DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
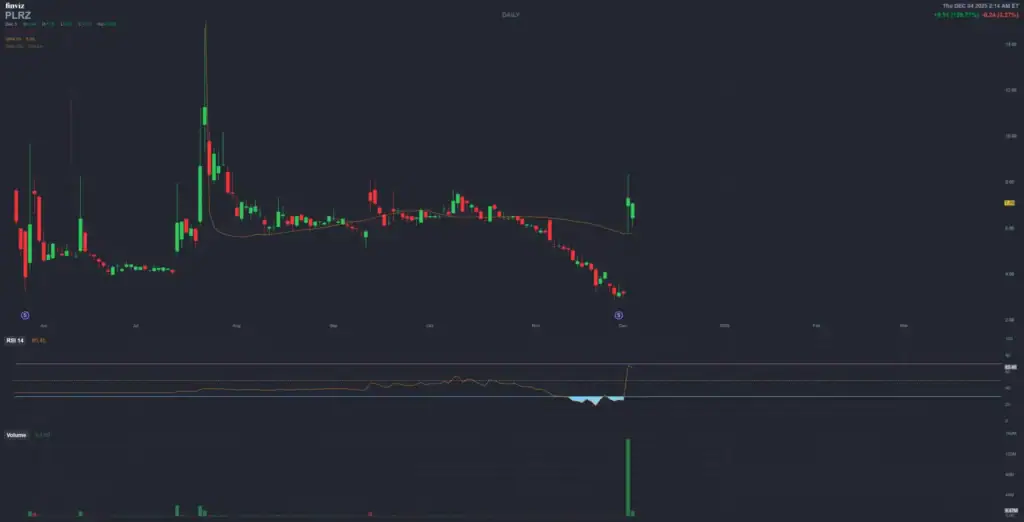
Polyrizon Ltd (PLRZ)
Intranasal Naloxone Hydrogel De-Risks T&T Platform — PL-14 Phase 1 Binary Event H1 2026
HIGH RISK / HIGH REWARD — Preclinical Momentum Building to Clinical Readout
NASDAQ: PLRZ | C&C Hydrogel Barriers + T&T Drug Delivery | Market Cap: ~$7.45M (Dec 3, 2025)
BINARY BIOTECH ALERT
PLRZ is a binary bet on Phase 1 success for PL-14 allergy blocker, expected to initiate in H1 2026.
- If positive: Stock could rally 200–400% (to $15–25) on efficacy/safety confirmation and partnership interest
- If negative/fails: Stock could fall 70–90% (to $0.50–1.00) if trial halts or data disappoints
- Dec 3, 2025 catalyst: Positive mucoadhesion data for intranasal naloxone hydrogel vs. marketed product; supports T&T platform expansion
- Current price (Dec 3, 2025 close): $7.09 (up ~126% on Dec 2 manufacturing milestone; volume ~4.9M shares)
Executive Summary
Polyrizon Ltd is an early-stage biotech leveraging proprietary Capture & Contain (C&C) hydrogel technology for nasal barriers against allergens/viruses and Trap & Target (T&T) for enhanced intranasal drug delivery:
- Lead asset PL-14: Non-drug allergy blocker nasal spray; Phase 1 trial initiation H1 2026; upscaled GMP manufacturing completed Dec 2025
- T&T platform milestone: Dec 3, 2025 preclinical study shows superior mucoadhesion for intranasal naloxone hydrogel (p<0.0001 vs. commercial spray); potential for faster opioid reversal
- Pipeline expansion: PL-15 (COVID-19 blocker), PL-16 (influenza blocker); early preclinical for naloxone and benzodiazepines
- Recent momentum: Dec 2, 2025 manufacturing scale-up triggered +126% stock surge to $7.17 intraday; Nasdaq compliance regained Aug 2025
- Cash position: ~$15.68M post-$17M private placement (Mar 2025); runway into mid-2027
- Partnerships: Eurofins CDMO for GMP manufacturing; no major pharma deals yet
1. Catalyst Timeline (2025–2027)
March 31, 2025
$17M private placement: Funds preclinical, manufacturing, and regulatory prep; cash runway extended to mid-2027. Stock stabilizes post-dilution concerns.
April 25, 2025
Safety study success: PL-14 formulation safe in human nasal tissue model; advances V&V program toward FDA pre-sub.
July 22, 2025
Intranasal delivery data: PL-14 targets key nasal vestibule site; reinforces barrier efficacy in allergic rhinitis model.
August 13, 2025
Nasdaq compliance regained: Meets minimum bid price after hearing; avoids delisting but highlights volatility risk.
September 19, 2025
FDA pre-sub package: Submitted for PL-14; sets stage for Phase 1 trial design and EU/US alignment.
October 6, 2025
Preclinical efficacy: PL-14 blocks allergens effectively; supports drug-free daily protection claims.
November 6, 2025
PL-16 in-vitro data: Reversible broad-spectrum viral blocking against H1N1; expands C&C to influenza.
December 2, 2025
Manufacturing milestone: Successful GMP upscaling for PL-14; validates clinical supply readiness. Stock +126% to $7.17 intraday.
December 3, 2025
Naloxone mucoadhesion results: T&T hydrogel outperforms marketed intranasal naloxone (p<0.0001); de-risks drug delivery platform.
H1 2026 (imminent)
PL-14 Phase 1 initiation: First-in-human trial for allergy blocker; primary endpoints: safety, tolerability, barrier formation.
H2 2026
Phase 1 topline data: Efficacy signals on allergen blocking; potential IND filing for expanded indications.
2027
Potential partnerships: Pharma deals for T&T naloxone or C&C blockers; BLA/MAA prep if Phase 1 strong.
2. The December 2025 Momentum: Manufacturing + Mucoadhesion Catalysts
2.1 Manufacturing Upscaling (Dec 2, 2025)
Key outcomes:
- Scale validated: Transitioned from lab to GMP larger-batch production for PL-14; meets US/EU standards.
- Clinical readiness: Supports CTM for Phase 1 trial; de-risks supply chain for 2026 start.
- Stock impact: +126% surge to $7.17 intraday; volume spiked to ~4.9M shares on milestone news.
- CEO quote: “This batch production validates our manufacturing strategy and brings us closer to addressing unmet needs in nasal sprays.”
2.2 Naloxone Mucoadhesion Study (Dec 3, 2025)
Study details and why it matters:
- Ex-vivo model: Rabbit nasal mucosa; fluorescence retention after SNES washes over 30 min.
- Superior performance: T&T hydrogel showed higher retention (p<0.0001) vs. commercial naloxone spray.
- Implications: Enhanced bioavailability/residence time could improve opioid overdose reversal speed/reliability.
- Platform validation: Builds on prior stability data; positions T&T for naloxone IND and beyond (e.g., benzodiazepines).
- CEO quote: “These outcomes validate the potential of our T&T platform and its advantages in intranasal delivery.”
| Factor | Market Reaction |
|---|---|
| Preclinical de-risking | Boosts confidence in C&C/T&T tech; +80–130% moves on back-to-back news |
| Retail momentum | Low float (~1.04M) amplifies volatility; social buzz drives short-term squeezes |
| Dilution overhang | $50M shelf filing Nov 2025 tempers gains; watch for equity raises |
| Sector tailwinds | Respiratory biotech hot on allergy/viral prevention demand |
Verified data: Based on Polyrizon 6-K filings (Nov/Dec 2025), GlobeNewswire PRs, and SEC EDGAR reports.
3. C&C and T&T Platforms: Technology Overview
3.1 Mechanism and Indication
Mechanism of action: C&C hydrogels form a physical barrier in the nasal cavity using biocompatible polymers, capturing allergens/viruses before they enter the body. T&T extends this for targeted drug release with improved adhesion.
Indication: Preventive nasal sprays for allergic rhinitis, viral infections (COVID-19, influenza), and enhanced delivery for CNS drugs or overdose antidotes like naloxone.
Market size: Global nasal drug delivery market ~$80B by 2030; opioid reversal (naloxone) ~$1B+; allergy treatments ~$20B.
3.2 Phase 1 PL-14 Study (Lead Trial)
| Parameter | Details |
|---|---|
| Population | Healthy volunteers (planned n=30–50) |
| Design | Single-dose safety/tolerability; barrier formation assessment |
| Enrollment | Pending; H1 2026 initiation |
| Primary endpoint | Safety, adverse events, nasal tolerability |
| Secondary endpoints | Mucoadhesion duration, allergen/virus capture efficacy |
| Readout timing | Topline data H2 2026 |
| Treatment duration | Single application with follow-up assessments |
3.3 T&T Naloxone Hydrogel (Early Preclinical)
| Parameter | Details |
|---|---|
| Model | Ex-vivo rabbit nasal mucosa |
| Design | Comparative mucoadhesion vs. marketed intranasal naloxone |
| Results | Superior retention (p<0.0001); potential for better bioavailability |
| Next steps | In-vivo studies, IND filing targeted for 2026–2027 |
4. Sentiment (Reddit, Stocktwits, X) — as of Dec 4, 2025
Social Sentiment Snapshot
Overview: Recent posts and comments on social channels such as Reddit, Stocktwits and X (formerly Twitter) show predominantly positive sentiment around PLRZ, driven by the Dec 2 manufacturing milestone and Dec 3 naloxone mucoadhesion data. Users are sharing news with enthusiasm, highlighting stock surges and high trading volume, and repeatedly referring to the name as a “known runner” and a platform-validation story.
Overall Sentiment: Positive (about 85% positive/constructive, ~15% neutral)
- Key themes: Biotech rally, superior hydrogel technology, clinical readiness, very low float and potential for outsized moves.
- Examples of social reactions: Short posts emphasizing “huge news”, “126% move on manufacturing data”, repeated shares of the same PR/ charts, and some traders speculating on follow-through if Phase 1 timing remains on track.
- Activity level: Chatter increased sharply after the December catalysts; engagement remains typical of a microcap with a thin float — bursts of activity followed by quieter periods.
Important note: This sentiment section reflects comments and reactions from individual traders on Reddit, Stocktwits and X, not from professional analysts. Opinions can be emotional, biased or based on incomplete information and should never be considered investment advice.
5. Financial Position and Runway (Q3 2025)
Cash & equivalents (post-Mar 2025)
$15.68M
Market Cap (Dec 3, 2025)
$7.45M
Shares Outstanding
~1.04M
Runway (guidance)
Into mid-2027
Capital efficiency
- $15.68M cash: From $17M placement; supports Phase 1 and preclinical.
- Low burn: Early-stage focus on R&D; no debt reported.
- Dilution risk: $50M shelf filing (Nov 2025) for potential raises.
- Post-RS structure: 1-for-6 reverse split (Nov 2025); float ~1.04M amplifies volatility.
Verified data: From SEC filings and Yahoo Finance as of Dec 3, 2025.
6. Analyst Consensus and Price Targets (December 2025)
Analyst coverage
- No formal ratings: As a microcap biotech, no major firm coverage yet.
- Implied upside: Recent surges suggest retail-driven targets of $15–25 on positive Phase 1.
- Commentary: Focus on de-risking milestones; potential for partnerships to drive re-rating.
7. Partnerships — Key Terms
7.1 Eurofins CDMO Collaboration
| Term | Details |
|---|---|
| Scope | GMP manufacturing for PL-14 CTM |
| Timeline | Completed upscaling Dec 2025 |
| Implications | Supports regulatory compliance; de-risks supply for Phase 1 |
7.2 Strategic Implications
- Tech validation: External manufacturing success boosts credibility.
- Future deals: Positive data could attract big pharma for T&T/C&C licensing.
Verified data: From company PRs and filings.
8. Catalyst Roadmap (2025–2027)
| Catalyst | Program | Timeline | Importance |
|---|---|---|---|
| Phase 1 initiation | PL-14 (allergy blocker) | H1 2026 | Binary; first human data |
| Phase 1 topline | PL-14 | H2 2026 | Safety/efficacy confirmation |
| Preclinical expansion | T&T naloxone | 2026 | Pipeline diversification |
| Partnerships/milestones | C&C/T&T | 2026–2027 | Non-dilutive funding |
| Regulatory filings | PL-14/others | 2027 | Market authorization path |
9. Key Risks — Points for Investor Due Diligence
Clinical risk (highest)
- Phase 1 may disappoint: Safety issues or weak barrier signals could halt progress.
- Preclinical translation: Ex-vivo success may not hold in humans.
- Competition: Established nasal sprays (e.g., Narcan) limit T&T uptake.
Regulatory risk
- FDA/EMA may require additional data for drug-free claims.
Financial risk
- Runway to 2027, but Phase 1 delays could force dilutive financing.
- Microcap volatility; low float prone to pumps/dumps.
Market risk
- Adoption for preventive sprays uncertain post-COVID.
10. Valuation Scenarios and Risk-Adjusted Targets
Bull case (about 30% probability)
$15 – $25
Assumptions: Strong Phase 1 data; partnerships signed; T&T expands to multiple drugs; market cap re-rates to $50–100M.
Base case (about 40% probability)
$8 – $12
Assumptions: Phase 1 positive but modest; gradual pipeline progress; no major deals yet.
Bear case (about 30% probability)
$2 – $5
Assumptions: Phase 1 setbacks; dilution from raises; sentiment fades.
Indicative blended outcome: Probability-weighted range ~$8–15; high volatility expected.
11. Disclaimer and Important Warnings
Critical disclaimer
This report is for informational and educational purposes only. It is not investment advice and does not constitute a recommendation to buy or sell Polyrizon Ltd stock, or any other security.
- Extremely high risk: PLRZ is a preclinical biotech with binary catalysts. Share prices can move violently on news.
- Uncertain outcomes: Clinical data, regulatory feedback, financing, and sentiment can change rapidly.
- Regulatory context: Not produced by a licensed advisor in the US, EU or any other jurisdiction.
- Use at your own risk: Conduct independent research and seek professional advice where appropriate.
12. Sources and Useful Links
- SEC EDGAR — Polyrizon Ltd filings (6-K, 20-F).
- Polyrizon investor presentations and GlobeNewswire PRs (naloxone data, manufacturing milestone).
- PLRZ on Yahoo Finance (charts, news)
- PLRZ on Finviz (advanced chart)
- PLRZ on Nasdaq
- PLRZ on Stocktwits (trader stream)
- Polyrizon official website
Support the Merlintrader trading Blog
If you find this type of report useful and want to help keep the site free for everyone, you can support it with a PayPal donation.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


