DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Ticker: TNGX (NASDAQ)
Tango Therapeutics (TNGX) – PRMT5, CoREST and a crowded short base into 2026
Synthetic lethality biotech with a PRMT5/CoREST pipeline, a sharp 2025 rerating, heavy short interest and a technical setup that now sits at the lower bound of a rising channel right before J.P. Morgan 2026.
News & Technical Snapshot – Why TNGX matters right now
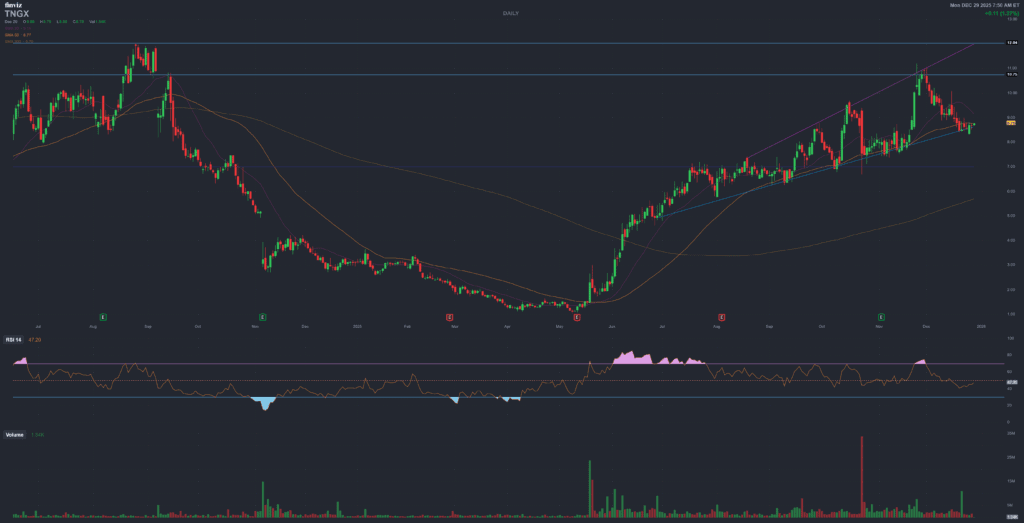
Late Dec 2025TNGX has tripled in 2025, pulled back from its recent high and now trades near the bottom of a rising channel – right as key clinical and strategic events line up into early 2026.
1) Price action – base of an ascending channel into mid-January
After a brutal washout to around $1.10 in May 2025, Tango rallied to a 52-week high of about $10.9 in late November 2025. As of late December 2025 the stock trades around $8.6–8.7 with a market cap near $1.1–1.2B, roughly 20% below the recent peak but still up several hundred percent from the lows.
On a simple trendline view, the current area (~$8–9) lines up with:
- the rising support connecting the May 2025 low and the September 2025 consolidation band;
- a prior volume node where large funds (including Third Rock) traded size between $7–8.
If that channel holds, a progressive grind back towards the $10–11 area into mid-January would not be technically surprising – and the timing would overlap almost perfectly with the J.P. Morgan Healthcare Conference presentation on January 14, 2026. Of course, patterns fail all the time: this is a technical observation, not a signal.
2) Fundamental story – PRMT5 as the core, NSCLC and pancreas as the test
Tango’s lead asset is vopimetostat (TNG462), a next-generation PRMT5 inhibitor for patients with MTAP-deleted cancers. Dose-expansion data in October 2025 showed:
- in second-line MTAP-deleted pancreatic cancer: median PFS ~7.2 months and ORR ~25%;
- signals of activity in lung and other solid tumors, with a pivotal trial in 2L MTAP-del pancreatic cancer planned to start enrolling in 2026.
In parallel, TNG260, a first-in-class CoREST inhibitor, is in Phase 1/2 dose expansion with pembrolizumab in STK11-mutant NSCLC, a population that usually does poorly on immunotherapy alone. Both programs are designed to “rescue” difficult molecular subsets that current drugs don’t treat well.
3) Cash, dilution and the “short squeeze” narrative
As of 31 March 2025, Tango reported about $216.7M in cash, cash equivalents and marketable securities, guiding that this should fund operations into Q1 2027 after cutting some preclinical spend. Since then, the company has:
- priced roughly $225M of equity and PIPE financing on the back of the October 2025 data;
- set up a new $100M at-the-market (ATM) equity program with Leerink Partners in November 2025.
At the same time, short interest has climbed to roughly 33M shares, around 25% of shares outstanding and >40% of free float, with a short-interest ratio near 10–12 days. That combination (heavy short base + high-profile catalysts + visible uptrend) is exactly what fuels “squeeze” talk on retail channels – but it also reflects real dilution risk and the complexity of the program.
Quick data – TNGX at a glance
Snapshot (late Dec 2025)Ticker / ExchangeTNGX – Nasdaq
Share price≈ $8.7 (late Dec 2025)
52-week range$1.10 – $10.91
Market cap≈ $1.1–1.2B
SectorOncology / Synthetic lethality
StageClinical-stage biotech (no product sales)
Lead assetVopimetostat (TNG462, PRMT5 inhibitor)
Key target subsetMTAP-deleted cancers
Second pillarTNG260 (CoREST inhibitor, STK11-mut NSCLC)
Cash & securities (Mar 31, 2025)~$216.7M
Runway (company guide)To ~Q1 2027
Short interest~33M shares (~25% OS, >40% float)
Short ratio~10–12 days to cover
Analyst 12-m PT (avg / range)≈ $12.5–13.25 (range $10–15)
Upcoming eventJPM 2026 presentation – Jan 14, 2026
Data compiled from Tango filings/press releases, SEC, StockAnalysis/Yahoo/MarketBeat, Fintel/Nasdaq and clinicaltrials.gov. Values rounded and for information only, not for trading decisions.
Synthetic lethality
MTAP-deleted cancers
STK11-mutant NSCLC
High short interest
Cash-intensive pipeline
Executive Summary – What Tango Therapeutics is (and is not)
Context for non-specialistsTango Therapeutics is a clinical-stage biotech built around the concept of synthetic lethality: exploiting specific genetic weaknesses in cancer cells (such as MTAP deletion or STK11 mutation) to kill tumors while sparing normal tissue. The company has become one of the more visible names in this niche thanks to strong early data with its PRMT5 inhibitor vopimetostat (TNG462) and a very vocal retail community around its ticker.
- Core of the story – PRMT5 and MTAP deletion: TNG462 is a next-generation PRMT5 inhibitor that relies on MTAP deletion to selectively attack cancer cells. Early Phase 1/2 data – especially in second-line MTAP-deleted pancreatic cancer – show meaningful responses and a median PFS that compares favorably with historical chemotherapy.
- Second pillar – CoREST and STK11-mutant NSCLC: TNG260 aims at STK11-mutant tumors via CoREST inhibition, restoring sensitivity to anti-PD-1 drugs like pembrolizumab. The Phase 1/2 dose expansion in NSCLC is ongoing, with proof-of-mechanism and acceptable safety already shown and further clinical data guided into 2026.
- Additional pipeline – TNG456 and TNG961: TNG456 brings PRMT5 into the brain for MTAP-deleted glioblastoma, while TNG961 is a first-in-class HBS1L molecular glue degrader targeting FOCAD-deleted tumors – still in IND-enabling/preclinical but important for long-term upside.
- Cash and dilution: Q1 2025 showed ~$216.7M in cash and an extended runway into Q1 2027, later reinforced – and diluted – by a ~$225M equity/PIPE deal and a new $100M ATM program.
- Short interest & volatility: short interest of roughly 33M shares (~25% of shares outstanding, >40% of float) on a name that has moved from ~$1 to >$10 within months makes TNGX structurally volatile and naturally feeds “squeeze” narratives.
- Analyst view: recent consensus from 6–10 covering analysts points to a Strong/Moderate Buy rating with average 12-month price targets clustering around $12.5–13.25 (roughly 40–45% above late-2025 levels), with individual targets ranging from $10 to $15.
This report is meant to lay out facts, timelines, risks and sentiment so that readers can form their own view. It is explicitly not a recommendation to buy, sell or hold TNGX or any other security.
1. Company & Platform – Synthetic lethality in practice
From CRISPR screens to clinical programsTango’s core idea is to systematically apply synthetic lethality: if gene A is lost in cancer cells (e.g. MTAP, FOCAD, STK11), then inhibiting gene B (PRMT5, HBS1L, CoREST complex) selectively kills those cancer cells while sparing normal tissue.
1.1 PRMT5 axis – Vopimetostat (TNG462) and TNG456
PRMT5 is an arginine methyltransferase involved in RNA splicing and gene expression. In MTAP-deleted cells, PRMT5 becomes particularly critical, making it an attractive synthetic lethal target. Tango initially advanced an older PRMT5 inhibitor (TNG908) but pivoted to vopimetostat (TNG462) as a more potent, MTA-cooperative, potentially best-in-class drug. TNG456 extends the same logic into the brain with a formulation designed to achieve sufficient CNS exposure for MTAP-deleted glioblastoma.
1.2 CoREST axis – TNG260 for STK11-mutant NSCLC
TNG260 targets the CoREST complex, which plays a role in chromatin remodeling and immune evasion. Preclinical work showed that CoREST inhibition can re-sensitize STK11-deficient NSCLC to anti-PD-1 therapy, leading to regression in mouse models when combined with PD-1 blockade. This translates into the current Phase 1/2 trial in STK11-mutant, RAS wild-type NSCLC combining TNG260 with pembrolizumab.
1.3 HBS1L / FOCAD – TNG961
TNG961 is a molecular glue degrader that targets HBS1L in tumors with coincident FOCAD deletion, a subset found in a meaningful fraction of MTAP-deleted cancers. It remains preclinical but shows how Tango wants to build a portfolio of genetically defined “niches” rather than broad, undifferentiated indications.
2. Lead Programs & 2025–2026 Catalyst Map
Vopimetostat, TNG260, TNG456, TNG9612.1 Vopimetostat (TNG462) – MTAP-deleted pancreatic and lung cancer
The October 23, 2025 data update from the ongoing Phase 1/2 study of TNG462 in MTAP-deleted cancers highlighted:
- in 2L MTAP-deleted pancreatic cancer (single-agent vopimetostat 250 mg daily): median PFS ≈ 7.2 months, ORR ≈ 25%;
- responses and durable disease control across multiple solid tumors, including lung cancer;
- manageable safety profile, with the main concern being chronic low-grade toxicity rather than acute DLTs;
- plans for a global pivotal study in 2L MTAP-deleted pancreatic cancer in 2026, enrolling ~300 patients vs four chemo regimens.
TNG462 is also being combined with Revolution Medicines’ RAS(ON) inhibitors daraxonrasib and zoldonrasib in pancreatic and lung cancer, with enrollment launched in 2025 and initial combo data guided for 2026. These combinations are important for:
- testing whether PRMT5 + RAS(ON) is synergistic in KRAS-driven tumors;
- supporting Tango’s ambition to be a “must-have” partner in RAS-mutant oncology rather than a niche MTAP story.
2.2 TNG260 – CoREST inhibitor for STK11-mutant NSCLC
The Phase 1/2 trial (NCT05887492) of TNG260 in combination with pembrolizumab is enrolling STK11-mutant, RAS wild-type solid tumors, with a focus on NSCLC. Key points from Tango’s 2025 updates include:
- proof-of-mechanism established via pharmacodynamic changes in on-treatment biopsies at 80 mg QD;
- favorable safety, tolerability and pharmacokinetics at that expansion dose so far;
- dose-expansion cohort in NSCLC ongoing, with more mature clinical data expected across 2026.
Preclinical work published in Cancer Research showed that TNG260 can sensitize STK11-mutant NSCLC to anti-PD-1 therapy, leading to tumor regression in mouse models. Turning that into reproducible human efficacy – with an acceptable immune-tox profile – is one of the most important “go or no-go” checkpoints for the CoREST program.
2.3 TNG456 – Brain-penetrant PRMT5 for glioblastoma
TNG456 is designed to bring MTAP-selective PRMT5 inhibition into the CNS. Tango has initiated a Phase 1/2 trial in MTAP-deleted glioblastoma and other solid tumors, with dose escalation underway. This program:
- expands the PRMT5 franchise into a notoriously hard-to-treat indication (GBM);
- acts as a technological proof-point that Tango can design CNS-penetrant synthetic lethality drugs.
2.4 TNG961 – HBS1L molecular glue degrader
TNG961 remains in the IND-enabling stage, targeting HBS1L in FOCAD-deleted tumors (often overlapping with MTAP deletion). For now it is more of a “future optionality” asset, but it reinforces the idea of Tango as a multi-target synthetic lethality platform.
2.5 Timeline – key milestones (2025–2026)
High-level timeline (approximate)
- May 12, 2025 – Q1 2025 results and business highlights; runway guided into Q1 2027; TNG462/TNG260/TNG456/TNG961 updates.
- 2H 2025 – Planned new data from TNG462 and TNG260 (monotherapy and combo, early conference readouts).
- Oct 23, 2025 – Positive Phase 1/2 data for TNG462; 2L pancreatic mPFS 7.2 months, ORR 25%; pivotal 2L pancreas study announced for 2026.
- Oct–Nov 2025 – Short interest rises as stock rerates; ~$225M equity/PIPE financing and new $100M ATM established.
- Late Nov 2025 – New 52-week high around $10.9; subsequent pullback into the current range.
- Jan 14, 2026 – Corporate presentation at the 44th Annual J.P. Morgan Healthcare Conference (9:45 am PST / 12:45 pm EST).
- 2026 – Expected start of pivotal Phase 3 for TNG462 in 2L MTAP-deleted pancreatic cancer; emerging data from RAS(ON) combos and expanded NSCLC cohorts.
3. Management & Governance – Who is steering Tango?
Barbara Weber and teamTango is led by Barbara L. Weber, M.D., President, Chief Executive Officer and co-founder of the company. She built Tango within Third Rock Ventures starting in 2015 and has served as CEO since 2017, leading it from Series A startup to a publicly-listed clinical-stage biotech.
Dr. Weber is a board-certified medical oncologist with more than 25 years of experience in cancer genetics and oncology drug development. Before Tango she served as:
- Senior Vice President & Global Head, Oncology Translational Medicine at Novartis (2009–2015);
- Vice President, Oncology Discovery & Translational Medicine at GlaxoSmithKline (2005–2009);
- Professor of Medicine and Genetics at the University of Pennsylvania (1990s–2000s), where she led a major clinical and translational research program in cancer genetics and contributed to early BRCA1/BRCA2 work.
As a Venture Partner at Third Rock Ventures she also helped build Neon Therapeutics (later acquired by BioNTech) and Relay Therapeutics from early seed stage through IPO. She currently sits on the boards of Revolution Medicines, FogPharma, Parabilis Medicines and other oncology-focused companies.
Dr. Weber holds a B.S. in Chemistry and an M.D. from the University of Washington, completed her internal medicine residency at Yale University and a medical oncology fellowship at Dana-Farber Cancer Institute. She is an elected member of the American Association of Physicians and the American Society for Clinical Investigation, has served as president of ASCI and on the boards of both ASCO and AACR.
The board is chaired by Alexis Borisy (independent) and the majority of directors are non-executive, which provides some balance to the CEO’s long-standing operational role.
4. Sentiment & Retail Chatter – What the crowd is saying
Reddit, Stocktwits, X (non-professional)On retail channels (Reddit, Stocktwits, X), Tango tends to split into two camps:
4.1 Bullish themes
- “Best-in-class PRMT5” narrative: many posts repeat that TNG462 looks superior to older PRMT5 attempts, especially in 2L pancreatic cancer, with comments about “potential next generation standard of care” in MTAP-deleted subsets if pivotal data hold up.
- “Short squeeze fuel”: the combination of ~25% of shares and >40% of float sold short, plus a small float and visible catalysts (JPM, pivotal start, further TNG260 data), naturally attracts squeeze scenarios, price targets in the $20–30+ range, and aggressive “diamond hands” rhetoric.
- Platform optionality: some more sophisticated posts emphasize that Tango is not just TNG462, but a broader synthetic-lethality engine (TNG260, TNG456, TNG961) in high unmet-need niches.
4.2 Bearish / skeptical themes
- Dilution fatigue: after the $225M offering and the new $100M ATM, plus a long road to potential commercialization, many traders focus on the risk of repeated equity raises and the question “how many times will they tap the market before approval?”.
- Execution and trial risk: concerns that moving from early Phase 1/2 data to a large pivotal pancreatic trial is a big step, with real risk of under-performance, unanticipated toxicity or competition from other targeted strategies.
- Over-crowded trade: some see TNGX as a “too popular” story where everyone already knows the bullish talking points, making it vulnerable to sharp corrections on any disappointing update or macro risk-off.
All of these are opinions from non-professional traders and investors. They can help frame positioning and psychology, but they are not research and should not be used as the sole basis for any decision.
These links may include affiliate/referral components. They do not change this editorial view and are not endorsements of any specific external service.
5. Key Risks & Red Flags
Where things can go wrong5.1 Clinical & regulatory risk
High TNG462 and TNG260 remain in Phase 1/2. Early signals, even good ones, frequently fail to hold in larger, more heterogeneous populations. Pivotal pancreatic and NSCLC trials carry meaningful risk of under-performance vs expectations or competing regimens.
5.2 Commercial and competitive risk
Medium / execution-driven
- MTAP deletion and STK11 mutation define narrow but complicated populations with evolving standards of care.
- Other companies are targeting overlapping biology (alternative PRMT5 approaches, KRAS combos, novel immunotherapies).
- Pricing, reimbursement and real-world tolerability can all erode the theoretical edge seen in early trials.
5.3 Balance sheet, dilution and funding
High Even after cost cuts and equity raises, Tango is still years away from potential product revenue. The company has openly prioritized its PRMT5 programs and deferred some studies to extend runway, but:
- large pivotal trials + combos are expensive;
- future dilution or non-equity financing structures remain a realistic scenario;
- ATM usage can pressure the stock during rallies if not carefully managed.
5.4 Short interest and volatility
Medium–High High short interest can cut both ways:
- in positive scenarios it can fuel violent upside squeezes;
- in negative or uncertain scenarios it can contribute to grinding downtrends as rallies are sold into;
- in all cases it adds volatility and makes position sizing more critical.
6. Scenario Map – Neutral high-level view
Not a recommendation6.1 Constructive scenario (what would need to go right)
- Vopimetostat confirms durable benefit in MTAP-deleted pancreatic cancer and advances into a well-designed pivotal study with clean safety.
- Early NSCLC signals with TNG462 and/or TNG260 are strong enough to justify expansion and eventual registrational plans.
- Management strikes one or more strategic partnerships (e.g. ex-US rights or combo deals) that bring non-dilutive capital.
- Cash burn remains controlled, with dilution coming at progressively higher prices rather than into weakness.
6.2 Cautious / downside scenario (what could break)
- TNG462 data flatten as trials grow, or safety becomes more complex in broader populations.
- TNG260 fails to show robust benefit in STK11-mutant NSCLC beyond biomarker shifts.
- Competing approaches in pancreatic or lung cancer outpace Tango’s development timeline.
- Equity and ATM usage increase faster than expected, eroding per-share upside and investor patience.
These are scenarios, not forecasts. They outline possible paths, without assigning probabilities or making any trade call.
7. Sources & Further Reading
Verifiable documents only-
Tango Therapeutics – Q1 2025 results and business highlights (GlobeNewswire, May 12, 2025):
https://www.globenewswire.com/news-release/2025/05/12/3078966/0/en/… -
Form 8-K – Tango Therapeutics Q1 2025 (SEC):
https://ir.tangotx.com/static-files/667e56fb-23d5-43ef-a0aa-42848637045e -
Vopimetostat (TNG462) Phase 1/2 MTAP-deleted data (GlobeNewswire, Oct 23, 2025):
https://www.globenewswire.com/news-release/2025/10/23/3171895/0/en/… -
“Pancreatic Revolution due? Tango PRMT5 data satisfy” – BioWorld (Oct 24, 2025):
https://www.bioworld.com/articles/725397-pancreatic-revolution-due-tango-prmt5-data-satisfy -
TNG260 CoREST preclinical paper – Cancer Research (2025):
https://aacrjournals.org/cancerres/article/85/20/3966/766095/… -
TNG260 clinical trial record (NCT05887492):
https://clinicaltrials.gov/study/NCT05887492 -
J.P. Morgan 2026 conference participation PR (Dec 17, 2025):
https://www.globenewswire.com/news-release/2025/12/17/3206852/0/en/… -
Analyst forecast & target price pages:
StockAnalysis forecast – https://stockanalysis.com/stocks/tngx/forecast/
Yahoo Finance analysis – https://finance.yahoo.com/quote/TNGX/analysis/
MarketBeat consensus – https://www.marketbeat.com/stocks/NASDAQ/TNGX/price-target/ -
Barbara Weber background:
SEC 10-K bio – 10-K 2021–2022
Parabilis/ITM profiles – https://parabilismed.com/board/barbara-weber-md/, https://www.itm-radiopharma.com/company/supervisory-board/
Educational / legal disclaimer (EN)
This content is for informational and editorial purposes only. It is not, and must not be interpreted as, investment advice, a solicitation, or a recommendation to buy or sell any security or financial instrument. All data are based on public sources (company filings, press releases, clinical trial registries and established financial data providers) believed to be reliable, but errors and omissions are always possible. Readers should independently verify all key figures (including dates, cash, revenue, trial status, short interest and regulatory milestones) using primary documents such as SEC filings, official regulatory websites and company disclosures.
Any reference to analyst price targets, ratings or community comments reflects third-party opinions and not the view of Merlintrader trading Blog. These opinions may change without notice and may be wrong. Trading and investing in biotech – especially small and mid caps with clinical-stage assets and high short interest – involves a high risk of capital loss and may be unsuitable for many investors. Always consult a qualified financial professional and consider local regulations (including CONSOB and SEC rules) before making any decision.
This content is for informational and editorial purposes only. It is not, and must not be interpreted as, investment advice, a solicitation, or a recommendation to buy or sell any security or financial instrument. All data are based on public sources (company filings, press releases, clinical trial registries and established financial data providers) believed to be reliable, but errors and omissions are always possible. Readers should independently verify all key figures (including dates, cash, revenue, trial status, short interest and regulatory milestones) using primary documents such as SEC filings, official regulatory websites and company disclosures.
Any reference to analyst price targets, ratings or community comments reflects third-party opinions and not the view of Merlintrader trading Blog. These opinions may change without notice and may be wrong. Trading and investing in biotech – especially small and mid caps with clinical-stage assets and high short interest – involves a high risk of capital loss and may be unsuitable for many investors. Always consult a qualified financial professional and consider local regulations (including CONSOB and SEC rules) before making any decision.
Legal resources of Merlintrader trading Blog:
https://merlintrader.com/disclaimer/
https://merlintrader.com/condizioni-duso-e-info-privacy/
My tale (personal view, not a recommendation)
Personally, I see Tango as a high-beta synthetic lethality story where structure matters: a credible scientific platform, a leading PRMT5 program with real pancreatic and lung data, and a CoREST play in one of the toughest immune-evasive NSCLC subsets – all wrapped inside a small-cap with heavy shorts and fresh dilution. The risk/reward is clearly asymmetric in both directions: strong execution and clean pivotal design could justify the 2025 rerating and more, while a mis-step in trial design, safety, or financing could unwind a big part of the move. This is my personal editorial view only and not a recommendation to buy, sell or hold TNGX.
Biotech Catalyst Calendar
For a broader view of upcoming biotech catalysts (PDUFA dates, trial readouts, regulatory events) beyond TNGX, you can consult the dedicated Biotech Catalyst Calendar maintained on Merlintrader.
Ticker: TNGX (NASDAQ)
Tango Therapeutics (TNGX) – PRMT5, CoREST e una base short affollata verso il 2026
Biotech di synthetic lethality con pipeline PRMT5/CoREST, forte rerating nel 2025, short interest elevato e un quadro tecnico che ora si trova sulla base di un canale ascendente proprio alla vigilia del J.P. Morgan 2026.
News & quadro tecnico – Perché TNGX è interessante adesso
Fine dic 2025Dopo un minimo sotto $1, TNGX è esplosa nel 2025, ha toccato nuovi massimi e ora sta ritracciando sulla base di un canale rialzista – proprio mentre si avvicinano dati e conferenze chiave a inizio 2026.
1) Prezzo – base del canale in coincidenza con J.P. Morgan
Dopo il minimo in area $1,10 di maggio 2025, il titolo è salito fino a circa $10,9 a fine novembre 2025. A fine dicembre 2025 TNGX scambia intorno a $8,6–8,7, con market cap in area $1,1–1,2 miliardi, circa il 20% sotto i massimi ma sempre multipli rispetto ai minimi di primavera.
A livello puramente grafico, questa fascia di prezzo (~$8–9) coincide con:
- la linea di tendenza rialzista tracciata dai minimi di maggio e dalla congestione di settembre;
- un’area di forte scambio istituzionale (es. vendite di Third Rock fra $7 e $8).
Se il canale resta intatto, una risalita graduale verso la zona $10–11 entro metà gennaio non sarebbe sorprendente dal punto di vista tecnico – e il timing combacerebbe quasi perfettamente con la presentazione al J.P. Morgan Healthcare Conference del 14 gennaio 2026. Naturalmente i pattern possono sempre fallire: è una lettura grafica, non un segnale operativo.
2) Storia fondamentale – PRMT5 al centro, NSCLC e pancreas come prova del nove
L’asset di punta è vopimetostat (TNG462), inibitore PRMT5 di nuova generazione per pazienti con delezione MTAP. I dati di ottobre 2025 nella fase 1/2 hanno mostrato:
- nel pancreas MTAP-del di seconda linea: mPFS ≈ 7,2 mesi e ORR ≈ 25%;
- attività anche in altri tumori solidi (incluso il polmone);
- piano per uno studio registrazionale globale in 2L pancreas MTAP-del con avvio enrollment nel 2026.
In parallelo TNG260, inibitore CoREST, è in fase 1/2 in combinazione con pembrolizumab in NSCLC con mutazione STK11, una popolazione notoriamente difficile per l’immunoterapia. L’idea complessiva è rimettere in gioco sottogruppi con prognosi scarsa (MTAP-del, STK11-mut) dove le terapie esistenti funzionano male.
3) Cassa, diluizione e narrativa “short squeeze”
Al 31 marzo 2025 Tango riportava circa $216,7M di cassa e titoli, con runway estesa fino a Q1 2027 grazie al taglio di parte della pipeline preclinica. Da allora la società ha:
- annunciato un aumento di capitale e PIPE complessivi per circa $225M nell’ottobre 2025;
- attivato un nuovo programma ATM fino a $100M nel novembre 2025.
In parallelo lo short interest è salito a circa 33 milioni di azioni, pari a circa un quarto dell’intero capitale e oltre il 40% del flottante, con uno short ratio a doppia cifra in giorni di copertura. Questo mix (short molto pesanti + catalyst visibili + chart in uptrend) alimenta ovviamente la narrativa dello squeeze sui canali retail, ma riflette anche una realtà: la storia resta intensamente diluitiva e binaria sul piano clinico.
Dati rapidi – TNGX in una schermata
Snapshot (fine dic 2025)Ticker / MercatoTNGX – Nasdaq
Prezzo azione≈ $8,7 (fine dic 2025)
Range 52 settimane$1,10 – $10,91
Market cap≈ $1,1–1,2 mld
SettoreOncologia / Synthetic lethality
StadioBiotech in fase clinica (no ricavi da farmaci)
Asset principaleVopimetostat (TNG462, inibitore PRMT5)
Sottogruppo targetTumori con delezione MTAP
Secondo pilastroTNG260 (CoREST, NSCLC STK11-mut)
Cassa + titoli (31 mar 2025)~$216,7M
Runway stimataFino a circa Q1 2027
Short interest~33M azioni (~25% OS, >40% float)
Short ratio~10–12 giorni di copertura
Target analisti 12 mesi≈ $12,5–13,25 (range $10–15)
Prossimo eventoJ.P. Morgan 2026 – 14 gennaio 2026
Dati ricavati da comunicati/filing Tango, SEC, StockAnalysis/Yahoo/MarketBeat, Fintel/Nasdaq e clinicaltrials.gov. Valori arrotondati e solo a fini informativi, non per decisioni operative.
Synthetic lethality
MTAP-del
STK11-mut NSCLC
Short interest elevato
Pipeline costosa
Executive summary – Cosa è (e cosa non è) Tango Therapeutics
Contesto per chi non è specialistaTango Therapeutics è una biotech in fase clinica costruita intorno al concetto di synthetic lethality: colpire vulnerabilità genetiche specifiche (MTAP-del, FOCAD-del, STK11-mut, ecc.) in modo selettivo sulle cellule tumorali. È diventata uno dei nomi più discussi del settore grazie ai dati iniziali di vopimetostat (TNG462) e a una community retail piuttosto rumorosa su TNGX.
- Cuore della storia – PRMT5 e delezione MTAP: TNG462 è un inibitore PRMT5 di nuova generazione, progettato per sfruttare la delezione MTAP nelle cellule tumorali. I dati iniziali nella fase 1/2, in particolare nel pancreas di seconda linea, mostrano risposte reali e una mPFS interessante rispetto alla chemioterapia storica.
- Secondo pilastro – CoREST e NSCLC STK11-mut: TNG260 mira al complesso CoREST per “ri-sensibilizzare” i tumori con mutazione STK11 all’anti-PD-1. La fase 1/2 in NSCLC è in espansione, con proof-of-mechanism già dimostrata e dati clinici più maturi attesi nel 2026.
- Pipeline aggiuntiva – TNG456 e TNG961: TNG456 porta PRMT5 nel cervello per tumori MTAP-del (glioblastoma), TNG961 è un degrader HBS1L per tumori con delezione FOCAD; per ora sono soprattutto opzionalità di medio-lungo periodo, ma rafforzano il profilo di piattaforma.
- Cassa e diluizione: la società ha runway ufficiale fino a Q1 2027, ma l’ha estesa e allo stesso tempo diluita con un aumento da ~$225M e un ATM da $100M a fine 2025.
- Short interest e volatilità: con ~33M di azioni short (≈25% delle azioni, >40% del flottante) e un titolo esploso da ~$1 a oltre $10 nel 2025, lo “squeeze trade” è inevitabile nelle discussioni, ma è anche un segnale di rischio percepito elevato.
- Vista degli analisti: le ultime sintesi indicano fra 6 e 10 analisti con rating complessivo Strong/Moderate Buy e target medi a 12 mesi intorno a $12,5–13,25 (range ~$10–15), cioè un upside stimato intorno al 40–45% dai prezzi di fine 2025.
L’obiettivo di questo report è offrire una mappa completa di dati, scenari e rischi, in modo che il lettore possa farsi una propria opinione. Non è in alcun modo una raccomandazione di acquisto, vendita o mantenimento del titolo.
1. Azienda e piattaforma – Synthetic lethality “sul campo”
Dai CRISPR screen ai trial cliniciL’idea di base è usare sistematicamente la synthetic lethality: se un gene chiave è perso nella cellula tumorale (MTAP, FOCAD, STK11, ecc.), colpire il partner sinteticamente letale (PRMT5, HBS1L, CoREST) dovrebbe uccidere il tumore lasciando relativamente indenni i tessuti sani.
1.1 Asse PRMT5 – Vopimetostat (TNG462) e TNG456
PRMT5 è un’arginina metiltransferasi coinvolta nello splicing dell’RNA e nell’espressione genica. Nelle cellule MTAP-del diventa particolarmente critica, rendendola un target ideale per approcci sinteticamente letali. Tango ha abbandonato un inibitore precedente (TNG908) per puntare su vopimetostat (TNG462), più potente e MTA-cooperativo. TNG456 estende la stessa logica nel SNC con un composto progettato per raggiungere concentrazioni cerebrali adeguate nel glioblastoma MTAP-del.
1.2 Asse CoREST – TNG260 per NSCLC STK11-mut
TNG260 prende di mira il complesso CoREST, coinvolto nel rimodellamento della cromatina e nell’evasione immunitaria. I dati preclinici mostrano che l’inibizione di CoREST può ri-sensibilizzare l’NSCLC STK11-mut all’anti-PD-1 con regressioni tumorali nei modelli murini. Da qui il trial di fase 1/2 in combinazione con pembrolizumab in tumori STK11-mut (focus NSCLC).
1.3 Asse HBS1L / FOCAD – TNG961
TNG961 è un degrader “molecular glue” che prende di mira HBS1L in tumori con delezione FOCAD, sottogruppo frequente nelle neoplasie MTAP-del. È ancora in fase preclinica, ma rappresenta il tassello successivo della pipeline oltre PRMT5 e CoREST.
2. Programmi principali e mappa catalyst 2025–2026
TNG462, TNG260, TNG456, TNG9612.1 Vopimetostat (TNG462) – Pancreas e polmone MTAP-del
L’aggiornamento del 23 ottobre 2025 sulla fase 1/2 di TNG462 nei tumori MTAP-del ha evidenziato:
- nel pancreas MTAP-del di seconda linea: mPFS ≈ 7,2 mesi, ORR ≈ 25% (monoterapia);
- attività anche in altri tumori (incluso il polmone), con ulteriori coorti in espansione;
- pianificazione di uno studio registrazionale 2L pancreas MTAP-del nel 2026, con ~300 pazienti randomizzati vs quattro regimi chemioterapici standard.
TNG462 viene inoltre combinato con gli inibitori RAS(ON) di Revolution Medicines (daraxonrasib e zoldonrasib) in pancreas e polmone, con enrollment avviato nel 2025 e dati iniziali attesi nel 2026.
2.2 TNG260 – CoREST per NSCLC STK11-mut
Il trial di fase 1/2 (NCT05887492) valuta TNG260 in combinazione con pembrolizumab in tumori STK11-mut, con focus sull’NSCLC. Dal Q1 2025 sappiamo che:
- il proof-of-mechanism è stato dimostrato su biopsie in trattamento a 80 mg QD;
- il profilo di sicurezza/tollerabilità è per ora favorevole;
- è in corso la coorte di dose-espansione NSCLC, con aggiornamenti clinici attesi in più step (incluso 2026).
2.3 TNG456 – PRMT5 cerebrale
TNG456 punta a portare l’approccio PRMT5 nel glioblastoma e in altri tumori MTAP-del del SNC. Tango ha avviato un trial di fase 1/2 con dose escalation in corso. Il programma:
- espande il franchise PRMT5 in un’indicazione notoriamente difficile (GBM);
- fa da prova tecnica sulla capacità di Tango di sviluppare farmaci synthetic lethality CNS-penetrant.
2.4 TNG961 – Degrader HBS1L
TNG961 resta in fase IND-enabling, con target HBS1L in tumori con delezione FOCAD e overlap con MTAP-del. È ancora lontano da catalyst clinici, ma il concept è rilevante per la storia di piattaforma.
2.5 Timeline sintetica 2025–2026
Timeline (indicativa)
- 12 maggio 2025 – Risultati Q1 2025: aggiornamenti su TNG462/TNG260/TNG456/TNG961; runway estesa a Q1 2027.
- 2H 2025 – Aggiornamenti clinici programmati su TNG462 e TNG260 (incluse presentazioni a congressi come SITC 2025).
- 23 ottobre 2025 – Dati positivi fase 1/2 TNG462; annuncio di piano per studio registrazionale pancreas 2L nel 2026.
- Ott–nov 2025 – Forte aumento di short interest e strutture di funding (aumento ~$225M, nuovo ATM $100M).
- Fine nov 2025 – Nuovo massimo 52 settimane ≈ $10,9 e successivo ritracciamento.
- 14 gennaio 2026 – Presentazione a J.P. Morgan Healthcare Conference (9:45 PST / 12:45 EST).
- 2026 – Avvio previsto dello studio registrazionale di TNG462 nel pancreas 2L MTAP-del e primi dati combinazioni con RAS(ON) e coorti NSCLC allargate.
3. Management & Governance – Chi guida Tango
Barbara Weber e teamTango è guidata da Barbara L. Weber, M.D., President, CEO e co-fondatrice della società. Ha iniziato a costruire Tango all’interno di Third Rock Ventures nel 2015 e la guida come CEO dal 2017, accompagnandola da startup Serie A a biotech quotata con pipeline clinica.
Weber è un’oncologa medica con oltre 25 anni di esperienza in genetica dei tumori e sviluppo di farmaci oncologici. Prima di Tango ha ricoperto ruoli di vertice come:
- Senior Vice President & Global Head, Oncology Translational Medicine in Novartis (2009–2015);
- Vice President, Oncology Discovery & Translational Medicine in GlaxoSmithKline (2005–2009);
- Professore di Medicina e Genetica all’Università della Pennsylvania, dove ha guidato uno dei primi programmi clinici e traslazionali su BRCA1/2 e genetica del tumore della mammella.
Come Venture Partner in Third Rock Ventures ha contribuito alla creazione di Neon Therapeutics (poi acquisita da BioNTech) e Relay Therapeutics, oltre a Tango. Siede nei board di Revolution Medicines, FogPharma, Parabilis Medicines e altre realtà di oncologia.
Ha una laurea in Chimica e una in Medicina (M.D.) presso l’Università di Washington, ha completato la residency in medicina interna a Yale e la fellowship in oncologia medica al Dana-Farber. È membro eletto dell’American Association of Physicians e dell’American Society for Clinical Investigation (di cui è stata Presidente), ed è stata nel board sia di ASCO che di AACR.
Il board è presieduto da Alexis Borisy (chair indipendente) e la maggioranza dei consiglieri è non-esecutiva, elemento che aggiunge un minimo di contrappeso alla forte figura operativa del CEO.
4. Sentiment & retail chatter – Cosa si legge in giro
Reddit, Stocktwits, X (non professionali)Sui canali retail (Reddit, Stocktwits, X) su TNGX si vedono pattern abbastanza ricorrenti.
4.1 Narrativa bullish
- “Best-in-class PRMT5”: molti utenti leggono i dati di TNG462 come potenzialmente superiori agli approcci PRMT5 precedenti, soprattutto nel pancreas 2L, e parlano apertamente di scenario “standard of care” se i dati si confermassero in uno studio registrazionale.
- “Fuel per lo squeeze”: short interest elevato (≈25% delle azioni, >40% del flottante), catalyst (JPM, avvio pivotal, ulteriori dati TNG260) e uptrend tecnico alimentano obiettivi di prezzo molto ambiziosi e narrativa da “diamond hands”.
- Piattaforma e non solo singolo farmaco: una parte della community più tecnica sottolinea che Tango non è solo TNG462, ma un’intera piattaforma di synthetic lethality con CoREST, HBS1L, PRMT5 cerebrale, ecc.
4.2 Narrativa scettica / bear
- Stanchezza da diluizione: dopo aumento + PIPE da ~$225M e nuovo ATM $100M, più la prospettiva di anni di trial, molti si chiedono quante altre volte la società tornerà sul mercato prima di vedere ricavi reali.
- Rischio di execution: passare da una coorte di fase 1/2 a un grande studio registrazionale nel pancreas non è banale: ci sono rischi su design, tollerabilità, potenza statistica e concorrenza.
- Trade affollato: TNGX viene percepita da alcuni come “già amata da tutti”, quindi vulnerabile a correzioni violente se arrivano news non perfette.
Tutto questo viene da trader/investitori retail non professionisti. Può aiutare a capire il clima, ma non è ricerca e non dovrebbe essere usato come base unica per decisioni operative.
Questi link possono includere elementi di affiliazione. Non modificano il contenuto del report e non sono endorsement di servizi specifici.
5. Rischi principali e red flags
Dove la storia può incepparsi5.1 Rischio clinico/regolatorio
Alto TNG462 e TNG260 sono ancora in fase 1/2. Segnali iniziali incoraggianti possono non replicarsi in studi più ampi e controllati; eventuali problemi di sicurezza o efficacia potrebbero cambiare la narrativa in modo drastico.
5.2 Rischio competitivo e commerciale
Medio, guidato dall’execution
- gli standard di cura nel pancreas e nel polmone stanno evolvendo rapidamente (nuovi combo, approcci RAS, immuno, ADC);
- altri player stanno entrando sugli stessi percorsi molecolari o in aree vicine;
- la traduzione da dati di early phase a pratica clinica reale richiede pricing, rimborsi e dati di vita reale convincente.
5.3 Cassa, diluizione e funding
Alto Anche con runway dichiarata fino a Q1 2027, la combinazione di studi registrazionali, combinazioni e pipeline ampia implica un rischio concreto di ulteriori aumenti di capitale o strutture di finanziamento non banali.
5.4 Short interest e volatilità
Medio–Alto Short interest elevato e trade affollato significano:
- movimenti molto ampi in entrambe le direzioni su news e rumor;
- rischio di over-reaction a ogni aggiornamento non perfetto;
- necessità di dimensionare con attenzione qualunque posizione (long o short).
6. Mappa scenari – Vista neutrale
Nessuna raccomandazione6.1 Scenario costruttivo (cosa dovrebbe andare bene)
- TNG462 conferma beneficio clinico robusto in pancreas MTAP-del e avanza in uno studio registrazionale ben disegnato.
- Emergono segnali convincenti in NSCLC (TNG462 e/o TNG260), tali da giustificare percorsi registrazionali successivi.
- Viene siglata una o più partnership strategiche (es. diritti ex-USA o co-sviluppo) che portano cassa non diluitiva.
- La diluizione futura avviene a valorizzazioni progressivamente più alte e non in fasi di debolezza marcata.
6.2 Scenario prudente / downside (cosa può rompersi)
- dati meno brillanti del previsto o safety più complessa nelle coorti allargate di TNG462/TNG260;
- concorrenti che si muovono più velocemente in pancreas o polmone;
- ulteriori aumenti di capitale/ATM in un contesto di prezzo debole che erodono il potenziale per azione;
- disallineamento fra aspettative della community e realtà dei tempi/regolatori, con conseguente compressione del multiplo.
Anche qui si tratta di scenari, non previsioni. Servono solo a mappare le strade possibili, non a suggerire operazioni.
7. Fonti e letture di approfondimento
Solo link verificabili-
Comunicato Q1 2025 Tango Therapeutics (GlobeNewswire, 12 maggio 2025):
https://www.globenewswire.com/news-release/2025/05/12/3078966/0/en/… -
Form 8-K Q1 2025 (SEC, bio e dati finanziari):
https://ir.tangotx.com/static-files/667e56fb-23d5-43ef-a0aa-42848637045e -
Comunicato vopimetostat (TNG462) fase 1/2 MTAP-del (23 ottobre 2025):
https://www.globenewswire.com/news-release/2025/10/23/3171895/0/en/… -
Articolo BioWorld “Pancreatic Revolution due? Tango PRMT5 data satisfy” (24 ottobre 2025):
https://www.bioworld.com/articles/725397-pancreatic-revolution-due-tango-prmt5-data-satisfy -
Paper preclinico TNG260 – Cancer Research (2025):
https://aacrjournals.org/cancerres/article/85/20/3966/766095/… -
Trial TNG260 + pembrolizumab (NCT05887492, clinicaltrials.gov):
https://clinicaltrials.gov/study/NCT05887492 -
PR partecipazione J.P. Morgan 2026 (17 dicembre 2025):
https://www.globenewswire.com/news-release/2025/12/17/3206852/0/en/… -
Target analisti e forecast:
StockAnalysis – https://stockanalysis.com/stocks/tngx/forecast/
Yahoo Finance – https://finance.yahoo.com/quote/TNGX/analysis/
MarketBeat – https://www.marketbeat.com/stocks/NASDAQ/TNGX/price-target/ -
Approfondimenti su Barbara Weber:
Bio SEC nel 10-K – 10-K Tango 2021–2022
Parabilis / ITM – https://parabilismed.com/board/barbara-weber-md/, https://www.itm-radiopharma.com/company/supervisory-board/
Disclaimer educativo / legale (IT)
Questo contenuto ha finalità esclusivamente informative ed editoriali. Non costituisce in alcun modo consulenza finanziaria, sollecitazione al pubblico risparmio, offerta o raccomandazione personalizzata di investimento. I dati sono tratti da fonti pubbliche (filing societari, comunicati, registri trial, provider di dati finanziari) ritenute affidabili, ma errori od omissioni sono sempre possibili. Il lettore deve verificare in autonomia tutte le informazioni rilevanti (date, importi, stati regolatori, short interest, ecc.) su documenti ufficiali (SEC, siti regolatori, comunicati aziendali).
I riferimenti a target di prezzo, rating o commenti di community riflettono opinioni di terzi e non l’opinione di Merlintrader trading Blog. Il trading/investing in titoli biotech – in particolare small e mid cap con pipeline in fase clinica e forte leva short – comporta un rischio elevato di perdita del capitale ed è spesso inadatto a molti investitori. Prima di qualsiasi decisione è opportuno rivolgersi a un consulente finanziario abilitato e considerare le normative applicabili (incluse quelle CONSOB e SEC).
Questo contenuto ha finalità esclusivamente informative ed editoriali. Non costituisce in alcun modo consulenza finanziaria, sollecitazione al pubblico risparmio, offerta o raccomandazione personalizzata di investimento. I dati sono tratti da fonti pubbliche (filing societari, comunicati, registri trial, provider di dati finanziari) ritenute affidabili, ma errori od omissioni sono sempre possibili. Il lettore deve verificare in autonomia tutte le informazioni rilevanti (date, importi, stati regolatori, short interest, ecc.) su documenti ufficiali (SEC, siti regolatori, comunicati aziendali).
I riferimenti a target di prezzo, rating o commenti di community riflettono opinioni di terzi e non l’opinione di Merlintrader trading Blog. Il trading/investing in titoli biotech – in particolare small e mid cap con pipeline in fase clinica e forte leva short – comporta un rischio elevato di perdita del capitale ed è spesso inadatto a molti investitori. Prima di qualsiasi decisione è opportuno rivolgersi a un consulente finanziario abilitato e considerare le normative applicabili (incluse quelle CONSOB e SEC).
Risorse legali Merlintrader trading Blog:
https://merlintrader.com/disclaimer/
https://merlintrader.com/condizioni-duso-e-info-privacy/
My tale (opinione personale, non raccomandazione)
In chiave personale vedo Tango come una storia di synthetic lethality ad altissimo beta: piattaforma credibile, un PRMT5 con numeri interessanti nel pancreas MTAP-del, un asse CoREST in uno dei sottogruppi NSCLC più ingrati da trattare – ma anche short interest pesante e storia di funding non banale. Se execution, design degli studi e safety reggono, il rerating 2025 può avere ancora spazio; se uno di questi pilastri scricchiola (dati, regolatorio o cassa), il downside può essere violento. È una mia lettura editoriale, non una raccomandazione operativa su TNGX.
Biotech Catalyst Calendar
Per una panoramica più ampia delle prossime date chiave biotech (PDUFA, readout di trial, eventi regolatori), puoi fare riferimento al Biotech Catalyst Calendar mantenuto su Merlintrader.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.



