DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Executive Summary
Sana Biotechnology is a high-risk, high-reward clinical-stage cell therapy company. The market focus has increasingly converged on whether its “hypoimmune” engineering can enable durable allogeneic cell therapies with reduced immune rejection and, in the long run, less reliance on chronic immunosuppression. The company has also executed a strategic refocus to prioritize fewer programs and reduce burn, which materially impacts runway and dilution expectations.
The most important “hard anchors” for this story are: (1) peer-reviewed and/or officially disclosed clinical evidence from the investigator-sponsored UP421 program, (2) program timelines and scope for prioritized assets (diabetes and autoimmune), and (3) the cash runway implied by the latest quarterly disclosures.
Verified anchors (primary sources)
- Q3 2025 cash and investments: Sana reported $153.1M in cash, cash equivalents and marketable securities as of Sep 30, 2025. (Company Q3 2025 results release / SEC-linked disclosure)
- Q3 2025 operating profile: Sana reported Q3 2025 R&D expense of $30.1M and G&A expense of $10.3M, reflecting lower spending after pipeline focus and restructuring. (Company Q3 2025 results release)
- Clinical evidence base: UP421 results and follow-ups have been disclosed in company updates, including a peer-reviewed publication in a major medical journal related to the “hypoimmune” islet cell approach. (Company update + journal link below)
Quick Data (aligned to filings and reputable quote pages)
| Item | Snapshot |
|---|---|
| Shares outstanding | 266.37M shares outstanding (reported in the company’s Q3 2025 Form 10-Q cover page / filing copy hosted by IR). |
| Cash + equivalents + marketable securities | $153.1M as of Sep 30, 2025 (company Q3 2025 results release). |
| Q3 2025 expenses |
R&D: $30.1M; G&A: $10.3M (company Q3 2025 results release). Note: some third-party summaries cite slightly different “operating expense totals”; for verification, the company release and the 10-Q are the definitive references. |
| 52-week range (reported by company quote page) | High: $7.30 | Low: $1.26 (company IR quote page). |
| Performance context (math on linked closes) | Using Jan 2, 2025 close $1.65 and Dec 12, 2025 close $5.05, implied YTD change is approximately +206.06%. Distance to 52-week high: -30.82%; distance to 52-week low: +300.79%. |
| Implied market cap (approx.) | $5.05 × 266.37M shares ≈ $1.35B (math shown; see sources section for linked quotes and filing). |
Company Overview and Proprietary Technology
Sana Biotechnology is building engineered cells as medicines. Its foundational platform is commonly described as “hypoimmune” engineering: a set of genetic modifications intended to reduce immune recognition and clearance of therapeutic cells. The investment debate is less about whether cell therapy works in principle, and more about whether Sana can demonstrate durable, scalable, and clinically meaningful outcomes in diabetes and autoimmune disease settings, under realistic safety constraints.
How the Hypoimmune (HIP) platform is described
- Goal: engineered cells that may better evade immune rejection, potentially reducing the need for long-term immunosuppression (conceptual objective; clinical reality depends on data and labels).
- Mechanistic framing (high level): modifications that reduce adaptive immune recognition plus signals that reduce innate immune clearance. (Detailed gene-level edits vary by program and disclosure.)
- Cross-program relevance: this platform concept is intended to be applicable to multiple cellular modalities (e.g., islet cell replacement, CAR-T variants, and other engineered cell approaches), but maturity differs by program.
Clinical and Preclinical Pipeline (structured and complete)
Prioritized programs (continuing)
| Program | Indication / Theme | Stage | Key notes and what matters |
|---|---|---|---|
| UP421 | Type 1 diabetes islet cell replacement (investigator-sponsored) | Clinical (investigator-sponsored) | Proof-of-concept relevance for hypoimmune islet cell approach. Public disclosures include follow-up data and a peer-reviewed publication link in the sources section. |
| SC451 | Stem cell-derived beta cells for Type 1 diabetes | Preclinical (IND-enabling) | Often framed as the scalable evolution beyond primary islets. Company communications point to IND-enabling work with an anticipated IND timeframe around 2026 (timing is subject to updates). |
| SC291 | B-cell mediated autoimmune (e.g., lupus / vasculitis concepts referenced in company materials) | Clinical development (autoimmune focus) | Program focus has shifted versus earlier oncology positioning; the key questions are durability, safety, and whether “immune reset” narratives translate into measurable, sustained outcomes. |
| SC262 | CD22 allogeneic CAR-T (historically studied in B-cell malignancies) | Clinical (Phase 1) | Often discussed as a follow-on/adjacent logic to CD19 targeting; clinical readouts and safety context are the primary drivers, and timelines can change. |
| SG299 | In vivo CAR-T concept using a “fusogen” delivery framing | Preclinical | Strategic optionality; also widely recognized as a competitive arena with multiple players. Relative positioning depends on who reaches human data first and what safety/efficacy look like. |
Paused or discontinued programs (historical completeness)
- Oncology reprioritization: the company announced a strategic pipeline focus (late 2024) that deprioritized certain oncology programs to concentrate resources on diabetes and autoimmune priorities.
- SC379 and other CNS-related efforts: company communications have discussed narrowing focus; some programs may be terminated, partnered, or otherwise restructured over time.
Clinical Evidence and Data Context (what is known, what remains uncertain)
UP421 (Type 1 diabetes) — disclosed follow-ups and peer-reviewed context
- Public company updates have described follow-up observations including insulin production markers (e.g., C-peptide) and imaging-based evidence consistent with graft activity in the investigator-sponsored setting.
- A peer-reviewed publication exists related to the hypoimmune islet cell approach (linked in Sources). Peer-review does not eliminate risk, but it improves transparency and technical scrutiny versus “headline only” coverage.
- Key limitation: small numbers and early follow-ups are not the same as broad, durable efficacy in larger trials. Interpretation should stay anchored to what is disclosed and how it generalizes (or does not).
SC451 (Type 1 diabetes) — why it is considered the “scalable” pillar
- The strategic logic is manufacturing scalability: stem cell-derived beta cells can, in principle, reduce dependence on donor islets.
- The timeline is commonly framed around an IND in 2026, but this is a moving target dependent on IND-enabling progress, manufacturing, and regulators.
Autoimmune programs (SC291) and CAR-T themes
- The autoimmune narrative often centers on deep B-cell depletion and an “immune reset” concept; the decisive proof is clinical durability and safety in defined autoimmune populations.
- Competitive context matters: other players are advancing both ex vivo and in vivo approaches, and the market can reprice quickly on peer readouts.
Financial Analysis (primary disclosure alignment)
Cash position and runway framing
As of Sep 30, 2025, Sana reported $153.1M in cash, cash equivalents and marketable securities. The company also described a financing path during 2025 that included an underwritten offering and ATM usage, and it guided to runway of at least one year from the filing timeframe (subject to spending and market access).
| Metric (Q3 2025 context) | Value | Why it matters |
|---|---|---|
| Cash + equivalents + marketable securities | $153.1M | Liquidity buffer; directly impacts dilution risk and operational flexibility. |
| R&D expense (Q3 2025) | $30.1M | Signals the level of active development work after strategic focus. |
| G&A expense (Q3 2025) | $10.3M | Corporate scale costs; matters as the company prepares for more complex execution. |
| Net loss (Q3 2025) | $42.2M | Baseline loss level; not unusual for pre-revenue biotech but relevant for runway math. |
| Financing actions in 2025 (company disclosed) | ~$109.7M net proceeds | Confirms the company’s access to capital; also expands share count over time. |
Runway math is sensitive to spending. “Straight-line” runway estimates are only heuristics: burn can rise if programs expand, manufacturing scales, or trial designs change. The authoritative anchor is the company’s latest guidance and subsequent filings.
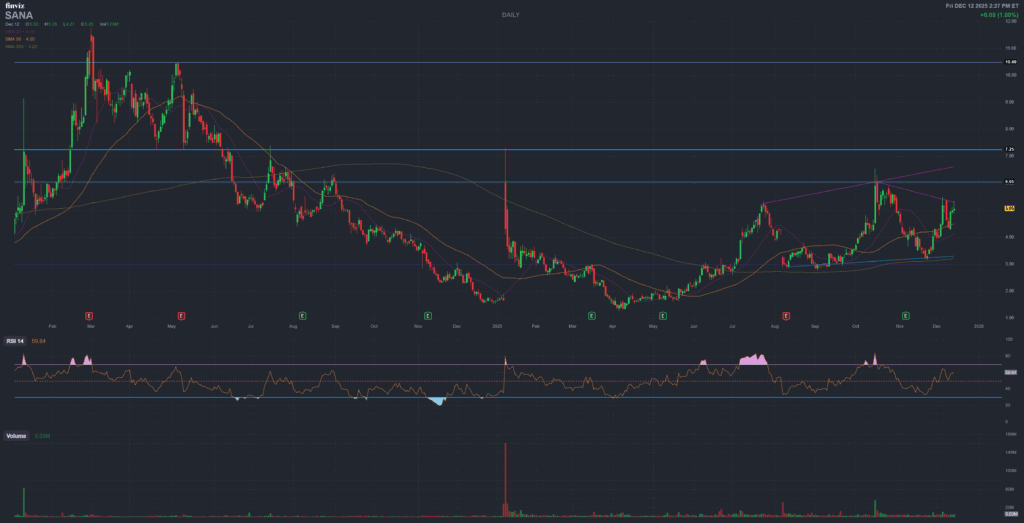
Stock Performance and Market Context
- Close used for this update: $5.05 (Dec 12, 2025).
- 52-week range (company quote page): $7.30 high and $1.26 low.
- Implied YTD (linked closes): approximately +206.06% (Jan 2 close $1.65 to Dec 12 close $5.05).
- Share count sensitivity: market cap and per-share math depend on the most recent share count in filings.
Analyst Coverage and Price Targets (opinion-based; linked)
Analyst ratings and targets are opinions that can change quickly. The most defensible way to use them is as a map of assumptions (timeline, differentiation, financing path, adoption, and risk tolerance), not as a forecast.
| Source / Firm | Rating (reported) | Price target (reported) | Date (reported) | Link |
|---|---|---|---|---|
| Citizens JMP (reported via Nasdaq) | Market Outperform | $8.00 | Oct 30, 2025 | Nasdaq |
| Wedbush (reported) | Outperform | $6.00 | Nov 7, 2025 | TipRanks / The Fly |
| H.C. Wainwright (reported) | Buy | $9.00 (from $11) | Nov 7, 2025 | TipRanks / The Fly |
| Morgan Stanley (reported) | Overweight | $12.00 | Jul 3, 2025 | Morgan Stanley via TipRanks/TheFly |
Sentiment and Media Framing (not evidence; linked where possible)
- News framing: tends to focus on diabetes “functional cure” narratives, hypoimmune differentiation, and runway/dilution risk.
- Retail discussion patterns: often rotate around “platform validation,” “partnership or M&A speculation,” and “funding overhang.” These are sentiment signals only and should be treated as mood, not proof.
- Practical constraint: sentiment can flip on peer readouts, financing headlines, or a single safety signal.
Key Catalysts (2025–2026, structured)
Potential value catalysts
Program milestones- UP421 follow-up disclosures (timeline depends on investigators and publication cadence).
- SC291 autoimmune clinical progress and any data disclosures.
- IND-enabling progress for SC451 with an anticipated IND timeframe around 2026 (subject to updates).
Neutral/ongoing items
Execution- Conference presentations (visibility events; content matters more than attendance).
- Enrollment updates where disclosed (timelines can shift).
- Manufacturing scale and CMC readiness for future INDs.
High-impact risk catalysts
Runway / legal- Financing decisions and market access (dilution sensitivity).
- Safety surprises in cellular therapies (can reprice the entire platform narrative).
- Ongoing litigation headlines (outcomes are uncertain; see Sources).
SEC Filings and Investor Relations (verification-first)
- Q3 2025 Form 10-Q (Sep 30, 2025): cash position, expense lines, share count, and runway statements.
- Company Q3 2025 results release: business updates, refined focus, and financial summary.
- Company business update and pipeline focus communications (2024–2025): prioritization decisions and program status changes.
Governance and Ownership (verification guardrails)
Institutional ownership and insider activity should be verified using SEC filings (Schedules 13D/13G, 13F, and Form 4). Third-party “ownership dashboards” can lag and can differ depending on methodology.
- Insiders: verify via Form 4 search.
- Institutions: verify via 13F and beneficial ownership filings.
- Short interest: verify using exchange/FINRA short interest publications and align by settlement dates.
Competitive Landscape (high-level mapping)
Type 1 diabetes cell therapy
- Vertex: advanced clinical efforts in islet replacement are often treated as the key “bar setter” for the space.
- Other approaches: encapsulation devices, alternative stem-cell derived beta cell strategies, and immune-modulating adjunct approaches.
- Sana differentiation claim: hypoimmune engineering is positioned as a potential way to reduce immune rejection; the decisive proof remains clinical durability and safety at scale.
Allogeneic and in vivo CAR-T themes
- In vivo delivery and fusogen-style concepts are competitive and fast-moving; first-in-human timing and safety profile matter more than preclinical “power” claims.
- Comparative positioning is fluid; peer readouts can re-rate expectations quickly.
Valuation Discussion (what is implied, what must happen)
- Market cap anchor: approximately $1.35B using $5.05 and 266.37M shares.
- Enterprise value context: EV depends on cash and any debt; see linked statistics pages for a current EV estimate.
- Interpretation: valuation is sensitive to the probability that diabetes and/or autoimmune programs become durable, scalable clinical assets before financing pressure dominates.
Key Risks (plain language, complete)
Financial risks
- Runway constraints: with $153.1M as of Sep 30, 2025 and ongoing operating losses, financing strategy is a central variable.
- Dilution risk: future equity raises can materially dilute existing holders.
- No commercial revenue base: typical for clinical biotech, but it makes financing cycles unavoidable.
Clinical and technical risks
- Early data limitations: small patient counts and early follow-ups can fail to generalize.
- Long-term safety uncertainty: engineered cellular therapies require long observation and robust safety databases.
- Manufacturing scale risk: transitioning from promising biology to reliable large-scale manufacturing is a common failure point.
Legal and reputational risks
- Securities litigation headlines: allegations and outcomes should be treated cautiously; the only reliable truth is in court records and company filings. Public law firm notices exist and are linked below for completeness.
Competitive risks
- Peer timelines can reset expectations abruptly, especially in diabetes cell therapy and CAR-T delivery themes.
Opportunities (what could create upside in a non-promotional way)
Scientific opportunities
- Platform validation: if hypoimmune engineering continues to show durable function with acceptable safety, it can re-rate platform probability.
- Broader applicability: a proven immune-evasion toolkit could be leveraged across multiple cell modalities.
Market opportunities
- Large unmet need: Type 1 diabetes remains a major global disease with high lifetime burden; durable cell-based functional solutions would be meaningful if validated.
- Autoimmune: certain B-cell mediated autoimmune diseases can support substantial value creation if durable remission-like outcomes are demonstrated.
Strategic opportunities
- Partnership optionality: partnerships can extend runway and add external validation, but should be treated as hypothetical until announced.
- M&A speculation: common in platform biotechs; not evidence unless supported by official communications.
Bull / Base / Bear Framework (educational, non-advisory)
Bull case (what would need to be true)
Illustrative- UP421-style signals prove durable and reproducible in broader settings.
- SC451 advances cleanly through IND-enabling milestones toward an IND timeframe around 2026.
- Autoimmune program execution produces meaningful, durable outcomes with acceptable safety.
- Financing is secured on terms that do not overwhelm the equity story.
Base case (what “normal” could look like)
Illustrative- Stock trades primarily on runway updates, financing terms, and intermittent milestone disclosures.
- Diabetes narrative remains “promising but early” until larger-scale evidence and durable follow-up accumulate.
- Program scope remains narrowed to preserve cash and maximize probability of one or two wins.
Bear case (what breaks the story)
Illustrative- Clinical durability or safety signals disappoint or do not replicate.
- Runway compresses faster than expected and financing becomes punitive.
- Competitive readouts raise the bar beyond what Sana can realistically match on timelines or outcomes.
How “rating labels” and “trading plans” appear in market commentary (context only)
The following items are included because they were present in the original research draft and are frequently seen in retail market commentary. They are not recommendations, not personalized, and not instructions. They are simply examples of how some commentary describes risk framing.
| Commentary pattern | Example phrasing seen in drafts/retail commentary | Why it exists |
|---|---|---|
| Labeling stance | “Cautious accumulate” (draft label) | A shorthand to express “high uncertainty + perceived upside” in informal commentary. |
| Range framing | Entry zone examples: $4.50–$5.50 (draft) | Downside trigger examples: $3.80 (draft) | Commentary often uses simple price anchors; this does not make them correct or appropriate. |
| Position sizing talk | “Max 3–5% position” (draft) | Retail risk language; real sizing depends on the individual and should not be copied. |
Monitoring Checklist (non-advisory)
- Cash and runway: quarterly cash position, cash used in operations, and any updated runway guidance.
- Financing terms: pricing, structure, and timing of any capital raises.
- Clinical updates: UP421 follow-ups, SC291 progress disclosures, and SC451 IND-enabling milestones.
- Safety narrative: any adverse event disclosures and how they compare to expectations for cellular therapies.
- Competitive readouts: diabetes cell therapy and autoimmune peers can re-rate assumptions quickly.
- Legal headlines: track only via reliable documents and official statements where possible.
Chart image source: Finviz (image load without affiliate parameters). Clickable Finviz links are provided in Tools & Partners.
Sources (primary first; then market/coverage; then legal notices)
- Company Q3 2025 results release (financials and update): GlobeNewswire link
- Company Form 10-Q (Q3 2025) copy hosted by IR (share count and filing-level detail): 10-Q PDF copy
- Company business update (Q2 2025; pipeline focus details): GlobeNewswire link
- UP421 follow-up and NEJM publication context (company update): GlobeNewswire link
- Peer-reviewed publication link (NEJM, as referenced in SEC/company disclosures): SEC Exhibit link
- Company strategic pipeline focus (historical, late 2024): GlobeNewswire link
- Company quote page (52-week range reference): IR quote page
- Historical price reference (Jan 2, 2025 close used for YTD math): Yahoo Finance history
- Analyst note links (reported): Citizens JMP via Nasdaq | Wedbush via TipRanks/TheFly | H.C. Wainwright via TipRanks/TheFly | Morgan Stanley via TipRanks/TheFly
- Litigation notices (for completeness; allegations only): Pomerantz notice | Rosen notice
Disclaimers (SEC / CONSOB / Affiliate) — EN + IT
EN: Educational and informational content only. This report reflects a personal interpretation of publicly available information and does not constitute investment advice, research, a recommendation, an offer, or a solicitation to buy or sell any security. Cell therapy and biotech stocks are volatile; clinical outcomes are uncertain; financing risk can be material. Always verify using primary sources (SEC filings, official company communications, regulators, peer-reviewed publications where applicable) and assess risks independently.
IT: Contenuto esclusivamente informativo ed educativo. Questo report rappresenta un’interpretazione personale di informazioni pubbliche e non costituisce consulenza finanziaria, ricerca, raccomandazione, offerta o sollecitazione all’acquisto/vendita di strumenti finanziari. Le biotech e le terapie cellulari sono altamente volatili; gli esiti clinici sono incerti; il rischio di finanziamento/diluizione può essere significativo. Verificare sempre tramite fonti primarie (filing SEC, comunicazioni ufficiali della società, autorità regolatorie, pubblicazioni peer-review quando applicabile) e valutare i rischi in autonomia.
Legal pages: Disclaimer | Terms & Privacy
Author Note
Navigation
Executive Summary
Quick Data
Company Overview
Hypoimmune Platform
Pipeline
Clinical Evidence
Financial Analysis
Stock Context
Analyst Coverage
Sentiment
Catalysts
SEC/IR
Governance
Competition
Valuation
Risks
Opportunities
Bull/Base/Bear
Market Commentary
Monitoring
Sources
Disclaimers
Author Note
Fast Links
Interpretation Guardrails
This page separates primary disclosures (SEC/company/journal) from opinion-based items (targets, sentiment). Any “scenario” language is illustrative only and is not a forecast.
Tools & Partners
Finviz
Charts, fundamentals, insider/institutional snapshots, quick market view.
ChartsWatcher
ChartsWatcher is a real-time, next-generation scanner for the US stock market.
Seeking Alpha
News flow, earnings coverage, analysis and investor debate.
Stocktwits
Retail sentiment stream and ticker-based discussions.
Medved Trader
Fast desktop platform for charts, scanning, alerts and automation (friend link; no referral).
Merlintrader trading Blog
Dashboards, calendars, reports and educational deep dives.
Affiliate note: some links above may be affiliate/referral links. If used, they may generate a commission that helps support the site at no extra cost.
Support the Project

Donate via PayPal
Biotech Catalyst Calendar
Track upcoming biotech catalysts in one place: open the calendar here.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


