DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
EKSO Ekso Bionics Holdings Inc

On December 29, 2025, Applied Digital (APLD) and Ekso Bionics (EKSO) announced a non-binding term sheet for a proposed business combination between Applied Digital’s cloud computing arm, Applied Digital Cloud, and EKSO.
PALI Palisade Bio Inc
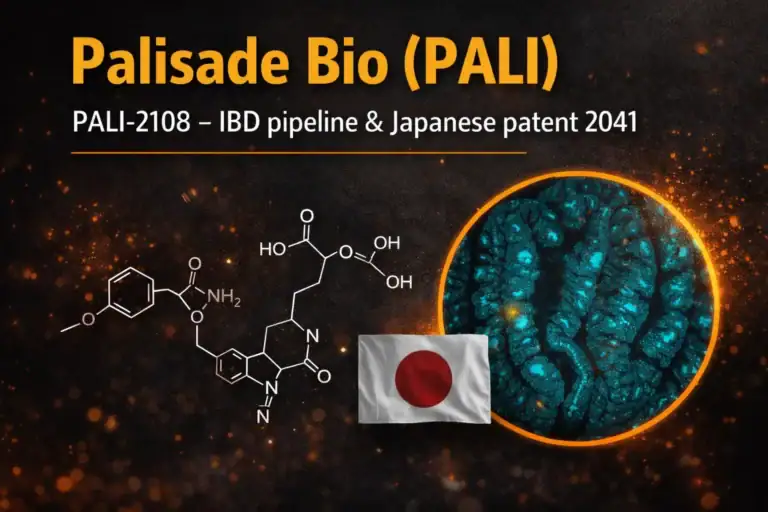
The Japan Patent Office has granted a patent titled “Gut Microbiota-Activated PDE4 Inhibitor Prodrug” covering the composition of matter for PALI-2108, Palisade Bio’s lead gut-restricted PDE4 B/D inhibitor for moderate-to-severe ulcerative colitis (UC) and fibrostenotic Crohn’s disease (FSCD). The base patent term runs into 2041 and is eligible for extension based on regulatory review timelines.
ARDX Ardelyx Inc Updated Jan 8

Q3 2025: revenue beat, IBSRELA guidance raised, Street now models ARDX turning profitable in 2026
October–December 2025 – Q3 revenue beat, IBSRELA guidance increased to $270–275M for 2025, XPHOZAH absorbs Medicare shock, analysts talk about 2026 breakeven.
GLSI Greenwich LifeSciences Inc

FLAMINGO-01 open-label arm shows ~80% reduction in breast cancer recurrences; Fast Track in place and lock-up extended to 2026
December 2025 – GLSI reports preliminary ~80% recurrence rate reduction in the 250-patient open-label arm of FLAMINGO-01, completes enrollment, and extends insider/pre-IPO lock-up to September 30, 2026, while the stock breaks to new 52-week highs.
CORT Corcept Therapeutics Inc DEC 31 UPDATE CRL

n March 3, 2025, the FDA filed Corcept’s NDA for relacorilant as a treatment for adult patients with endogenous hypercortisolism (Cushing’s syndrome), assigning a PDUFA target action date of December 30, 2025. The NDA is based on the pivotal Phase 3 GRACE study, confirmatory evidence from Phase 3 GRADIENT, long-term extension data and a Phase 2 study in hypercortisolism.
INO Inovio Pharmaceuticals Inc
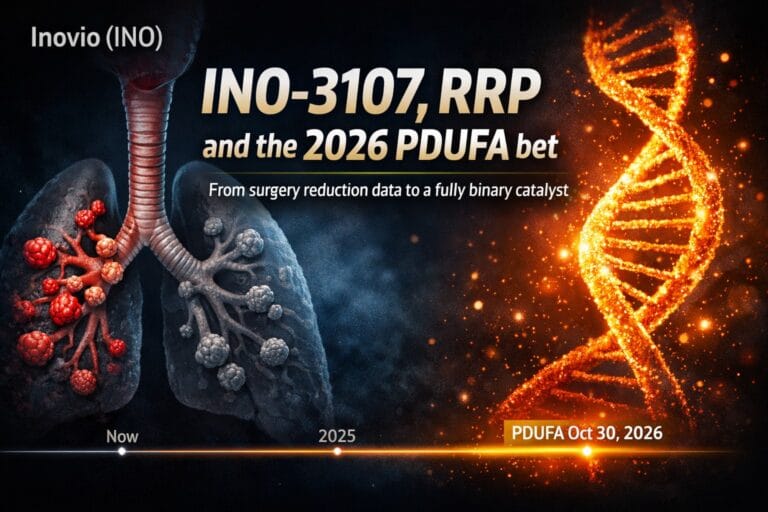
FDA accepts INO-3107 BLA, Standard Review, PDUFA October 30, 2026
On December 29, 2025 the FDA accepted Inovio’s Biologics License Application for INO-3107 as a potential treatment for adults with Recurrent Respiratory Papillomatosis (RRP). The review classification is Standard and the agency set a PDUFA target action date of October 30, 2026. The FDA also indicated that it is currently not planning to hold an Advisory Committee for this dossier.
BTAI BioXcel Therapeutics Inc

On 12 December 2025 BioXcel held its virtual Annual Meeting. Shareholders approved all proposals, including a flexible reverse stock split authorization (from 1-for-5 up to 1-for-30) that the Board may implement within 12 months if needed to keep Nasdaq listing compliance. The company is currently compliant, but this vote gives management an emergency tool in case of further price pressure
MREO Mereo Biopharma Group

On 29 December 2025, Mereo BioPharma announced Phase 3 ORBIT and COSMIC results for setrusumab (UX143) in osteogenesis imperfecta. Neither trial met its primary endpoint of reduced annualised clinical fracture rate versus placebo (ORBIT) or bisphosphonates (COSMIC),
RARE Ultragenyx Pharmaceutical Inc

On 29 December 2025 Ultragenyx announced that the Phase 3 ORBIT and COSMIC studies of setrusumab (UX143) in osteogenesis imperfecta did not meet their primary endpoints of reducing annualized clinical fracture rate versus placebo (ORBIT) or bisphosphonates (COSMIC).
LPCN Lipocine Inc

16 December 2025: Lipocine reports that its Phase 3 trial of LPCN 1154 (oral brexanolone) in postpartum depression has reached 80% enrollment (66 of 80 planned participants). The second independent Data Safety Monitoring Board (DSMB) review is scheduled for mid-January 2026 and topline results are still guided for the second quarter of 2026. The study is intended to support a 505(b)(2) NDA submission referencing IV brexanolone.
TDOC Teladoc Health Inc

Teladoc Health – From pandemic superstar to underpriced cash-flow machine?
Virtual care incumbent trading at about 0.6× revenue and mid-teens implied FCF yield, with a new CEO, a shrinking BetterHelp segment and a slow but steady reset toward profitable integrated care
Big Pharma Goes Shopping – Q4 2025 Biotech M&A Wave and When Retail Fantasy Came True

4 2025 turned into a real M&A season for Big Pharma: multi-billion deals in diagnostics, cardio-metabolic and rare diseases, plus a few cases where retail dreams of a buyout actually became reality. Below you find a structured overview of the main transactions, the strategic themes behind them and the “fantasy came true” section based on frequently mentioned retail ideas on social platforms.
TNGX Tango Therapeutics Inc

Tango Therapeutics (TNGX) – PRMT5, CoREST and a crowded short base into 2026 | Tango Therapeutics (TNGX) – PRMT5, CoREST e una base short affollata verso il 2026 English Italiano Ticker: TNGX (NASDAQ) Tango Therapeutics (TNGX) – PRMT5, CoREST and…
IBRX ImmunityBio Inc

On December 12, 2025, the European Medicines Agency recommended granting a conditional marketing authorization for ANKTIVA (nogapendekin alfa inbakicept) in combination with BCG for adults with BCG-unresponsive non-muscle invasive bladder cancer (NMIBC) with CIS, with or without papillary tumors – effectively mirroring the U.S. label in the EU and leveraging the pivotal QUILT-3.032 data set.
OCGN Ocugen Inc

November 2025 – In its Q3 2025 update, Ocugen reports 1.75 million USD in revenue and a net loss of about 20 million USD, with operating expenses of 19.4 million USD driven mainly by gene therapy R&D. Cash and equivalents stand around 33 million USD after a 20 million USD financing, which management believes funds operations into 2026.
GERN Geron Corp

After decades of telomerase debates and more than one boom-and-bust cycle, Geron finally has an FDA-approved product on the market. Rytelo is generating real revenue, IMpactMF is fully enrolled, and a very loyal retail base is watching every quarterly print and every slide deck, trying to understand whether this is the beginning of a long compounding story or just the payoff of a very long wait.
VKTX Viking Therapeutics Inc
Viking is trying to crash a party dominated by Eli Lilly and Novo Nordisk with a dual GLP-1/GIP agonist, VK2735, in both weekly injectable and daily oral forms. Phase 2 data are eye-catching, Phase 3 VANQUISH trials are fully underway, and the balance sheet is still heavy with cash – but there is real execution, timing and dilution risk.
IOVA Iovance Biotherapeutics Inc

6 November 2025 – Iovance reports Q3 2025 revenue of about $68 million, up 13% sequentially, with Amtagvi driving the growth. Gross margin improves to 43%, but the company still posts a net loss of over $90 million and burns cash.
DCTH Delcath Systems Inc

20 November 2025 – Delcath’s board authorizes a $25 million share repurchase program, at a time when the stock has dropped more than 50% in six months and trades close to its 52-week low around $8 per share. The program can be executed via open-market and privately negotiated transactions, with no fixed end-date, funded from cash and operating cash flow.
BHVN Biohaven Ltd

24 December 2025 – Biohaven reports that BHV-7000 did not meet the primary endpoint in a six-week Phase 2 MDD study (MADRS change vs placebo). Shares fall about 16–17% after hours. Management says it will stop psychiatric trials with BHV-7000 and focus 2026 resources on immunology, obesity and epilepsy.