DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
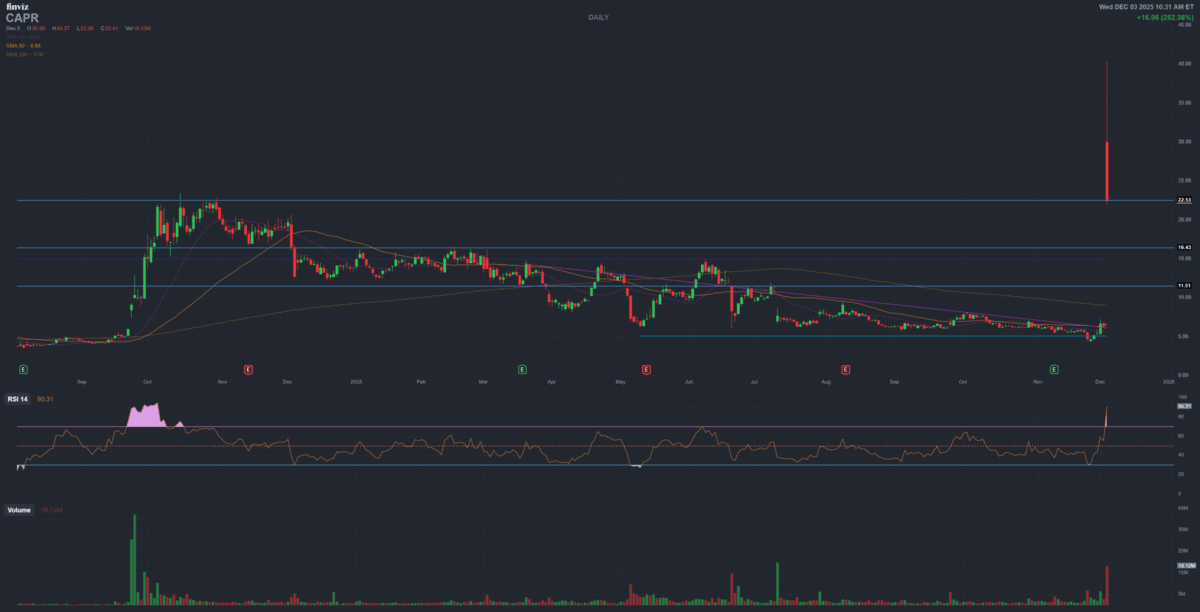
Biotech Catalyst Emergency Report
Capricor Therapeutics (CAPR) – HOPE-3 Positive Phase 3 Catalyst
NASDAQ: CAPR
Duchenne muscular dystrophy cardiomyopathy – cell therapy (Deramiocel / CAP-1002)
Positive Phase 3 HOPE-3 topline results in Duchenne muscular dystrophy cardiomyopathy with statistically significant skeletal and cardiac benefit, reopening the regulatory path after the 2025 CRL.
Updated: December 3, 2025 – HOPE-3 topline data day
Primary endpoint (PUL v2.0)
Met – p≈0.03
Key cardiac endpoint (LVEF)
Met – p≈0.04
Trial size
n = 106 (Phase 3)
Intraday move (today)
~+300% range
Contents
- 1. Executive summary – the catalyst
- 2. HOPE-3 topline results (Dec 3, 2025)
- 3. Detailed efficacy data (PUL & LVEF)
- 4. HOPE-3 trial design & population
- 5. Regulatory path & timeline
- 6. How HOPE-3 addresses the July 2025 CRL
- 7. Financials, cash runway & ATM
- 8. Scenario analysis & valuation frames
- 9. Key risks, volatility & upcoming catalysts
- 10. Verified sources & filings
- 11. Tools, charts & support
- 12. Legal & risk disclaimer
1. Executive Summary – HOPE-3 as a Major De-Risking Event
⚡ What happened today?
On December 3, 2025, Capricor Therapeutics reported positive topline results from its pivotal Phase 3 HOPE-3 trial of Deramiocel (CAP-1002) in boys and young men with Duchenne muscular dystrophy (DMD) cardiomyopathy. The randomized, double-blind, placebo-controlled study (n=106) met both the:
- Primary endpoint: Performance of Upper Limb (PUL v2.0) – statistically significant benefit
- Key secondary endpoint: Left Ventricular Ejection Fraction (LVEF) – statistically significant preservation of cardiac function
- All type-1 error–controlled secondaries: also achieved statistical significance
- Safety: Deramiocel maintained a favorable profile consistent with prior clinical experience
As a result, shares spiked in triple-digit percentage terms intraday, reflecting a sharp reassessment of regulatory odds and commercial potential after the July 2025 Complete Response Letter (CRL). :contentReference[oaicite:4]{index=4}
Skeletal muscle benefit
~54% slowing
Cardiac function
~91% LVEF preservation
PUL v2.0 p-value
≈ 0.03
LVEF p-value
≈ 0.04
SEC / company verified: efficacy data are taken from the official
Capricor press release and 8-K filed December 3, 2025, which describe a Phase 3
randomized, double-blind, placebo-controlled trial (n=106) meeting both the primary PUL
v2.0 endpoint and key cardiac LVEF endpoint with p-values around 0.03 and 0.04,
respectively, and confirm a favorable safety profile. :contentReference[oaicite:5]{index=5}
2. The Catalyst: HOPE-3 Topline Results (December 3, 2025)
After roughly eight months of uncertainty following the FDA CRL, HOPE-3 delivers the “adequate and well-controlled” Phase 3 dataset the Agency had requested. Capricor now plans to use these data in its formal response to the CRL and BLA resubmission.
Key messages from today’s data
- Statistically and clinically meaningful skeletal benefit – slowing loss of upper limb function in a population with advanced DMD.
- Clinically relevant cardiac benefit – preservation of left ventricular ejection fraction in a setting where cardiomyopathy is the leading cause of death.
- All pre-specified, type-1 error–controlled secondary endpoints met, strengthening the overall efficacy package.
- Safety and tolerability in line with prior Deramiocel experience from HOPE-2 and its open-label extension (>48 months of follow-up).
As CEO Linda Marbán, Ph.D., emphasized, HOPE-3 confirms and extends the durability signal seen in HOPE-2 and the long-term OLE, and is intended to directly address the issues raised in the 2025 CRL, in line with prior FDA guidance that a successful HOPE-3 could be sufficient to support approval. :contentReference[oaicite:6]{index=6}
3. Detailed Efficacy Results
3.1 Primary Endpoint – PUL v2.0 (Skeletal Muscle Function)
| Metric | Result | Interpretation |
|---|---|---|
| Change in PUL v2.0 vs. placebo | ~54% slowing of disease progression | Statistically significant treatment effect in upper limb function |
| Population (ITT) | n = 105 evaluable subjects | Randomized, double-blind, placebo-controlled trial |
| P-value | ≈ 0.03 (company disclosure) | Meets conventional 5% significance threshold |
| Clinical meaning | Slowing the decline in upper limb function by roughly half vs. placebo | Relevant to preserving independence and daily activities |
Clinician perspective – skeletal muscle
According to Craig McDonald, M.D. (UC Davis; National PI for HOPE-3), a nearly 54% slowing of skeletal muscle disease progression is described as “extraordinary in Duchenne” and closely linked to maintaining independence and quality of life in severely affected, mostly non-ambulatory patients. :contentReference[oaicite:7]{index=7}
3.2 Key Secondary Endpoint – LVEF (Cardiac Function)
| Metric | Result | Interpretation |
|---|---|---|
| Left Ventricular Ejection Fraction (LVEF) | ~91% preservation vs. placebo | Statistically significant stabilization of cardiac function |
| Evaluable patients | n = 83 with cardiac MRI | Majority with established cardiomyopathy at baseline |
| P-value | ≈ 0.04 | Statistically significant cardiac benefit |
| Clinical context | DMD cardiomyopathy is the leading cause of death in DMD | Cardiac preservation may translate into improved survival |
Clinician perspective – cardiomyopathy
Jonathan Soslow, M.D., MSCI (Vanderbilt) called the HOPE-3 cardiac findings “a significant advance in the management of Duchenne cardiomyopathy”, noting that stabilizing LVEF has been a major unmet need and that the statistically and clinically significant preservation seen with Deramiocel targets one of the most critical drivers of mortality in DMD. :contentReference[oaicite:8]{index=8}
4. HOPE-3 Trial Design & Population
| Trial parameter | Details (HOPE-3) |
|---|---|
| Phase | Pivotal Phase 3 |
| Design | Randomized, double-blind, placebo-controlled, multi-center |
| Sites | ~20 leading U.S. DMD centers |
| Enrollment | 106 eligible subjects (boys and young men with DMD) |
| Ambulatory status | Both ambulatory and non-ambulatory; skewed towards non-ambulatory population |
| Dosing | Deramiocel 150M cells/infusion vs. placebo, IV, every 3 months × 4 doses (12 months) |
| Concomitant therapy | Stable corticosteroids; ~90% on cardiac medications; >75% with diagnosed cardiomyopathy |
| Primary endpoint | Change from baseline in PUL v2.0 at 12 months |
| Key secondary endpoints | LVEF by cardiac MRI; additional motor performance measures (type-1 error controlled) |
Baseline characteristics
- Average age ~15 years across both arms.
- All participants on stable corticosteroid regimens.
- High background use of cardiac medications at baseline (~90%).
- More than three-quarters with a clinical diagnosis of cardiomyopathy.
- Demographics broadly representative of real-world DMD cardiomyopathy burden.
5. Regulatory Path & Timeline
5.1 Timeline – from CRL to HOPE-3 topline
July 2025
FDA issues a Complete Response Letter (CRL) for Deramiocel, citing insufficient efficacy evidence (reliance on Phase 2 + historical controls) and chemistry, manufacturing and controls (CMC) questions. Stock draws down sharply.
August 2025
Type A meeting with FDA: the Agency signals that a successful randomized, controlled Phase 3 (HOPE-3) could address efficacy concerns and form the basis of a complete response to the CRL. :contentReference[oaicite:9]{index=9}
September 2025
Capricor announces completion of the 12-month treatment phase for HOPE-3 and reiterates its plan to use HOPE-3 data in a resubmission package.
December 3, 2025
HOPE-3 topline results: primary and key cardiac endpoints met with statistical significance, all type-1 error–controlled secondary endpoints also positive, and favorable safety. Company confirms its intention to submit a response to the CRL incorporating these data. :contentReference[oaicite:10]{index=10}
5.2 Next steps – BLA response and potential approval window
- Near term (Q4 2025–Q1 2026): Capricor prepares and submits its formal response to the CRL, incorporating full HOPE-3 data, updated CMC documentation and long-term HOPE-2 / OLE safety.
- Review clock: management has indicated a 6-month FDA review window typical of a Type 2 resubmission, once the response is formally accepted for review.
- Potential decision timing: a reasonable working window for an FDA decision is mid-to-late 2026, assuming timely acceptance and no major new deficiencies.
- Priority Review Voucher (PRV) potential: Deramiocel holds Rare Pediatric Disease Designation, which may qualify Capricor for a PRV upon approval – historically monetizable for roughly $100M+ in other programs. :contentReference[oaicite:11]{index=11}
6. How HOPE-3 Addresses the July 2025 CRL
6.1 Main issues raised by the FDA
- Efficacy evidence: initial BLA relied heavily on Phase 2 HOPE-2 plus historical controls; FDA requested a more robust randomized, controlled Phase 3 dataset.
- CMC / manufacturing: questions on consistency and reproducibility of cell therapy manufacturing across lots.
- Long-term safety & durability: need for additional data on sustained benefit and safety in DMD, given the novel cell therapy modality.
6.2 HOPE-3 as a direct response
| FDA concern | HOPE-3 / recent data package |
|---|---|
| Efficacy evidence | Phase 3, randomized, double-blind, placebo-controlled trial (n=106) meeting primary PUL v2.0 and key LVEF endpoints with statistically significant, clinically meaningful treatment effects, plus positive type-1 error–controlled secondary endpoints. :contentReference[oaicite:12]{index=12} |
| CMC / manufacturing | Capricor has completed pre-license inspections of its San Diego GMP facility and provided extensive manufacturing data across >100 lots, aiming to show lot-to-lot consistency for Deramiocel (details in SEC filings and prior updates). :contentReference[oaicite:13]{index=13} |
| Long-term safety & durability | HOPE-3 expands exposure to 12-month randomized treatment; HOPE-2 and its open-label extension now provide >48 months of safety/efficacy follow-up, with no new safety signals highlighted in recent disclosures. :contentReference[oaicite:14]{index=14} |
| Cardiac benefit proof | HOPE-3 demonstrates statistically and clinically significant preservation of LVEF in a population where cardiomyopathy is the leading cause of mortality, directly addressing questions around the cardiac indication. :contentReference[oaicite:15]{index=15} |
Bottom line vs. the CRL
HOPE-3 does not guarantee approval, but it goes a long way toward resolving the explicit efficacy objections in the CRL. The remaining regulatory risk is mainly around CMC, any detailed subgroup analyses the FDA may request, and the Agency’s overall comfort with long-term safety for a first-in-class cell therapy in DMD.
7. Financial Update – Q3 2025 Snapshot (SEC-Verified)
7.1 Income statement (Q3 and 9M 2025)
| Item | Q3 2025 | Q3 2024 | 9M 2025 | 9M 2024 |
|---|---|---|---|---|
| Revenue | $0.0M | $2.3M | $0.0M | $11.1M |
| R&D expense | $20.4M | $11.8M | $61.3M | $35.4M |
| G&A expense | $5.9M | $3.5M | $17.7M | $10.6M |
| Total operating expenses | $26.3M | $15.3M | $79.0M | $46.0M |
| Net loss | ($24.6M) | ($12.6M) | ($74.9M) | ($33.4M) |
| Diluted EPS | ($0.54) | ($0.38) | ($1.64) | ($1.04) |
7.2 Balance sheet (as of September 30, 2025)
| Item | Sep 30, 2025 | Dec 31, 2024 | Δ vs. FY 2024 |
|---|---|---|---|
| Cash & marketable securities | $98.6M | $151.5M | ($52.9M) |
| Total assets | $126.4M | $170.5M | ($44.1M) |
| Total liabilities | $42.6M | $25.0M | +$17.6M |
| Stockholders’ equity | $83.9M | $145.5M | ($61.6M) |
SEC-verified financials: figures above are taken from Capricor’s
Form 10-Q for the quarter ended September 30, 2025 and associated earnings materials,
which report cash and marketable securities of approximately $98.6M, and confirm the
September 2025 ATM program with an aggregate capacity of $150M and no shares sold as of
the filing date. :contentReference[oaicite:16]{index=16}
7.3 Cash runway & ATM capacity
- Cash & marketable securities: ~$98.6M (Sep 30, 2025).
- Quarterly operating spend (Q3): ~$26.3M total OpEx implies an annualized burn a little above $100M.
- Runway: on simple math, this supports ~12–15 months of operations at the current burn rate, roughly into Q4 2026, consistent with management commentary that current resources fund operations through at least the next 12 months.
- ATM program: a $150M at-the-market facility was established in September 2025; according to the 10-Q, no shares had been sold under this program as of the filing date. :contentReference[oaicite:17]{index=17}
8. Scenario Analysis & Valuation Frames (Not Investment Advice)
HOPE-3 materially de-risks the regulatory and clinical profile of Deramiocel, but valuation remains highly sensitive to assumptions on approval probability, launch execution, pricing and competition. The ranges below are illustrative scenario frames, not price targets or recommendations.
? Bull scenario (optimistic)
$35–50
HOPE-3 data are accepted without major caveats; BLA response is filed promptly, and FDA grants approval in 2026. Deramiocel achieves strong uptake in DMD cardiomyopathy, with peak sales in the U.S./Japan region in the $200M–$400M+ range. Priority Review Voucher is monetized at >$100M.
? Base scenario (central case)
$15–25
FDA accepts HOPE-3 but may request additional analyses or modest label constraints. Approval still occurs around 2026–2027. Commercial ramp is more gradual, with peak sales in the $100M–$200M range by around 2030 and more moderate economics from ex-U.S. partners.
⚠️ Bear scenario (downside)
$3–8
Additional FDA objections emerge during the resubmission review; approval is delayed materially or potentially withheld. Deramiocel’s commercial adoption is limited, competition intensifies, and Capricor must rely heavily on equity financing, leading to substantial dilution.
These scenario bands are for educational purposes only and are not price targets or recommendations. They simplify a complex reality and ignore many variables (macro-environment, broader pipeline, exosome programs, etc.).
9. Key Risks, Volatility & Upcoming Catalysts
9.1 Regulatory risks
- CMC re-inspection risk: additional FDA inspections or questions at the manufacturing site could delay approval if issues arise.
- Subgroup / sensitivity analyses: if pivotal benefit is driven by a narrow subgroup, FDA could request more data or limit the label.
- Long-term safety: regulators may require more follow-up to confirm durability and rule out late safety signals for a chronic cell therapy.
- Competitive landscape: further progress in gene therapies or other DMD treatments could raise the efficacy bar or shift payer focus.
9.2 Commercial & financial risks
- Market size: DMD cardiomyopathy is an orphan indication; peak sales could be lower than optimistic ranges if eligible patients or duration of treatment are more limited than expected.
- Pricing & reimbursement: payers may push back on high cell-therapy pricing, require step-through, or narrow coverage criteria.
- Manufacturing scale-up: moving from clinical to commercial scale in cell therapy is operationally demanding and capital intensive.
- Dilution: the $150M ATM program provides flexibility but also creates the possibility of significant future equity issuance.
9.3 Near-term catalysts (next ~12 months)
- Full HOPE-3 dataset presentation: conference and/or peer-reviewed publication with more granular data.
- Formal response to the CRL / BLA resubmission: key milestone that may clarify review timelines.
- Regulatory feedback: additional FDA information requests or preliminary views on adequacy of HOPE-3 data.
- Business development: potential updates on existing or new partnerships (e.g., Nippon Shinyaku) and economics.
9.4 Volatility reminder
CAPR remains an event-driven, high-volatility small-cap biotech name. Historically, similar situations have seen daily moves of 15–20% (or more) around key regulatory and clinical news. Position sizing, diversification and risk management are critical considerations for any investor analysing such profiles.
10. Verified Sources & Filings
10.1 HOPE-3 topline & regulatory
- Capricor Therapeutics – press release, “Capricor Therapeutics Announces Positive Topline Results from Pivotal Phase 3 HOPE-3 Study of Deramiocel in Duchenne Muscular Dystrophy”, December 3, 2025.
- Capricor Therapeutics – Form 8-K, December 3, 2025, including HOPE-3 topline disclosure and attached press release.
- Capricor Therapeutics – press release, “Capricor Therapeutics Provides Regulatory Update on Deramiocel Program for Duchenne Muscular Dystrophy Following Type A Meeting”, September 2025.
- ClinicalTrials.gov – HOPE-3 trial record (NCT05126758) with protocol and design summary.
10.2 Financials & ATM program
- Capricor Therapeutics – Form 10-Q for the quarter ended September 30, 2025 (filed November 10, 2025), including full financial statements and ATM description.
- Capricor – press release, “Capricor Therapeutics Reports Third Quarter 2025 Financial Results and Provides Corporate Update”, November 2025.
10.3 Market & media coverage
- Reuters / MarketScreener – coverage of HOPE-3 topline results and intraday share move on December 3, 2025.
- StockTitan / other news aggregators – HOPE-3 topline headlines and sentiment tags.
Methodology note: all numerical data (trial size, p-values, cash,
expenses, ATM size) in this report are sourced from official SEC filings, company
communications or widely followed newswires and cross-checked where possible. No
third-party blogs or unverified social media sources were used.
11. Tools, Charts & Support
View CAPR on Finviz (chart & data)
CAPR coverage on Seeking Alpha
Support Merlintrader trading Blog via PayPal
External links lead to third-party platforms for charts, fundamental data and community discussion. Always verify information across multiple primary sources before making any financial decision.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


