DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Hoth Therapeutics Inc. – Pipeline & Catalyst Report
HOTH – GDNF Metabolic Program, HT-001 Phase 2 and HT-KIT Orphan Drug
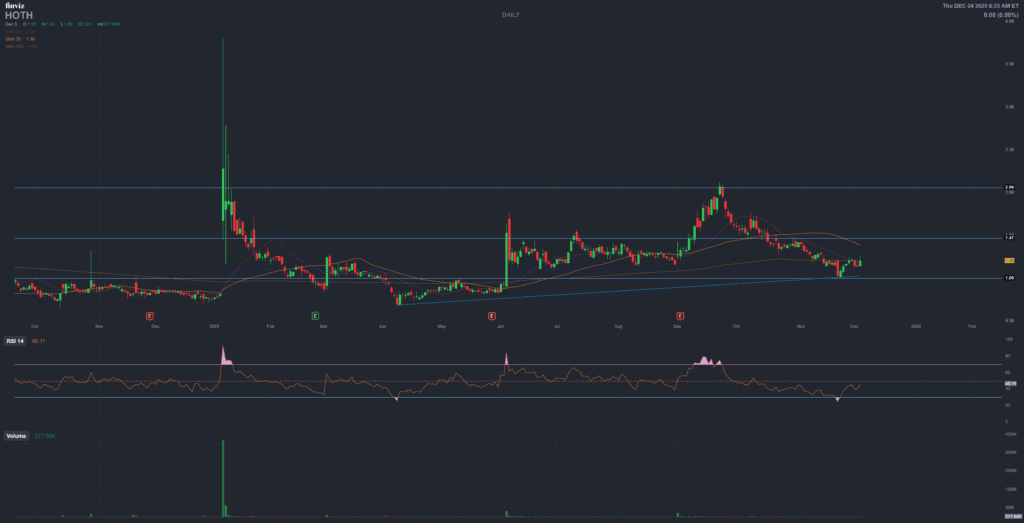
Price & Market Cap
$1.20
Approx. extended-hours price on 4 Dec 2025, market cap around $18M
(Finviz / Nasdaq). :contentReference[oaicite:1]{index=1}
Balance Sheet
$7.8M
Cash and cash equivalents as of 30 Sep 2025; no debt on the balance sheet. :contentReference[oaicite:2]{index=2}
Programs in Focus
4
HT-001 (Phase 2), HT-KIT (IND-enabling), HT-ALZ (GLP / PK), GDNF metabolic program (preclinical). :contentReference[oaicite:3]{index=3}
Executive Summary
Hoth Therapeutics is a micro-cap, clinical-stage biotech focused on oncology-supportive care, mast-cell–driven diseases, neurodegeneration and metabolic/obesity indications. The company just issued a comprehensive pipeline update covering four core programs: HT-001 (topical therapy for EGFR-inhibitor rash), HT-KIT (KIT-driven mast-cell diseases, Orphan Drug Designation), HT-ALZ (Alzheimer’s / CNS inflammation) and a newly launched GDNF-based metabolic program in collaboration with the Atlanta VA Medical Center. :contentReference[oaicite:4]{index=4}
- HT-001 continues to enroll in the CLEER-001 Phase 2 trial for EGFR-inhibitor–associated rash, with a favourable safety profile and encouraging trends in rash severity and pruritus. :contentReference[oaicite:5]{index=5}
- HT-KIT holds FDA Orphan Drug Designation for mast-cell diseases and is moving through IND-enabling toxicology and manufacturing scale-up, with an IND filing targeted for 2026. :contentReference[oaicite:6]{index=6}
- HT-ALZ is advancing GLP and PK work with positive CNS penetration modelling; a regulatory package is expected to mature in 2026. :contentReference[oaicite:7]{index=7}
- The GDNF weight-loss and metabolic program is VA-backed, targeting obesity and fatty liver disease, with early data expected in 2026. :contentReference[oaicite:8]{index=8}
The equity remains highly speculative: HOTH is loss-making, reliant on capital markets, and trades with very high volatility. This report is descriptive only and does not contain buy/sell recommendations.
Quick Data – HOTH Snapshot
| Item | Value (approx.) | Notes / Source |
|---|---|---|
| Share price | $1.20 | Finviz intraday / extended hours around 4 Dec 2025. :contentReference[oaicite:9]{index=9} |
| Market cap | ~$18M | Micro-cap range; precise value fluctuates with price. :contentReference[oaicite:10]{index=10} |
| Cash (30 Sep 2025) | $7.85M | Q3 2025 financial results, no outstanding debt disclosed. :contentReference[oaicite:11]{index=11} |
| Q3 2025 net loss | Approx. −$3.0M | Rough order-of-magnitude from Q3 filing and Rhea-AI summary. :contentReference[oaicite:12]{index=12} |
| Core programs | 4 | HT-001, HT-KIT, HT-ALZ, GDNF metabolic program. :contentReference[oaicite:13]{index=13} |
Pipeline Overview
Phase 2 – CLEER-001
HT-001 – EGFR-inhibitor rash
Topical therapy aimed at managing skin toxicity from EGFR inhibitors and potentially other targeted oncology agents. The CLEER-001 Phase 2 trial is ongoing, with no dose-limiting toxicities reported and encouraging trends in rash severity and pruritus. :contentReference[oaicite:14]{index=14}
- Unmet need: patients often reduce or interrupt life-saving cancer therapy because of rash.
- Near-term milestone: updated Phase 2 data in coming months. :contentReference[oaicite:15]{index=15}
Orphan – IND enabling
HT-KIT – Mast-cell diseases and KIT-driven cancers
Precision KIT-inhibitor program for systemic mastocytosis and other rare KIT-driven diseases. HT-KIT already has FDA Orphan Drug Designation and has shown potent suppression of KIT signalling and mast-cell activation in preclinical models. :contentReference[oaicite:16]{index=16}
- IND-enabling toxicology and CMC activities ongoing.
- Company guides to IND filing and first-in-human study in 2026. :contentReference[oaicite:17]{index=17}
GLP / PK work
HT-ALZ – Alzheimer’s / CNS inflammation
CNS-directed therapeutic candidate with supportive data across pharmacokinetics, biodistribution and blood–brain-barrier modelling. GLP studies are underway and a regulatory-facing package is expected to mature in 2026. :contentReference[oaicite:18]{index=18}
Preclinical – VA collaboration
GDNF-based Weight-loss & Metabolic Program
Novel biologic approach using GDNF to target obesity, hepatic steatosis and metabolic dysfunction, developed with the Atlanta VA Medical Center. Preclinical diet and dosing work has started, with first data expected in 2026. :contentReference[oaicite:19]{index=19}
Key 2025–2026 Catalysts
| Catalyst | Timing (approx.) | Details |
|---|---|---|
| HT-001 Phase 2 CLEER-001 update | Coming months (2026) | Safety and efficacy trends in EGFR-inhibitor rash – important proof-of-concept for the platform. :contentReference[oaicite:20]{index=20} |
| HT-KIT IND filing | 2026 | Completion of IND-enabling toxicology and CMC, followed by first-in-human trial in mast-cell diseases. :contentReference[oaicite:21]{index=21} |
| HT-ALZ GLP / PK package | 2026 | More detailed CNS penetration and neuroinflammation data, foundation for future clinical plans. :contentReference[oaicite:22]{index=22} |
| GDNF metabolic program read-out | Early 2026 | First preclinical results from the VA-backed obesity and fatty-liver study. :contentReference[oaicite:23]{index=23} |
Sentiment – Stocktwits, X and Reddit
Stocktwits
Recent messages around the pipeline update are predominantly bullish, with many retail traders focusing on the combination of a very small market cap and multiple shots on goal (HT-001, HT-KIT, HT-ALZ, GDNF). The tone is clearly speculative and driven by catalysts rather than fundamentals.
X (Twitter)
Fin-twit accounts commenting on HOTH highlight the obesity/GLP-1 narrative and the VA collaboration, together with the EGFR-rash opportunity. There are also sceptical comments regarding dilution risk and the size of the addressable market for a small company.
Threads on small-cap and biotech subreddits show a mix of enthusiasm and caution. Bullish posts emphasise “multiple catalysts at micro-cap valuation”; bearish voices point to the cash runway and historical drawdowns after spikes.
Sentiment comments are anecdotal, based on a manual review of recent public posts
on Stocktwits, X and Reddit. These are opinions from non-professional traders and
should never be used as a stand-alone decision tool.
Bull vs Bear – Scenario Framework
Bull case
- HT-001 delivers convincing Phase 2 data, enabling global expansion and potential partnerships.
- HT-KIT IND accepted, early human data show biological activity in mast-cell disease.
- VA GDNF program produces strong weight-loss and liver-fat signals, attracting attention in the obesity space.
- Company manages to fund operations without extreme dilution at current prices.
Bear case
- HT-001 data are modest or inconclusive, limiting commercial potential.
- Delays or issues in IND-enabling work for HT-KIT and HT-ALZ push timelines beyond 2026.
- Preclinical GDNF results are weak or difficult to translate into a clear clinical plan.
- Heavy dilution is required to finance trials, eroding any upside for existing shareholders.
Sources & Tools
Primary sources used for this report
- Hoth Therapeutics – Comprehensive pipeline update press release (Dec 3, 2025). :contentReference[oaicite:24]{index=24}
- Q3 2025 financial results and company summaries via Rhea-AI / StockTitan. :contentReference[oaicite:25]{index=25}
- News coverage and analysis on Investing.com. :contentReference[oaicite:26]{index=26}
- Additional background from market-data platforms (Finviz, Nasdaq, SEC EDGAR). :contentReference[oaicite:27]{index=27}
Legal and regulatory disclaimer (EN)
This document is provided strictly for educational and informational purposes. It is not, and must not be interpreted as, investment advice, a solicitation, or a recommendation to buy or sell any financial instrument. All information is based on public sources believed to be reliable, but cannot be guaranteed for completeness or accuracy. Micro-cap biotech stocks are highly speculative and can result in the total loss of capital.
Nothing in this report has been prepared in accordance with the requirements designed to promote the independence of investment research. The author is not an investment advisor registered with the SEC, FINRA or CONSOB. Readers remain solely responsible for their own decisions and should consult a qualified, licensed financial professional before making any investment, especially in high-risk sectors such as biotechnology. Affiliate links may be present: if you choose to open tools or services through those links, the author may receive a small commission, at no extra cost to you.
This document is provided strictly for educational and informational purposes. It is not, and must not be interpreted as, investment advice, a solicitation, or a recommendation to buy or sell any financial instrument. All information is based on public sources believed to be reliable, but cannot be guaranteed for completeness or accuracy. Micro-cap biotech stocks are highly speculative and can result in the total loss of capital.
Nothing in this report has been prepared in accordance with the requirements designed to promote the independence of investment research. The author is not an investment advisor registered with the SEC, FINRA or CONSOB. Readers remain solely responsible for their own decisions and should consult a qualified, licensed financial professional before making any investment, especially in high-risk sectors such as biotechnology. Affiliate links may be present: if you choose to open tools or services through those links, the author may receive a small commission, at no extra cost to you.
Hoth Therapeutics Inc. – Report Pipeline e Catalyst
HOTH – Programma metabolico GDNF, HT-001 Fase 2 e HT-KIT Orphan
Prezzo e Market Cap
$1,20
Prezzo indicativo in pre-/post-market al 4 dicembre 2025, market cap intorno
a 18 milioni di dollari (Finviz / Nasdaq). :contentReference[oaicite:28]{index=28}
Situazione Cassa
$7,8M
Cassa e equivalenti al 30 settembre 2025; nessun debito riportato in
bilancio. :contentReference[oaicite:29]{index=29}
Programmi chiave
4
HT-001, HT-KIT, HT-ALZ e programma metabolico GDNF in collaborazione con la
VA di Atlanta. :contentReference[oaicite:30]{index=30}
Executive Summary
Hoth Therapeutics è una biotech micro-cap in fase clinica, focalizzata su supportive care in oncologia, patologie da mast-cellule, neurodegenerazione e indicazioni metaboliche/obesità. L’ultimo comunicato ufficiale riassume quattro programmi centrali: HT-001 (terapia topica per rash da EGFR-inibitori), HT-KIT (patologie mast-cell–driven, con designazione Orphan), HT-ALZ (Alzheimer / infiammazione del SNC) e un nuovo programma metabolico basato su GDNF in collaborazione con l’Atlanta VA Medical Center. :contentReference[oaicite:31]{index=31}
- HT-001 prosegue nello studio CLEER-001 di Fase 2 per il rash da EGFR-inibitori, con profilo di sicurezza favorevole e trend incoraggianti su severità del rash e prurito. :contentReference[oaicite:32]{index=32}
- HT-KIT ha ricevuto la designazione Orphan Drug FDA per malattie mast-cell–driven e sta completando gli studi tossicologici e di produzione propedeutici alla sottomissione IND nel 2026. :contentReference[oaicite:33]{index=33}
- HT-ALZ avanza con studi GLP e PK, con modellistica positiva di penetrazione nel SNC; il pacchetto regolatorio è previsto in maturazione nel 2026. :contentReference[oaicite:34]{index=34}
- Il programma metabolico GDNF è supportato dalla VA e mira a obesità e steatosi epatica, con primi dati attesi nel 2026. :contentReference[oaicite:35]{index=35}
Il titolo resta altamente speculativo: HOTH è in perdita, dipende dal mercato dei capitali e mostra una volatilità molto elevata. Questo report è esclusivamente descrittivo e non contiene raccomandazioni di acquisto o vendita.
Disclaimer legale (IT)
Questo documento ha finalità esclusivamente informative e didattiche. Non costituisce consulenza in materia di investimenti, né sollecitazione o raccomandazione ad acquistare o vendere strumenti finanziari. Tutte le informazioni derivano da fonti pubbliche ritenute affidabili, ma non ne è garantita l’accuratezza o completezza. Le small e micro-cap biotech sono tra gli strumenti più rischiosi in assoluto e possono portare alla perdita totale del capitale.
Il presente report non è redatto secondo le regole che disciplinano la ricerca finanziaria indipendente. L’autore non è un consulente finanziario abilitato da CONSOB o SEC. Ogni lettore resta pienamente responsabile delle proprie decisioni e dovrebbe confrontarsi con un professionista abilitato prima di effettuare investimenti, in particolare in settori ad alto rischio come le biotecnologie. Possono essere presenti link di affiliazione; se decidi di utilizzare servizi tramite tali link, l’autore può ricevere una piccola commissione, senza costi aggiuntivi per te.
Questo documento ha finalità esclusivamente informative e didattiche. Non costituisce consulenza in materia di investimenti, né sollecitazione o raccomandazione ad acquistare o vendere strumenti finanziari. Tutte le informazioni derivano da fonti pubbliche ritenute affidabili, ma non ne è garantita l’accuratezza o completezza. Le small e micro-cap biotech sono tra gli strumenti più rischiosi in assoluto e possono portare alla perdita totale del capitale.
Il presente report non è redatto secondo le regole che disciplinano la ricerca finanziaria indipendente. L’autore non è un consulente finanziario abilitato da CONSOB o SEC. Ogni lettore resta pienamente responsabile delle proprie decisioni e dovrebbe confrontarsi con un professionista abilitato prima di effettuare investimenti, in particolare in settori ad alto rischio come le biotecnologie. Possono essere presenti link di affiliazione; se decidi di utilizzare servizi tramite tali link, l’autore può ricevere una piccola commissione, senza costi aggiuntivi per te.
Hoth Therapeutics (HOTH) – Addendum
December 2025 Update – Pipeline, SEC Filings, Analyst Scenarios & Retail Sentiment
1. Snapshot vs original report
Position in early December 2025 (approximate):
- Share price: around 1.20 USD, micro-cap (~18M USD market cap, no financial debt).
- Stage: clinical-stage, no approved products; value concentrated in HT-001, HT-KIT, HT-ALZ and the new GDNF metabolic program.
- Balance sheet: cash around 7–8M USD, total liabilities ~1M USD, debt-free structure.
The big change vs the initial report is not a single binary event, but a sequence of pipeline, regulatory and financing moves that re-position HOTH as a high-risk, high-optionality micro-cap with multiple shots on goal and clear financing risk.
2. SEC filings & capital structure – key points
Main filings since the original report
- 28 Mar 2025 – Form 10-K (FY 2024): restates prior periods for R&D and prepayments classification; confirms a small company with limited cash, no debt and ongoing operating losses.
- 12 May / 12 Aug / 12 Nov 2025 – 10-Q Q1–Q3 2025: R&D focused on HT-001, HT-KIT, HT-ALZ; operating cash burn of a few million USD year-to-date, no revenue.
- 21 Mar 2025 – 8-K, accounting restatement: audit committee declares non-reliance on several 2023–2024 quarters and restates them; cash and cash equivalents are unchanged, but control weaknesses are flagged.
- 14 Nov 2025 – S-3 shelf registration: registers securities based on the 10-K and 10-Q series, allowing future equity raises at management’s discretion. This is a clear dilution overhang for any multi-year scenario.
Capital & dilution setup
| Metric (approx.) | Value |
|---|---|
| Shares outstanding (Mar 2025) | ~13.2M common shares |
| Debt | 0 USD (no financial debt) |
| Cash | ~7.8M USD |
| Shelf registration | S-3 effective Nov 2025, enabling future raises |
For a trader, the combination of micro-cap size, no debt, limited cash and a fresh S-3 is exactly the typical setup where binary catalysts can produce sharp upside, but also where equity dilution can be used to fund the pipeline after positive news or even into strength.
3. Clinical, regulatory and strategic events (timeline)
Condensed timeline of material events after the base report
- 12 May 2025 – HT-KIT preclinical efficacy: antisense therapy shows >80% reduction in KIT expression and strong tumor inhibition in GIST/mast-cell models, with favourable safety in animals.
- 18 Jun 2025 – HT-KIT preclinical safety: positive safety data, no observed toxicity; company prepares GLP tox and IND pathway.
- 2 Sep 2025 – HT-KIT milestones exceeded: GLP-validated bioanalytical data, potent anti-tumor activity; HT-KIT is now positioned as an orphan-focused oncology asset (c-KIT driven cancers).
- 10 Sep 2025 – EMA submission for HT-001: Hoth files a CTA with the European Medicines Agency to expand the ongoing Phase II trial (HT-001 for EGFR-inhibitor skin toxicities) into Europe, a key regulatory milestone beyond the US.
- Oct 2025 – HT-KIT FDA Orphan Drug Designation: HT-KIT receives FDA orphan designation for certain rare c-KIT-driven cancers, strengthening the regulatory package and potential exclusivity.
- 27 Oct 2025 – GDNF metabolic/obesity program: launches a VA-backed study of a GDNF-based therapy targeting obesity and fatty liver disease; early data expected in 2026.
- 20 Nov 2025 – NVIDIA Connect Program: Hoth is accepted into NVIDIA’s Connect Program, gaining AI/accelerated computing resources to support discovery and modelling across the pipeline.
- 1–3 Dec 2025 – Noble Capital Markets conference: company presents at Noble’s Emerging Growth Equity Conference, positioning the story in front of small-cap-focused investors.
- 3 Dec 2025 – Comprehensive pipeline update: latest PR summarises progress across HT-001 (Phase 2 CLEER-001), HT-KIT (orphan oncology), HT-ALZ (Alzheimer’s) and the GDNF metabolic program, with IND for HT-KIT targeted in 2026.
In short: the story that used to be mostly about HT-001 is now marketed as a platform micro-cap with dermatology, oncology, neurology and metabolic optionality, supported by AI infrastructure.
4. Analyst outlook – price targets and scenarios
Consensus: Strong Buy (2–3 covering analysts)
12-month average PT: ~4.0–4.5 USD
Upside vs ~1.2 USD: +230–280%
EPS 2025–2027: negative
| Source | Rating / PT |
|---|---|
| MarketBeat | 3 analysts, average PT ~4.50 USD (range 4–5 USD), implied upside ~+275%. |
| StockAnalysis / Public.com | 2 analysts, “Strong Buy”, average PT 4.50 USD, high 5.00 USD. |
| Nasdaq article | Separate piece quoting an average PT ~4.08 USD (+240% upside) around mid-November 2025. |
| EPS forecast sites | EPS forecast still negative: around –0.53 USD in 2025, –0.60 USD in 2026, with losses persisting into 2027. |
| Long-term model sites | Aggressive algorithmic forecasts (multi-thousand-percent upside by 2035–2050) must be treated as speculative sentiment, not as a realistic base case. |
4.1 Bull case – what the optimists are implicitly assuming
- HT-001 delivers a clean Phase 2 readout that clearly reduces EGFRi-related rash and itching, with a favourable safety profile and practical topical use for oncologists.
- Regulatory leverage from EMA and orphan hooks: EMA expansion for HT-001 plus FDA Orphan Drug Designation for HT-KIT create credible partnership and pricing angles in oncology.
- HT-KIT IND in 2026 comes with compelling GLP data, allowing HOTH to position itself as a rare-cancer antisense player in a niche where even small markets can justify meaningful valuation.
- GDNF obesity/metabolic program shows early signs of efficacy in VA-backed work, giving optionality in a very large market even if it remains pre-clinical.
- AI angle via NVIDIA Connect and internal platforms speeds up target selection and trial design, improving the perception of HOTH as a technology-enabled, partner-ready platform.
- Micro-cap float amplifies moves: with a small market cap and low institutional ownership, any positive surprise can translate into outsized price swings, especially if coupled with news-driven volume spikes.
4.2 Bear case – what can go wrong
- No revenue and persistent losses: all analyst models keep EPS negative several years out, and the company remains fully dependent on capital markets to fund R&D.
- Dilution overhang: after the S-3 shelf and the existing ATM, equity raises are structurally on the table, especially if price spikes on news.
- Binary clinical risk: if HT-001 Phase 2 data are noisy or not clearly positive, the thesis loses its main near-term value driver; if HT-KIT or HT-ALZ stumble in tox or early studies, the platform narrative weakens.
- Crowded obesity and oncology space: even if HOTH generates positive signals, competition in supportive oncology care and metabolic disease is intense, with much larger and better-funded players.
- Accounting restatement and governance: the need to restate past periods is a reminder of execution and control risk in a small organisation.
- High volatility and thin liquidity: as seen when the stock tripled on interim data and then retraced, moves can be extreme both up and down, and slippage for real-money positions can be material.
5. Retail sentiment – Reddit, Stocktwits and X (Twitter)
5.1 What traders are saying
- Reddit (pennystocks, last 3–6 months): one detailed DD thread highlights the breadth of the pipeline, consensus PT around 4–5 USD, cash in the ten-million range and no financial debt. The tone is constructive but clearly retail and speculative, with emphasis on asymmetric upside rather than fundamental valuation.
- Stocktwits (2025): sentiment swung to extremely bullish when HOTH tripled on interim HT-001 data, with retail messages talking about multi-bagger potential and double-digit price targets; message volume is now lower but interest tends to spike again around every new PR or conference headline.
- X / Twitter: posts mix the official corporate feed (pipeline and PR amplification) with trader accounts focusing on short-term setups, breakouts above specific levels and the digital-asset treasury angle (plan to hold Bitcoin/Ethereum/Solana as part of reserves).
- Noise vs signal: algorithmic forecast sites quote extreme long-term targets, which are often shared in retail channels without context; these are models, not research, and should be treated as sentiment indicators rather than inputs for a serious thesis.
5.2 Sentiment summary
My read of the crowd:
Retail sentiment is tilted bullish but very fragile. Enthusiasm increases sharply around news and fades just as fast on quiet days or minor sell-offs. Most public comments clearly come from non-professional traders.
For a report like this, Reddit/Stocktwits/X are useful mainly as sentiment thermometers: they show how quickly the story can become crowded, but they do not replace SEC filings, company presentations or proper risk analysis.
6. Useful links and tools
This addendum is based exclusively on public information from SEC filings, official company communications and major news outlets, cross-checked as of 4 December 2025. It is an informational trading/educational note and does not contain, and must not be read as, any form of investment advice or buy/sell recommendation. Markets, prices and opinions can change quickly; data and interpretations here may become outdated or contain errors despite best efforts to verify them.
1. Fotografia rispetto al report iniziale
Situazione a inizio dicembre 2025 (approssimativa):
- Prezzo per azione: intorno a 1,20 USD, micro-cap (circa 18M USD di capitalizzazione, senza debiti finanziari).
- Stadio: società in fase clinica, senza prodotti approvati; la creazione di valore ruota soprattutto attorno a HT-001, HT-KIT, HT-ALZ e al nuovo programma metabolico GDNF.
- Stato patrimoniale: cassa intorno a 7–8M USD, passività totali ~1M USD, struttura priva di debito finanziario.
Il cambiamento principale rispetto al primo report non è un singolo evento binario, ma una serie di mosse su pipeline, regolatorio e funding che trasformano HOTH in una micro-cap a rischio elevato, ma con più “colpi in canna” e un rischio di finanziamento evidente.
2. Filings SEC e struttura del capitale – punti chiave
Principali documenti dopo il report base
- 28 mar 2025 – Form 10-K (FY 2024): rettifica diversi periodi per la contabilizzazione di R&D e prepagate; conferma profilo di piccola biotech con poca cassa, nessun debito e perdite operative ricorrenti.
- 12 mag / 12 ago / 12 nov 2025 – 10-Q Q1–Q3 2025: R&D concentrata su HT-001, HT-KIT, HT-ALZ; cash burn operativo di qualche milione di dollari nell’anno, assenza di ricavi.
- 21 mar 2025 – 8-K, restatement contabile: il comitato di controllo contabile dichiara la non-affidabilità di vari trimestri 2023–2024 e li ri-dichiara; cassa invariata, ma emergono debolezze nei controlli interni.
- 14 nov 2025 – Shelf registration S-3: registra strumenti azionari facendo leva sul 10-K e sui 10-Q, consentendo future emissioni di capitale a discrezione del management. Questo crea un chiaro overhang di diluizione in qualunque scenario pluriennale.
Impostazione finanziaria
| Indicatore (circa) | Valore |
|---|---|
| Azioni in circolazione (mar 2025) | ~13,2M azioni ordinarie |
| Debito finanziario | 0 USD (nessun debito) |
| Cassa | ~7,8M USD |
| Shelf S-3 | attiva da novembre 2025, abilita nuove emissioni |
Per chi fa trading, la combinazione di micro-cap, assenza di debito, cassa limitata e nuovo S-3 è il classico setup in cui i catalyst binari possono dare movimenti esplosivi, ma in cui la diluizione azionaria è uno strumento sempre pronto per finanziare la pipeline, specie dopo news positive.
3. Eventi clinici, regolatori e strategici (timeline)
Timeline sintetica degli eventi materiali post-report
- 12 mag 2025 – Efficacia preclinica HT-KIT: la terapia antisense mostra >80% di riduzione dell’espressione di KIT e forte inibizione tumorale in modelli GIST/mastocitari, con profilo di sicurezza favorevole negli animali.
- 18 giu 2025 – Dati di sicurezza HT-KIT: dati preclinici di sicurezza positivi, senza tossicità osservata; l’azienda prepara gli studi GLP e il percorso verso IND.
- 2 set 2025 – Milestone HT-KIT: dati GLP e bioanalitici validati, forte attività anti-tumorale; HT-KIT viene posizionato come asset oncologico orphan in tumori rari c-KIT-dipendenti.
- 10 set 2025 – Submission EMA HT-001: Hoth presenta un CTA all’EMA per estendere il trial di Fase II di HT-001 (tossicità cutanee da EGFRi) in Europa, passo regolatorio importante oltre gli Stati Uniti.
- ott 2025 – Designazione Orphan HT-KIT: HT-KIT riceve la qualifica Orphan Drug da FDA per alcune neoplasie rare c-KIT-driven, con potenziale beneficio in termini di esclusiva e incentivi.
- 27 ott 2025 – Programma metabolico GDNF: parte uno studio supportato dal VA su un candidato GDNF per obesità e malattia epatica; primi dati attesi nel 2026.
- 20 nov 2025 – Ingresso in NVIDIA Connect: Hoth viene accettata nel programma NVIDIA Connect, con accesso a risorse AI/accelerated computing per la ricerca e la modellistica lungo tutta la pipeline.
- 1–3 dic 2025 – Conference Noble: presentazione alla Noble Emerging Growth Equity Conference, con esposizione mirata verso investitori focalizzati sulle small-cap.
- 3 dic 2025 – Aggiornamento completo di pipeline: l’ultimo comunicato riassume i progressi su HT-001 (Fase 2 CLEER-001), HT-KIT (oncologia orphan), HT-ALZ (Alzheimer) e programma metabolico GDNF, con IND di HT-KIT prevista per il 2026.
In sintesi: da storia quasi solo HT-001, HOTH viene ora raccontata come micro-cap piattaforma con dermatologia, oncologia, neurologia e metabolico, sostenuta da infrastruttura AI.
4. Analisti – target price e scenari
Consenso: Strong Buy (2–3 analisti)
PT 12 mesi: ~4,0–4,5 USD
Upside vs ~1,2 USD: +230–280%
EPS 2025–2027: in perdita
| Fonte | Rating / PT |
|---|---|
| MarketBeat | 3 analisti, PT medio ~4,50 USD (forchetta 4–5 USD), upside potenziale intorno a +275%. |
| StockAnalysis / Public.com | 2 analisti, “Strong Buy”, PT medio 4,50 USD, massimo 5,00 USD. |
| Articolo Nasdaq | Articolo separato cita PT medio ~4,08 USD (circa +240% di upside) a metà novembre 2025. |
| EPS forecast | Le proiezioni su EPS restano negative: circa –0,53 USD nel 2025 e –0,60 USD nel 2026, con perdite attese anche nel 2027. |
| Modelli long-term | Alcuni siti algoritmici mostrano stime ultra-ottimistiche (migliaia di punti percentuali di rialzo entro il 2035–2050): numeri utili per capire il sentiment, non per costruire una valutazione seria. |
4.1 Scenario bull – cosa deve andare bene
- HT-001 esce con una Fase 2 chiara, che riduce in modo credibile la rash da EGFRi e il prurito, con profilo di sicurezza pulito e facilità d’uso per gli oncologi.
- Leve regolatorie EMA + Orphan: l’espansione EMA di HT-001 e la designazione Orphan per HT-KIT danno sostanza al discorso partnership/prezzi in oncologia.
- IND HT-KIT nel 2026 accompagnata da dati GLP solidi, che permettono di posizionare HOTH come player antisense in neoplasie rare dove anche mercati piccoli possono sostenere valutazioni interessanti.
- Programma GDNF mostra primi segnali incoraggianti in obesità/metabolico, offrendo optionalità in un mercato molto grande anche se ancora pre-clinico.
- Angolo AI (NVIDIA Connect + piattaforme interne) rende più credibile il racconto di pipeline “data-driven” e partner-friendly.
- Micro-cap con flottante ridotto: come già visto a inizio 2025, dati positivi possono innescare movimenti molto violenti al rialzo, se combinati con volumi e FOMO retail.
4.2 Scenario bear – dove si rompe la storia
- Mancanza di ricavi e perdite persistenti: tutte le proiezioni mantengono l’EPS in rosso per anni; la società resta totalmente dipendente dal mercato dei capitali.
- Overhang di diluizione: dopo S-3 e ATM, ogni spike di prezzo diventa un’occasione potenziale di raccolta, con impatto diretto sulla struttura per azione.
- Rischio binario sui trial: se i dati di Fase 2 di HT-001 risultano deboli o ambigui, salta il principale driver di valore nel breve; eventuali problemi di tox o efficacia in HT-KIT/HT-ALZ indeboliscono il racconto di piattaforma.
- Concorrenza pesante: supportive care in oncologia e obesità/metabolico sono aree con big pharma e mid-cap molto più capitalizzate.
- Restatement e governance: la necessità di riaprire bilanci recenti ricorda che nei micro-cap la execution risk include anche la contabilità.
- Volatilità estrema e liquidità sottile: come già sperimentato sul triplo di prezzo e successivo ritracciamento, i movimenti possono essere violenti in entrambe le direzioni, con slippage rilevante.
5. Sentiment retail – Reddit, Stocktwits e X
5.1 Cosa emerge dai canali
- Reddit (pennystocks, ultimi mesi): un thread di DD mette in evidenza la pipeline ampia, il PT di consenso intorno a 4–5 USD, la presenza di cassa a doppia cifra di milioni e l’assenza di debito finanziario. Il tono è costruttivo ma evidentemente retail, con focus su upside asimmetrico più che su valutazione fondamentale.
- Stocktwits (2025): il sentiment è passato a “estremamente bullish” nel momento in cui il titolo è triplicato sui dati intermedi di HT-001; ora i volumi di messaggi sono più bassi, ma tendono a esplodere di nuovo ad ogni nuovo comunicato o conference call.
- X / Twitter: convive il flusso ufficiale della società (pipeline, PR) con account di trader che guardano soprattutto a setup di breve, livelli tecnici e all’angolo “crypto treasury” (piano per detenere Bitcoin/Ethereum/Solana in riserva).
- Modelli automatici: forecast estremi pubblicati da siti algoritmici vengono condivisi come “prova” di enormi target di lungo periodo; in realtà sono indicatori di sentiment, non ricerca analitica.
5.2 Sintesi del sentiment
La mia lettura del sentiment:
Il sentiment retail è sbilanciato sul lato rialzista ma molto fragile. L’entusiasmo cresce forte sulle news e svanisce rapidamente nei periodi di silenzio o sulle correzioni. La quasi totalità dei commenti è chiaramente generata da trader non professionisti.
In un report di questo tipo, Reddit/Stocktwits/X servono soprattutto come termometro di sentiment: indicano quanto in fretta la storia può diventare affollata, ma non sostituiscono SEC, presentazioni della società o un’analisi del rischio strutturata.
6. Link e strumenti utili
Questo addendum si basa esclusivamente su informazioni pubbliche tratte da filings SEC, comunicati ufficiali della società e principali testate giornalistiche, verificate al 4 dicembre 2025. È una nota informativa a scopo didattico/trading e non contiene, né va letta come, alcuna forma di raccomandazione di investimento o indicazione di acquisto/vendita. Mercati, prezzi e opinioni possono cambiare rapidamente; dati e interpretazioni qui riportati possono diventare obsoleti o contenere errori nonostante il doppio controllo delle fonti.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


