DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Viking Therapeutics (VKTX) – VK2735, GLP-1/GIP and the Obesity Arms Race
Viking is trying to crash a party dominated by Eli Lilly and Novo Nordisk with a dual GLP-1/GIP agonist, VK2735, in both weekly injectable and daily oral forms. Phase 2 data are eye-catching, Phase 3 VANQUISH trials are fully underway, and the balance sheet is still heavy with cash – but there is real execution, timing and dilution risk.
News of the day
November 2025 – Viking completes enrollment in its 4,650-patient Phase 3 VANQUISH-1 trial for injectable VK2735,
while the oral VENTURE Phase 2 trial reports up to 12.2% mean weight loss at 13 weeks with statistically significant separation versus placebo.
At the same time, the FDA has just approved Novo Nordisk’s once-daily Wegovy oral pill, giving Novo a first-mover advantage in oral GLP-1s.
VKTX sits exactly where volatility loves to live: no approved drugs yet, a lead asset with best-in-class ambitions, a balance sheet that can carry Phase 3 but not an entire commercial build-out forever, and a retail community that treats every dip like a referendum on the GLP-1 future.
1. Quick thesis – where VKTX stands right now
If you want the one-page version before we dive into 2027 endpoints and 2030 EV/Sales math, it’s this:
- Science and early data look real. Subcutaneous VK2735 has shown up to 14.7% mean weight loss at just 13 weeks with no plateau in Phase 2, and the oral tablet has shown up to 12.2% at 13 weeks with strong responder rates.
- The program is de-risked mechanistically. VK2735 is a dual GLP-1/GIP agonist similar in concept to Eli Lilly’s tirzepatide (Mounjaro/Zepbound), which is already a commercial monster.
- But timing is a problem. VANQUISH Phase 3 trials run 78 weeks; with enrollment now done in VANQUISH-1 and ongoing in VANQUISH-2, top-line data aren’t expected before 2027, with potential FDA approval and launch around 2028–2029 if all goes well.
- Competition is brutal. Novo now has an FDA-approved Wegovy pill; Lilly dominates injectable market share; Pfizer just paid up to $10B for Metsera and its MET-097i once-monthly GLP-1 with 14.1% placebo-adjusted weight loss at 28 weeks.
- Valuation is “cheap for success, rich for failure.” With an enterprise value around $3.5B and analyst models that see multi-billion revenue potential by 2030–2031, VKTX trades somewhere near 1–2× out-year sales depending on which forecast you use – a deep discount to LLY, NVO and the price PFE just paid for Metsera.
- Balance sheet is solid but not infinite. $715M in cash and investments as of Q3 2025 easily fund VANQUISH, but not a full global launch plus combination programs forever. Dilution risk later in the decade is real.
- Retail culture is a feature, not a bug. VKTX has a hyper-engaged fanbase on Reddit and Stocktwits, plus >20% of float sold short, which makes the stock behave more like a swing-trade instrument than a sleepy biotech – think multi-week squeezes and 30–40% gaps on news.
Net: VKTX looks attractive as a high-beta GLP-1/GIP options ticket on 2027–2029 obesity data, not as a low-vol, sleep-well-at-night hold. Whether that’s “Buy” or “Too spicy” depends entirely on your risk tolerance and time horizon.
2. Timeline – from anonymous small cap to GLP-1 cult stock
VKTX was a quiet NASH / thyroid-beta story before it rediscovered itself as a pure-play GLP-1/GIP platform.
2.1 Before the GLP-1 hype
2015–2020
Early Viking – NASH and rare diseases
Viking’s first wave focused on VK2809 (thyroid-beta agonist) in NASH / NAFLD and VK0214 in X-linked adrenoleukodystrophy,
with multiple Phase 2 studies delivering encouraging lipid and liver-fat reductions.
The market largely ignored the story while bigger NASH names imploded one by one.
2021–2023
Dual GLP-1/GIP appears
VK2735, a dual GLP-1/GIP agonist similar in concept to tirzepatide, enters early clinical development in injectable form.
Initial Phase 1 data show up to ~8% weight loss after just 28 days of weekly injections, with tolerable GI side-effects,
putting Viking on the GLP-1 map for the first time.
2.2 2024–2025 – From quiet success to volatility magnet
Feb 2024
Subcutaneous VENTURE Phase 2
Viking reports that weekly injectable VK2735 in obesity achieves up to 14.7% mean weight loss after 13 weeks in the VENTURE study,
with high proportions of patients crossing the 10% threshold and no plateau visible at the end of treatment.
Social media starts calling VKTX “mini-Lilly” and the share price goes vertical.
Jun 2025
VANQUISH Phase 3 program launched
Viking initiates VANQUISH-1 (obesity without diabetes, ~4,500+ patients) and VANQUISH-2 (obesity + T2D, ~1,100 patients),
both 78-week weekly injection trials testing three VK2735 doses versus placebo.
Aug 2025
VENTURE-Oral Phase 2 – and an ugly sell-off
Oral VK2735 tablet shows up to 12.2% mean weight loss at 13 weeks; placebo-adjusted effect up to 10.9%,
with 80% of patients achieving at least 10% weight loss.
However, discontinuations from GI side-effects run higher than investors hoped. Reuters, Barron’s and others highlight the dropout rate,
and VKTX drops ~35–40% in a day as fast-money GLP-1 tourists bail.
Oct–Nov 2025
Q3 results, VANQUISH-1 fully enrolled
Q3 2025: no revenue yet, net loss $90.8M, but $715M cash and investments on the balance sheet.
November: Viking announces that VANQUISH-1 reached ~4,650 patients enrolled, exceeding the original 4,500 target –
a sign of strong investigator and patient interest.
Dec 2025
Novo’s Wegovy pill gets FDA approval
On December 22, 2025, the FDA approves Novo Nordisk’s 25 mg once-daily Wegovy pill,
the first oral GLP-1 approved for obesity, with ≈16–17% mean weight loss over 64 weeks.
Traders immediately start asking one question: “By 2028, will there still be room for VKTX’s oral and injectable combos?”
3. Mechanism – what exactly is VK2735?
3.1 Dual GLP-1/GIP agonist 101
VK2735 is a peptide that activates both the GLP-1 receptor and the GIP receptor, aiming to replicate and extend the biology behind tirzepatide:
- GLP-1 decreases appetite, slows gastric emptying, improves glycemic control and drives weight loss – the same mechanism used by semaglutide (Ozempic/Wegovy) and other GLP-1s.
- GIP enhances insulin secretion and may add a complementary metabolic effect when co-activated with GLP-1, as demonstrated by tirzepatide’s superior efficacy versus semaglutide in multiple trials.
- Preclinical and early clinical work suggests dual agonists can deliver faster and deeper weight loss than GLP-1 alone, at the cost of similar class-typical GI side-effects (nausea, vomiting, diarrhea).
Conceptually, VK2735 is trying to be “tirzepatide-like” but with both injectable and tablet formulations built from the same core molecule, which may simplify switch-overs between weekly injections and low-dose oral maintenance.
3.2 How the program is structured
- Subcutaneous VK2735 – once-weekly injection; now in Phase 3 (VANQUISH-1 and VANQUISH-2) for obesity with and without Type 2 diabetes.
- Oral VK2735 – once-daily tablet; Phase 2 VENTURE trial met primary and secondary endpoints (up to 12.2% mean loss at 13 weeks).
- Maintenance concept – Viking is already running a trial exploring whether patients can transition from high-dose VK2735 (injectable or oral) to low oral doses as a maintenance strategy, building on the exploratory maintenance arm in VENTURE.
That “single molecule / two formulations” strategy is one of the clearest differentiators versus Lilly and Novo, which use different molecules and titration schemes across their portfolios.
4. Clinical data – what VK2735 has shown so far
The market freak-out after the oral read-out hid a simple truth: both injectable and oral VK2735 still look very competitive on raw efficacy, especially on speed of weight loss.
4.1 Subcutaneous VENTURE Phase 2 – 14.7% at 13 weeks
| Endpoint | Injectable VK2735 |
|---|---|
| Mean weight loss at 13 weeks | Up to 14.7% from baseline |
| Placebo-adjusted difference | ≈13.1% in the top dose cohort (per third-party analysis) |
| ≥10% weight loss | Up to ~88% of VK2735 patients vs ~4% placebo (exploratory data) |
| Trajectory | Weight loss curve still trending down at week 13 – no clear plateau |
| Safety | Mostly mild/moderate GI events; discontinuations similar to placebo |
These numbers come from company presentations at ObesityWeek and follow-up analyses. The key point: VK2735’s early slope is steeper than what tirzepatide showed at 12 weeks in SURMOUNT-1, though final 78-week data will be the real comparison.
4.2 Oral VENTURE Phase 2 – 12.2% with drama
| Endpoint | Oral VK2735 |
|---|---|
| Mean weight loss at 13 weeks | Up to 12.2% from baseline |
| Placebo-adjusted effect | Up to 10.9% better than placebo |
| ≥5% weight loss | Up to 97% on VK2735 vs 10% on placebo |
| ≥10% weight loss | Up to 80% on VK2735 vs 5% on placebo |
| Safety / discontinuations | Higher GI-related drop-outs: depending on dose, ~20–38% on drug vs ~18% placebo in media and trial reports |
The numbers above are taken from the company’s August 19, 2025 press release and an Applied Clinical Trials detailed write-up, combined with media coverage from Reuters, MarketWatch and others.
The oral controversy isn’t about efficacy (which is clearly strong at 13 weeks); it’s about
how much GI tolerability and drop-out risk investors are willing to accept in a world
where Wegovy and future orals will have years of real-world data by the time VK2735 hits the market.
5. Competitive landscape – who is VKTX really fighting?
The GLP-1 race is no longer a two-horse matchup between Lilly and Novo. Pfizer’s Metsera deal and a wave of next-gen programs make “third place” a much harder slot to win.
5.1 The incumbents: Lilly and Novo
- Eli Lilly – tirzepatide (Mounjaro/Zepbound) is a dual GLP-1/GIP RA with ~20–22.5% mean weight loss at 72 weeks in SURMOUNT-1. Lilly now controls a dominant share of the US “incretin analog” market and the majority of new prescriptions.
- Novo Nordisk – semaglutide (Ozempic/Wegovy) has slightly lower peak efficacy but a massive installed base, and now a Wegovy pill approved with ~16–17% weight loss at 64–68 weeks.
- Both companies already have global supply chains, payer deals and telehealth partnerships; Wegovy pill will be launched heavily via retail pharmacies and online channels at relatively aggressive pricing.
By 2027, when VK2735 Phase 3 reads out, Lilly and Novo won’t just have first-mover advantage – they’ll have brand loyalty, outcome data (CV benefit, etc.) and dozens of ongoing label expansions.
5.2 The new kid with a giant wallet: Pfizer / Metsera
- Metsera MET-097i – once-weekly and once-monthly GLP-1 RA; Phase 2b VESPER-1 showed placebo-adjusted mean weight loss up to 14.1% at 28 weeks, with some patients losing 26.5% of body weight.
- Pfizer is paying up to ~$10B to buy Metsera, explicitly to get back into obesity after its own danuglipron program was scrapped.
- Sell-side work and media coverage see Metsera’s pipeline at >$5B peak sales across GLP-1 and amylin combo products.
Pfizer’s bet underlines why VK2735 matters: if a GLP-1 program with ~14% at 28 weeks can justify a $7–10B M&A price tag, Viking’s data are clearly “within the conversation.” The issue is more: can Viking either (a) sell itself at a good price, or (b) go it alone without wrecking the cap table.
5.3 Where VK2735 fits today
- Injectable VK2735 looks competitive on early-time weight loss (14.7% at 13 weeks) and could match or exceed tirzepatide at later time points if curves keep diverging – but this is still a “to be proven” assumption.
- Oral VK2735 is no longer a “slam-dunk differentiator” now that Wegovy pill is approved and Lilly’s orforglipron has Phase 3 under way; 12.2% at 13 weeks is solid, but long-term data, safety and real-world convenience will decide the winner.
- Viking’s key angle is flexibility: same molecule in both weekly injection and tablet, plus potential combination later with its own amylin program.
6. Financials and valuation – how much are you paying for this lottery ticket?
6.1 Current P&L and burn
- Q3 2025 net loss: $90.8M, versus $24.9M a year earlier – mainly due to R&D jumping to $90M as VANQUISH and VENTURE scaled.
- 9M 2025 net loss: $202M, almost triple the 9M 2024 loss.
- Cash, cash equivalents and short-term investments: $715M. No debt.
- R&D for the first nine months of 2025: $191.5M; G&A: $37.1M.
At a simple “current burn” run-rate, Viking has roughly 3–3.5 years of runway – which lines up roughly with the Phase 3 duration and NDA review period.
6.2 Multiples and 2030+ revenue debate
Because VKTX has no revenue today, investors lean on 2030-plus forecasts. These are noisy, but we can sketch the range:
- Consensus 2031 revenue for Viking around $3.3B on Fintel-type aggregators.
- Some bullish sell-side and media pieces cite “bold” scenarios of ~$3.5B by 2030 and $10B by 2034 if VK2735 takes a meaningful slice of the obesity market.
With an enterprise value of roughly $3.5B, that implies:
- EV / 2031 sales ≈ 1.0–1.1× if you take the $3.3B consensus at face value.
- Even if you haircut to ~$1.5B sales by 2030 (roughly “half of early hype”), you still sit around 2–2.5× EV/sales.
Compare that to:
- Big GLP-1 players like Lilly trading on mid-teens forward EV/sales and high single-digit 2030 EV/sales.
- Novo Nordisk around mid single-digit forward EV/sales and 3–3.5× 2030 EV/sales.
- Pfizer paying up to $7–10B for Metsera, whose lead asset is still in Phase 2 but has mid-teens weight loss and analysts modeling >$5B peak sales.
On that simple lens, VKTX looks cheap relative to success, but of course the denominator (future revenue) is highly speculative and the numerator (EV) will grow if Viking issues more shares to fund commercialization.
6.3 Capital allocation and pipeline focus
One thing investors give Viking credit for: focus. At recent conferences the CEO has been clear that, near-term, the company is essentially putting the spotlight on VK2735 (injectable and oral) and a next-gen amylin program, while older assets VK2809 and VK0214 are “available for licensing” rather than near-term cash sinks.
That discipline is an important ingredient in making the current cash stockpile last through Phase 3 and early launch – but it also means the investment thesis is even more concentrated on one mechanism.
7. Retail culture – VKTX as a cult ticker
VKTX doesn’t trade like a sleepy development-stage biotech. It trades like a cult GLP-1 name with a lot of smart people, a lot of YOLO and a lot of shorts.
7.1 Reddit and Stocktwits – “mini-Lilly” and “GLP-1 lottery ticket”
- A popular due-diligence thread on r/Vikingtherapeutics bluntly describes VK2735’s mechanism as “basically what Zepbound does but earlier in the life-cycle,” arguing the mechanism is “de-risked” because tirzepatide already works.
- A WallStreetBets-style DD frames VKTX as both a long-term GLP-1 play and a swing-trade on upcoming trial readouts, with some users openly running both a “core” and a “trading” position.
- On r/investing and r/stocks you regularly see posts along the lines of “I loaded the boat on VKTX, this is my best trade of the year” and speculation that Viking will eventually be bought, often with Pfizer named as the most likely acquirer.
None of this is “data,” but it matters because these communities do move short-term flows, especially when short interest is high.
7.2 Short interest and sentiment spikes
- Short interest sits around 22% of free float, with roughly 24M shares sold short and about 8–9 days to cover on average volume.
- Stocktwits data show repeated sentiment spikes where chatter jumps >200% in 24 hours when VKTX is in the news.
- When oral data came out in August 2025, message volume went from normal to “high” while sentiment swung from bullish to panic in hours – a reminder that crowd expectations were arguably ahead of the science.
For new investors dropping into VKTX because “GLP-1 is hot,” it’s crucial to realize that the stock is a playground for sophisticated longs, fast-money traders and
active shorts at the same time. That mix makes big intraday swings a feature, not a bug.
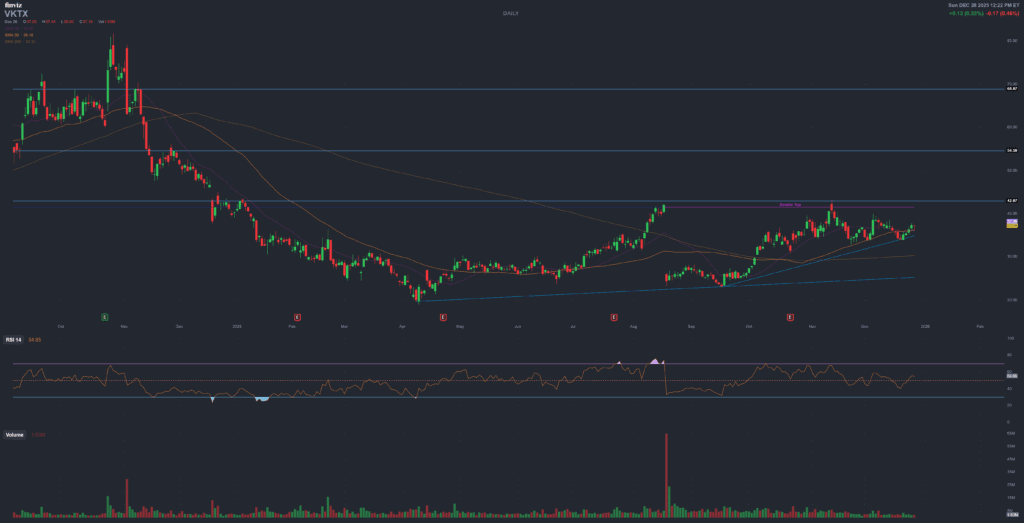
8. Trading setup – support, resistance and swing-trade logic
This is not a suggestion to trade VKTX, but if you’re going to anyway, it’s worth thinking in terms of levels and catalysts rather than “all-in forever”.
8.1 Technical context (late 2025)
- VKTX has been in a broad uptrend since early 2023, but 2024–2025 price action is dominated by violent rallies and sell-offs.
- 52-week range sits roughly between $18.9 and $43–48 depending on the source; current price is in the mid-30s.
- Chart watchers often highlight a support zone around $33 (recent Q4 2025 floor) and resistance around $39–40, corresponding to the post-Q3 earnings spike and multiple failed breakouts.
That 33–40 band roughly matches where short interest tends to reload and where retail enthusiasm tends to spike on good headlines.
8.2 How swing traders think about it
- Reddit swing-trading posts frequently mention VKTX as one of their “best trades of the year”, with typical holds of a few days to a couple of weeks around news events.
- Common playbook: buy near the uptrend support / recent floor, sell near the top of the range or just after a catalyst spike, leaving the long-dated clinical outcome risk to someone else.
- With short interest >20% and heavy options action, squeezes are possible in both directions – especially if oral/subQ news surprises versus consensus expectations from Lilly/Novo comparisons.
If you’re a long-term investor, it’s worth accepting that your P&L will sit inside someone else’s swing-trade battleground
until the 2027 Phase 3 data arrive. Position size accordingly.
9. Scenarios – what could VKTX look like in 2030?
These are educational sketches, not predictions and not recommendations.
Scenario A – Execution win, VK2735 becomes a serious third player
- VANQUISH-1 and VANQUISH-2 show strong, sustained weight loss (≥20% at 78 weeks) with manageable GI profile; maintenance data prove that a low-dose oral works for long-term control.
- FDA approves VK2735 for obesity and obesity + T2D around 2028; payer coverage is acceptable and Viking either partners or sells ex-US rights on attractive terms.
- By 2030, VK2735 has carved out, say, mid-single-digit global market share as a “Zepbound-class” drug with flexible dosing, and Viking’s revenue runs in the low-to-mid single-digit billions.
- Under this view, today’s ~1–2× EV/future sales multiple is a bargain if dilution is controlled.
Scenario B – Good drug, crowded market, “ok” equity outcome
- Phase 3 confirms attractive weight-loss but not clearly superior to tirzepatide/semaglutide combos; safety is acceptable but GI profile somewhat worse than best-in-class.
- Launch is solid but not spectacular; big players aggressively defend share with price cuts, combo regimens and telehealth ecosystems.
- Viking ends up with hundreds of millions to low billions in sales but needs several capital raises to finance global launches, diluting equity significantly; stock ends up “okay” but not spectacular relative to the risk taken.
Scenario C – Clinical or commercial disappointment
- One or both VANQUISH trials fail to hit primary endpoints with convincing margins, or safety / dropout issues worsen with longer follow-up.
- Even if approved, physicians and payers default to Wegovy/Mounjaro/Metsera combos, leaving VK2735 as a niche or second-line option with limited uptake.
- Cash burn forces Viking into dilutive financings or a sale at a price that looks modest relative to today’s market cap.
In this downside case, VKTX would become another example of how being “scientifically right” doesn’t automatically translate into equity outperformance in crowded mega-markets.
Whichever scenario you lean toward, the common theme is that VKTX is a high-variance bet on one platform.
For many readers, that makes it a candidate for small, satellite positions or swing trades – not a core holding.
10. Sources and further reading
Key sources used to build this report (only primary / high-quality sources; no low-quality blogs).
- Viking Therapeutics – Q3 2025 earnings release and corporate update: Q3 2025 financial results and Form 10-Q (quarter ended Sept 30, 2025).
- Viking Therapeutics press releases and presentations on VK2735 subcutaneous and oral formulations: ObesityWeek 2025 VK2735 obesity program data and Phase 2 VENTURE-Oral dosing top-line results.
- Phase 3 VANQUISH program: completion of enrollment in VANQUISH-1 and related VANQUISH clinical-trials and news coverage.
- Applied Clinical Trials – “Oral GLP-1/GIP Dual Agonist VK2735 Achieves Up to 12% Weight Loss in Phase II VENTURE Trial”.
- Mainstream coverage of the August 19, 2025 VK2735 oral sell-off and discontinuation rates: Reuters, Investor’s Business Daily, MarketWatch and Barron’s.
- Novo Nordisk Wegovy pill approval – FDA-related news and company releases: Novo Nordisk December 22, 2025 press release and global coverage from Reuters.
- Pfizer / Metsera deal and MET-097i data: Pfizer press release on Metsera acquisition and Phase 2b efficacy summaries from BioSpace and ClinicalTrialsArena.
- Valuation, consensus and short-interest data for VKTX: Fintel revenue and valuation estimates, MarketBeat short-interest statistics and Finviz fundamentals/price history.
- Obesity and GLP-1 market outlook: Evaluate Pharma 2030 GLP-1 projections, PharmaVoice analysis of Novo/Lilly GLP-1 dominance and Future Market Insights GLP-1 obesity market report.
- Retail sentiment and community discussion: r/Vikingtherapeutics, WallStreetBets VKTX DD thread and Stocktwits VKTX page (price and sentiment).
11. Disclaimer – educational only
This article is for educational and informational purposes only. It is not, and must not be interpreted as, investment advice, investment research, a recommendation to buy, sell or hold any financial instrument, or a solicitation to engage in any investment activity.
The information is based on publicly available sources believed to be reliable at the time of writing, but no representation or warranty is given as to its accuracy or completeness. Biotech and small-cap securities are highly volatile and speculative; investors can lose all or a substantial portion of their capital. Past performance and past clinical data are not indicative of future results.
Readers remain solely responsible for their own investment decisions and should consult qualified, independent financial and legal advisers before taking any action. The author and Merlintrader trading Blog accept no liability for any direct or indirect loss arising from the use of this material.
For full legal and risk disclosures, including privacy and terms of use, please refer to: Merlintrader – Disclaimer and Terms of use & privacy information.
Viking Therapeutics (VKTX) – VK2735 e la guerra GLP-1/GIP nell’obesità
Viking non ha ancora un farmaco approvato, ma punta a entrare nella stessa arena di Eli Lilly e Novo Nordisk con VK2735, un dual agonist GLP-1/GIP in versione iniettabile settimanale e in compressa orale giornaliera. I dati di Fase 2 sono notevoli, le Fasi 3 VANQUISH sono avviate, ma il timing è scomodo, la concorrenza è feroce e la cassa – pur robusta – non è infinita.
News del giorno
Fine 2025 – VKTX completa l’arruolamento di circa 4.650 pazienti nel trial di Fase 3 VANQUISH-1 per VK2735 sottocute (78 settimane)
e annuncia risultati positivi dello studio VENTURE-Oral, con fino al 12,2% di perdita media di peso a 13 settimane e chiara separazione dal placebo.
Nel frattempo la FDA approva la pillola Wegovy di Novo, primo GLP-1 orale per l’obesità, che parte subito in vantaggio sul fronte “pill vs iniezione”.
VKTX è diventato il classico titolo dove convivono fondi, hedge, retail fanatici e short aggressivi: perfetto per swing trade, ma anche per chi vuole seguire da vicino l’evoluzione delle terapie anti-obesità di nuova generazione.
1. Tesi in breve – cosa rappresenta VKTX oggi
Senza girarci troppo intorno, VKTX oggi è:
- Una scommessa concentrata su VK2735, con dati di Fase 2 che lo mettono nella parte alta della “classifica” GLP-1/GIP su 13 settimane, sia in versione iniettabile (14,7%) sia orale (12,2%).
- Un titolo ancora pre-ricavi con cassa ampia (715 M$) ma perdite in accelerazione (–202 M$ nei primi 9 mesi del 2025).
- Un “terzo incomodo” che prova a farsi spazio tra Lilly, Novo e ora Pfizer/Metsera, con tempistiche che portano la Fase 3 a chiudersi non prima del 2027 e un’eventuale commercializzazione nel 2028–2029.
- Un magnete per trader e short, con short interest sopra il 20% e community retail super-attiva su Reddit e Stocktwits che amplifica ogni news in volatilità.
Per un investitore paziente con orizzonte 2027–2030, VKTX può essere un’opzione interessante sul “terzo player” GLP-1/GIP. Per chi cerca solo stabilità, è probabilmente troppo caldo.
2. Timeline – da NASH di nicchia a GLP-1 di culto
Rapida carrellata, concentrandoci su ciò che conta per la tesi attuale.
2.1 Le origini (VK2809 / VK0214)
2015–2020
Viking “vecchia maniera”
Pipeline focalizzata su NASH (VK2809, agonista TRβ), NAFLD e X-ALD (VK0214), con studi Fase 2 positivi su LDL-C e grasso epatico.
Tema interessante ma poco sexy per il grande pubblico rispetto ai GLP-1.
2021–2023
Appare VK2735
I primi studi di VK2735 in versione iniettabile mostrano fino a ~8% di perdita di peso in 28 giorni di dosi settimanali,
con safety gestibile.
2.2 2024–2025: esplosione GLP-1 e scossoni
2024
VENTURE subQ e hype
Dati VENTURE in obesità: VK2735 sottocute arriva a 14,7% di perdita di peso media in 13 settimane senza plateau.
VKTX raddoppia più volte e diventa “mini-Lilly” per molti trader.
Giugno 2025
Partenza delle Fasi 3 VANQUISH
Parte il programma VANQUISH: due studi di Fase 3 da 78 settimane con VK2735 settimanale (obesità ± diabete).
19 agosto 2025
VENTURE-Oral e sell-off
Lo studio VENTURE-Oral raggiunge l’endpoint primario: fino al 12,2% di perdita peso media a 13 settimane, 10,9% vs placebo.
Ma i tassi di interruzione per eventi GI più alti del previsto scatenano un “panic sell” con il titolo giù anche del 40% nella stessa giornata.
Ott–Nov 2025
Q3, cassa e VANQUISH-1 full
Q3 2025: perdita 90,8 M$, ma cassa 715 M$; R&D esplosa a 90 M$ nel trimestre.
VANQUISH-1 completa l’arruolamento a 4.650 pazienti.
Dic 2025
La pillola Wegovy
La FDA approva la pillola Wegovy (semaglutide orale 25 mg) con circa 16–17% di perdita peso media a 64–68 settimane.
VKTX non è più il “solo” futuro GLP-1 orale alternativo.
3. Meccanismo – cosa fa davvero VK2735
3.1 Dual GLP-1/GIP
VK2735 è un agonista duale dei recettori GLP-1 e GIP, sulla falsariga di tirzepatide:
- GLP-1 riduce l’appetito, rallenta lo svuotamento gastrico e migliora glicemia e peso.
- GIP potenzia la secrezione insulinica e può amplificare l’effetto sul metabolismo quando viene co-attivato.
- L’idea è che la combinazione porti a cali di peso più rapidi e profondi rispetto a un GLP-1 puro.
3.2 Struttura del programma
- Formulazione sottocute settimanale (VANQUISH 1–2, Fase 3).
- Formulazione orale giornaliera (VENTURE, Fase 2 conclusa positivamente).
- Studio di maintenance in corso: idea di passare da dosi alte (iniettabile o orale) a basse dosi orali per mantenere il peso perso.
Un unico principio attivo, due vie di somministrazione e potenziale logica “induction + maintenance” tutta interna alla piattaforma.
4. Dati clinici – dove sta VK2735 rispetto agli altri
L’hype di agosto è stato tradito dal sentiment, non dai numeri brutti: i numeri di efficacia restano forti.
4.1 Iniettabile VENTURE (Fase 2)
| Endpoint | Risultato |
|---|---|
| Perdita peso media a 13 settimane | fino al 14,7% da baseline |
| Placebo-adjusted | ≈13% nei dosaggi più alti (stima da analisi terze) |
| ≥10% perdita peso | circa 80–90% dei pazienti vs ~4% placebo (dati esplorativi) |
| Trend | curva ancora in discesa a settimana 13, nessun plateau evidente |
| Safety | AE GI per lo più lievi/moderati, discontinuation simili al placebo |
4.2 Orale VENTURE (Fase 2)
| Endpoint | Risultato |
|---|---|
| Perdita peso media a 13 settimane | fino al 12,2% da baseline |
| Placebo-adjusted | fino al 10,9% meglio del placebo |
| ≥5% perdita peso | fino al 97% vs 10% placebo |
| ≥10% perdita peso | fino all’80% vs 5% placebo |
| Discontinuation | tassi più alti del previsto per eventi GI (20–38% vs ~18% placebo a seconda del dosaggio) |
Il punto debole dello studio non è la perdita di peso (molto buona per 13 settimane),
ma il profilo di tollerabilità GI e i dropout – da gestire con titrazioni più lente e selezione paziente migliore in Fase 3.
5. Concorrenza – Lilly, Novo, Pfizer/Metsera & co.
È impossibile valutare VKTX senza posizionarlo rispetto ai colossi e ai nuovi arrivi.
5.1 I due big
- Eli Lilly – tirzepatide (Mounjaro/Zepbound) dual GLP-1/GIP con ~20–22,5% di perdita peso media a 72 settimane; quota dominante del mercato incretin negli USA.
- Novo Nordisk – semaglutide (Wegovy) con efficacia leggermente inferiore ma presenza enorme, ora anche in formulazione pill con 16–17% di perdita peso nei trial.
5.2 Pfizer / Metsera
- MET-097i – GLP-1 RA long-acting, una volta al mese; Fase 2b con 14,1% di perdita peso placebo-adjusted a 28 settimane, con singoli pazienti oltre il 26%.
- Pfizer paga fino a 7–10 Mrd $ per Metsera, dopo aver abbandonato il proprio programma orale.
Questo deal è la migliore “validation” del fatto che programmi come VK2735 hanno valore significativo, anche se non saranno mai market leader assoluti come Zepbound o Wegovy.
6. Numeri e valutazione – quanto paghi il rischio
6.1 P&L e cassa
- Q3 2025: perdita netta 90,8 M$ (–0,81$/azione), R&D 90 M$, G&A 8,6 M$.
- 9M 2025: perdita 202 M$ vs 74,5 M$ l’anno precedente.
- Cassa + equivalenti + short term: 715 M$ a fine settembre 2025, nessun debito.
6.2 Multipli e stime 2030+
A questi livelli, l’enterprise value ~3,5 Mrd $ si confronta con:
- Stime 2031 intorno a 3,3 Mrd $ di ricavi (fonte Fintel / consensus).
- Scenari più “bull” che parlano di 3,5 Mrd $ nel 2030 e 10 Mrd $ nel 2034 se VK2735 diventasse un player importante nel GLP-1.
Tradotto: multipli EV/Sales futuri intorno a 1–2×, contro gli 8–15× che il mercato assegna oggi a Lilly/Novo, o l’EV/Sales implicito nella valutazione Metsera.
Ma attenzione: questi multipli hanno senso solo se le stime di ricavi si materializzano davvero – e prima bisogna passare indenni da Fase 3, EMA/FDA e concorrenza.
6.3 Allocazione del capitale
Il management ha chiarito, in più occasioni, che nel breve concentrerà la spesa su VK2735 (subQ + orale) e sul nuovo programma amylin, lasciando VK2809/VK0214 “pronti per licensing” ma non in focus operativo. È una scelta che piace ai fondamentali (focus e disciplina), ma aumenta la dipendenza da un singolo asset.
7. Community retail – VKTX come “ticker di culto”
VKTX non è solo un titolo biotech: è un piccolo ecosistema social.
7.1 Reddit / Stocktwits
- Su r/Vikingtherapeutics spopolano i thread di “scientific DD”, con utenti che spiegano il meccanismo GLP-1/GIP e fanno confronti numerici contro Zepbound, Wegovy, Metsera.
- Su r/wallstreetbets & co. circola da mesi un DD che presenta VKTX come “best GLP-1 pure play” e “swing + long insieme”, con target price molto ambiziosi.
- Stocktwits registra spesso raddoppi del volume di messaggi (+200% o più in 24h) quando arriva una news clinica o un report di broker importante.
7.2 Short interest e dinamica swing
- Short interest ~22% del flottante, 8–9 giorni per coprire su volumi medi: numeri che rendono possibili squeeze violenti in entrambe le direzioni.
- Molti trader dichiarano esplicitamente di aver fatto “il trade dell’anno” proprio su VKTX, spesso con cicli di 5–10 giorni tra ingresso e uscita.
Tutto questo aumenta l’appeal per chi cerca swing trade, ma è un avvertimento per chi vuole semplicemente “comprare e dimenticare”: il rumore di breve resterà elevato fino almeno alle letture di Fase 3.
8. Setup di trading – livelli chiave e logica
Non è un consiglio operativo, ma una fotografia di come molti trader stanno ragionando sul titolo.
8.1 Livelli
- Range 52-week circa 18,9–43–48 $; prezzo attuale in area metà 30.
- Supporto “psicologico” e tecnico recente intorno ai 33 $, dove più volte il titolo ha rimbalzato nell’ultimo trimestre.
- Area di resistenza tra 39 e 40 $, dove i tentativi di breakout 2025 sono stati respinti più volte.
8.2 Swing view
Schema tipico che si vede nei thread:
- Accumulo graduale vicino al supporto/uptrend (mid-30 o sotto), stop relativamente stretto sotto i minimi precedenti.
- Uscita parziale o totale in area 39–40 o su spike di news (upgrade broker, headline positive su GLP-1, rumor M&A).
- Zero leva o leva moderata: la combinazione short interest + news-flow può trasformare rapidamente un “trade figo” in drawdown pesante.
Anche per chi non fa trading attivo, sapere che molti altri lo fanno su VKTX aiuta a non farsi spaventare (o euforizzare) dal rumore di breve.
9. Scenari – come potrebbe essere VKTX nel 2030
Stesso approccio della parte EN: non sono previsioni, ma mappe mentali.
Scenario A – Esecuzione perfetta, VK2735 diventa “terzo polo”
- Fase 3 VANQUISH con risultati solidi, profilo di safety accettabile e buona gestibilità GI.
- FDA ed EMA approvano VK2735 per più indicazioni; arrivano partnership e forse M&A su multipli EV/Sales simili (o migliori) rispetto al deal Metsera.
- VKTX converte la cassa in una piattaforma GLP-1/GIP + amylin, con ricavi a 2030 nell’ordine dei miliardi e multipli di mercato molto più alti del livello attuale.
Scenario B – Buon farmaco, equity outcome “così così”
- VK2735 approvato ma non superiore a Mounjaro/Zepbound/Wegovy; mercato GLP-1 maturo e molto competitivo.
- Ricavi a qualche centinaio di milioni/pochi miliardi, ma con equity raise successivi che diluiscono molto l’azionista attuale.
- Il titolo resta interessante, ma non esplosivo rispetto al rischio corso.
Scenario C – Dati deludenti o execution debole
- Fase 3 non replica la forza di Fase 2 o emergono problemi di tollerabilità/aderenza a lungo termine.
- Lilly, Novo e Pfizer saturano il mercato con combo e prezzi aggressivi; VK2735 resta spiazzato come “me too tardivo”.
- VKTX finisce per vendere gli asset a valutazioni non entusiasmanti, dopo altra diluizione per completare i programmi.
Morale: VKTX è una storia high-risk/high-reward da dosare con attenzione in portafoglio.
Per chi ama il settore GLP-1 può avere senso come “satellite” accanto ai big, non come posizione dominante.
10. Fonti e approfondimenti
Stesse fonti della versione inglese, elencate qui per comodità.
- Risultati Q3 2025 e 10-Q Viking Therapeutics: comunicato Q3 2025 e Form 10-Q.
- Comunicati VENTURE (subQ e orale) e presentazioni ObesityWeek su VK2735: ObesityWeek 2025 e VENTURE-Oral top-line.
- Programma VANQUISH (Fase 3) e completamento arruolamento VANQUISH-1: comunicato ufficiale.
- Applied Clinical Trials – articolo su VENTURE-Oral.
- Articoli su sell-off di agosto 2025 (dati orali VK2735): Reuters, Investor’s Business Daily, MarketWatch, Barron’s.
- Wegovy pill (semaglutide orale 25 mg): comunicato Novo Nordisk e copertura Reuters.
- Deal Pfizer / Metsera e dati MET-097i: press release Pfizer, BioSpace, ClinicalTrialsArena.
- Dati di consenso e short interest VKTX: Fintel, MarketBeat short interest e Finviz.
- Studi e report su mercato obesità/GLP-1: Evaluate, PharmaVoice, Future Market Insights.
- Esempi di sentiment retail: r/Vikingtherapeutics, thread DD su r/wallstreetbets e pagina VKTX su Stocktwits.
11. Disclaimer – solo a scopo informativo
Questo articolo ha esclusivamente finalità informative e didattiche. Non costituisce e non deve essere interpretato come consulenza in materia di investimenti, raccomandazione personalizzata, ricerca in materia di investimenti, sollecitazione al pubblico risparmio o offerta di strumenti finanziari.
Le informazioni sono basate su fonti ritenute affidabili al momento della redazione, ma non se ne garantisce accuratezza o completezza. I titoli biotech e le small cap sono altamente speculativi e volatili; è possibile perdere una parte rilevante o la totalità del capitale investito. Rendimenti e dati clinici passati non sono indicativi di risultati futuri.
Ogni decisione di investimento rimane a esclusivo carico del lettore, che dovrebbe rivolgersi a consulenti finanziari e legali abilitati prima di assumere qualunque decisione. L’autore e Merlintrader trading Blog declinano ogni responsabilità per eventuali perdite dirette o indirette derivanti dall’uso di questo materiale.
Per testi completi su rischi, responsabilità, privacy e trattamento dati si vedano: Disclaimer Merlintrader e Condizioni d’uso e info privacy.
Biotech Catalyst Calendar
Anche per VKTX, il punto non è solo il singolo titolo ma il contesto:
PDUFA, letture di Fase 3, nuovi GLP-1 di big pharma e M&A possono cambiare la percezione del rischio in poche settimane.
Apri il Biotech Catalyst Calendar
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


