese technology company specializing in holographic LiDAR technology, autonomous driving ADAS systems, and holographic digital twin solutions. The company represents a **recovery play** from a post-reverse-split penny stock to a **profitable, cash-rich operation** with significant exposure to the autonomous vehicle and advanced driver assistance systems (ADAS) market