DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
TVTX Travere Therapeutics Inc

Today is about one thing: uncertainty around the FDA decision timing/outcome for the FILSPARI FSGS sNDA. The company explicitly highlighted FDA information requests right before the target date.
PL Planet Labs PBC

Focus: multi-year low 9-figure Satellite Services contract with the Swedish Armed Forces – third sovereign-space deal in 12 months, >$500M combined.
TENX Tenax Therapeutics Inc

Tenax Therapeutics (TENX) – Deep Dive / Analisi Approfondita (Jan 2023 – Oggi) Tenax Therapeutics (TENX) – Deep Dive / Analisi Approfondita (Jan 2023 – Oggi) Full deep dive on TENX: financials, pipeline, catalysts, shareholder structure, insider activity, dilution, analyst…
IDYA Ideaya Biosciences Inc

Precision oncology company built around synthetic lethality, with darovasertib in uveal melanoma and a broad pipeline (MAT2A, PRMT5, PARG, ADCs) backed by more than 1.1 billion dollars in cash and a guided runway into 2030.
RCKT Rocket Pharmaceuticals

Focus: FDA PDUFA for KRESLADI™ (LAD-I) on March 28, 2026 – gene therapy, rare pediatric disease.
ATRA Atara Biotherapeutics Inc Jan 12 Update

Yesterday’s report framed ATRA around a PDUFA-window setup, with the assumption that the 2025 CRL was primarily a CMC/GMP story and that the ALLELE trial remained acceptable as the pivotal efficacy dataset. Today that premise is broken: the FDA has issued a second Complete Response Letter (CRL) for tabelecleucel/tab-cel in EBV+ PTLD.
Market Review | Preview for the Week of Jan 12

Biotech catalysts, small-cap rotation, space & defence names already on the Merlintrader radar, Trump factor, geopolitics (Ukraine, Red Sea, Venezuela, Greenland) and J.P. Morgan Healthcare Conference 2026.
SANA Biotechnology

As of January 10, 2026, Sana Biotechnology (NASDAQ: SANA) stands as a clinical-stage pioneer focused on the development of engineered cells as a new modality of medicine. Founded on the belief that cell therapy will follow small molecules and biologics as the "third pillar" of therapeutics, Sana has built a platform to address the two primary bottlenecks of the industry: Immune Evasion and Scalable Manufacturing.
MDGL Madrigal Pharmaceuticals Inc

Madrigal (MDGL) is a commercial-stage biopharma focused on metabolic dysfunction-associated steatohepatitis (MASH, formerly NASH). Its lead product, Rezdiffra (resmetirom), received FDA accelerated approval on March 14, 2024 as the first treatment for adults with noncirrhotic MASH with moderate to advanced fibrosis (F2–F3), to be used with diet and exercise.
CRMD Cormedix Inc jan 12 Legislative update (KCAPA 2026)
CorMedix Inc. has crossed a line that many small biotechs never reach. It is no longer a binary, pre-revenue story waiting for a single FDA decision. With DefenCath approved, the Melinta acquisition closed and several hundred million dollars of revenue on the board, CRMD now behaves like a small-cap specialty pharma focused on preventing and treating serious infections linked to vascular access.
SMMT Summit Therapeutics Inc

Summit Therapeutics Inc. (NASDAQ: SMMT) has effectively transformed itself into a single-asset, late-stage oncology company centered on ivonescimab (SMT112), a PD-1 × VEGF bispecific antibody licensed from Akeso in January 2023 (FY2024 PR). Ivonescimab is already approved in China (Akeso) and is now in a broad, global Phase III program led by Summit in NSCLC and colorectal cancer.
RVMD Revolution Medicines

Revolution Medicines, Inc. (Nasdaq: RVMD) enters the 44th Annual J.P. Morgan Healthcare Conference as a late-stage, pre-commercial oncology company focused on RAS(ON) inhibitors. The company’s FY24 results release (link) positions RVMD as “a late-stage clinical oncology company developing targeted therapies for patients with RAS-addicted cancers,” with a deep pipeline led by daraxonrasib, elironrasib and zoldonrasib.
OCGN Ocugen Inc

Ocugen, Inc. (Nasdaq: OCGN) is a clinical-stage biotechnology company focused on gene therapies for blindness diseases and related ophthalmic biologics. Its core asset base is a “modifier gene therapy” platform built around nuclear hormone receptors (NHRs) designed to reset dysfunctional retinal gene networks rather than replace single mutated genes. The three flagship programs are OCU400 for retinitis pigmentosa (RP), OCU410 for geographic atrophy (GA, late-stage dry age-related macular degeneration) and OCU410ST for Stargardt disease, each targeting sizeable unmet-need populations in inherited and multifactorial retinal disease.
CLRB Cellectar Biosciences Inc

Cellectar Biosciences (Nasdaq: CLRB) is a late-stage clinical radiopharmaceutical company built around a proprietary phospholipid drug conjugate (PDC) platform. Its lead asset, iopofosine I 131, delivers radioactive iodine-131 directly to tumor cell membranes; the most advanced program is in relapsed/refractory Waldenström macroglobulinemia (WM) after BTK inhibitor therapy, supported by the pivotal CLOVER-WaM Phase 2 data described in the company’s Form 10-K 2024 (Business section).
DVLT Datavault AI Inc

Datavault AI Inc. (Nasdaq: DVLT) is a small/micro-cap technology company that wants to sit at the intersection of AI, Web3 and data monetization. It licenses a portfolio of patents and software platforms to tokenize and value “real-world assets” (RWA) – from genomic data and prescription flows to commodities and luxury goods – and still carries the legacy wireless-audio business acquired from WiSA.
AQST Aquestive Therapeutics
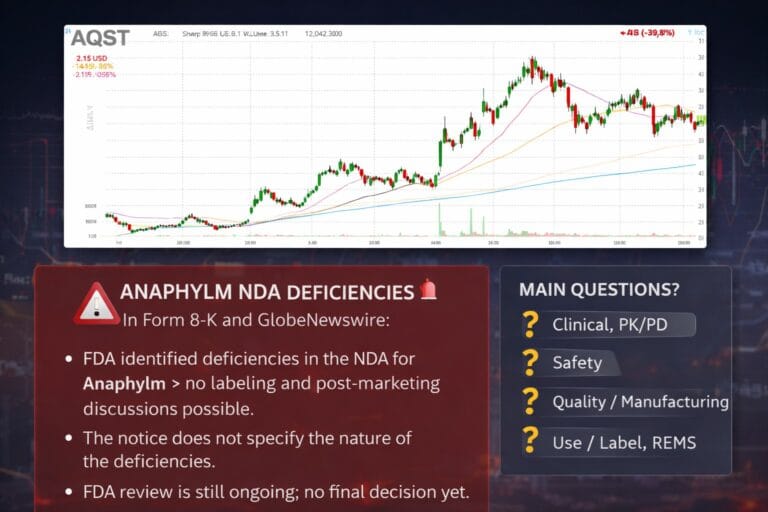
On 9 January 2026, Aquestive Therapeutics announced that the US FDA had identified “deficiencies” in the NDA for Anaphylm (dibutepinephrine sublingual film). These deficiencies currently preclude discussions of labeling and post-marketing commitments.
SAVA Cassava Sciences Inc

A factual deep dive on where SAVA really stands after shutting down Alzheimer’s, pivoting to TSC-related epilepsy, facing a full FDA clinical hold, settling a $31.25M class action and attracting heavy call-option interest and insider buying.
KRYS Krystal Biotech Inc

HSV-1 gene therapy platform moves from skin to lung: robust CFTR expression in CF lungs, repeat dosing on deck, and JPM 2026 as the launchpad for a mutation-agnostic CF strategy.
SPACE & DEFENCE 2026

SIDU, ASTS, LUNR, ONDS and PL are not just “hot tickers”. Their satellites, landers, drones and imaging constellations are becoming building blocks of a Western answer to China’s dual-use space programs, Russia’s drone war and a world where infrastructure and data are contested every single day.
ONDS Ondas Holdings Inc jan 8

This is not a buy/sell note, but a way to show readers how Ondas (ONDS) has been covered on Merlintrader over the last months, how the stock moved from November into early January, and what the upcoming Needham Growth Conference presentation actually adds (or doesn’t add) to the picture. Educational only, not investment advice.