DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
ATRA Atara Biotherapeutics Inc JAN 2 Update

Atara Biotherapeutics is a small-cap cell therapy company whose equity story is now essentially a single binary catalyst: the U.S. FDA decision on tab-cel (tabelecleucel) for EBV-positive PTLD, with a Priority Review PDUFA action date on 10 January 2026. The company has already divested or discontinued its other programs and has transferred most tab-cel operational responsibilities to Pierre Fabre Laboratories. What remains for shareholders is a high-volatility, high-uncertainty exposure to the outcome of that regulatory event and its knock-on impact on liquidity and solvency.
ALDX Aldeyra Therapeutics Update dec 12

Aldeyra Therapeutics is a clinical-stage biotech focused on inflammation, oxidative stress and ocular / metabolic diseases via its RASP platform. The lead asset, Reproxalap, is currently under FDA review for dry eye disease with a target action date of December 16, 2025 — a highly binary event coming after previous Complete Response Letters (CRLs) in 2023 and 2025.
OMER The Path of the Force Update dec 9

Omeros Corporation is a US-based biotechnology company focused on complement-related diseases. Its lead asset, narsoplimab (OMS721), targets transplant-associated thrombotic microangiopathy (TA-TMA), with a PDUFA target action date of December 26, 2025 for its resubmitted BLA. This is the core binary catalyst currently driving the stock.
SNTI Senti Biosciences Inc

Senti Biosciences is a micro-cap cell therapy company built around programmable gene-circuit technology. The lead asset, SENTI-202, is an off-the-shelf, logic-gated CAR-NK therapy in Phase 1 for relapsed / refractory acute myeloid leukemia (R/R AML).
VOR BIOPHARMA
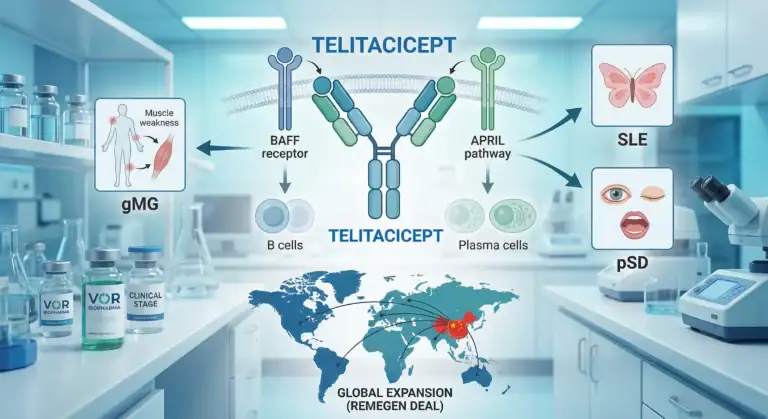
VOR transformed from a dying cell therapy company into a focused autoimmune player with a globally licensed, China-approved dual BAFF/APRIL inhibitor (telitacicept). The stock has crashed/rallied 5 times in 2025 alone.
XCUR Exicure Inc

Exicure has repositioned itself around burixafor, a CXCR4 antagonist designed to mobilize hematopoietic progenitor cells (HPCs) for autologous stem-cell transplant in multiple myeloma and potentially other settings. At the ASH 2025 annual meeting, the company presented Phase 2 data showing high rates of successful HPC collection, including in daratumumab-exposed patients who are traditionally harder to mobilize.
PHAR Pharming Group (Updated dec 19)

Pharming Group has quietly moved into an unusual position in the biotech universe: it is a profitable, cash-generating rare-disease company, yet still trades in the small/mid-cap bracket and continues to carry several meaningful catalysts.
AQST Aquestive Therapeutics

Aquestive Therapeutics is a small-cap specialty pharma player built around its PharmFilm oral film technology and a mix of manufacturing revenue and proprietary pipeline assets. The stock is now tightly linked to a single binary catalyst: the FDA decision on Anaphylm, its epinephrine sublingual film for severe allergic reactions.
TVTX Travere Therapeutics

Travere Therapeutics is a commercial-stage rare-disease biotech focused on kidney and metabolic disorders. Its lead product, FILSPARI (sparsentan), is approved in the United States for IgA nephropathy (IgAN) and is under FDA review for focal segmental glomerulosclerosis (FSGS). A supplemental NDA for FSGS has an FDA target action date in early 2026, turning TVTX into a classic binary catalyst name with an already meaningful revenue base from IgAN.
FULC Fulcrum Therapeutics
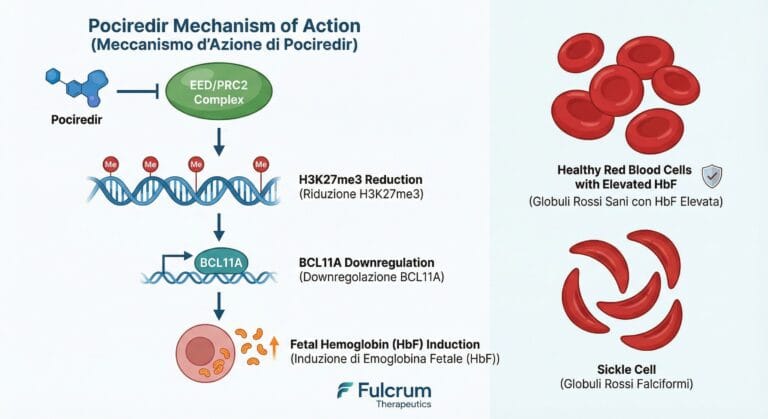
Fulcrum Therapeutics (FULC) – Pociredir SCD Deep Dive | Merlintrader trading Blog EN IT Fulcrum Therapeutics (FULC) – Pociredir in Sickle Cell Disease Deep Dive (December 2025) Last updated: December 8, 2025 – Authors: Merlintrader and Jane – Educational, non-promotional…
INVA Innoviva Inc
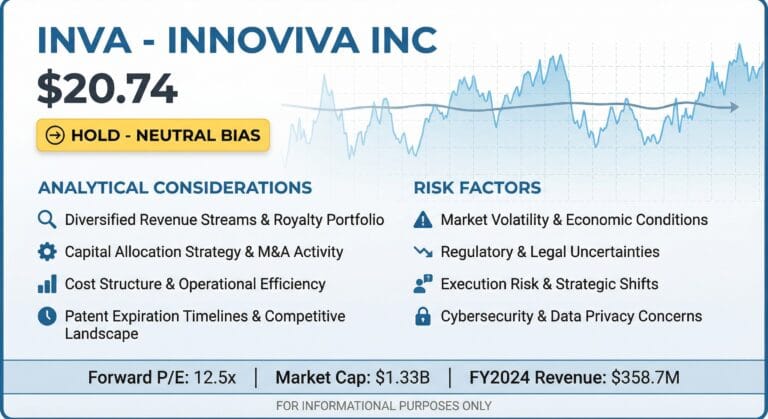
Innoviva Inc (NASDAQ: INVA) is a diversified holding company with a core royalty portfolio derived from respiratory assets in partnership with GlaxoSmithKline (GSK) and a growing commercial platform focused on critical care and infectious disease therapies through Innoviva Specialty Therapeutics (IST).
PDUFA dates december 2025
December 2025 is the densest FDA decision month of the year. In only 26 days the agency is expected to rule on fourteen key dossiers across oncology (Breyanzi), cardiology (Aficamten), ophthalmology (EYLEA HD, LYTENAVA), rare diseases (Narsoplimab, Mitapivat), endocrine disorders (Relacorilant), infectious diseases (Zoliflodacin) and more.
PDSB PDS Biotechnology Corporation

On December 3, 2025, PDS Biotech announced via an official 8-K filing that the FDA has granted a Type C Meeting to discuss a potential accelerated approval pathway based on PFS for PDS0101 + pembrolizumab in first-line HPV16+ HNSCC.
The market reaction was immediate: the stock gained roughly +18% on Dec 3 and closed at $0.899 on Dec 4, about +12% across 48 hours.
HOTH Hoth Therapeutics Inc

Hoth Therapeutics is a micro-cap, clinical-stage biotech focused on oncology-supportive care, mast-cell–driven diseases, neurodegeneration and metabolic/obesity indications. The company just issued a comprehensive pipeline update covering four core programs: HT-001 (topical therapy for EGFR-inhibitor rash), HT-KIT (KIT-driven mast-cell diseases, Orphan Drug Designation), HT-ALZ (Alzheimer’s / CNS inflammation) and a newly launched GDNF-based metabolic program in collaboration with the Atlanta VA Medical Cente
PLRZ Polyrizon Ltd

Polyrizon Ltd is an early-stage biotech leveraging proprietary Capture & Contain (C&C) hydrogel technology for nasal barriers against allergens/viruses and Trap & Target (T&T) for enhanced intranasal drug delivery
MREO Mereo BioPharma

LONDON, Nov. 10, 2025 (GLOBE NEWSWIRE) -- Mereo BioPharma Group plc (NASDAQ:MREO) ("Mereo" or the "Company"), a clinical-stage biopharmaceutical company focused on rare diseases, today announced its financial results for the third quarter ended September 30, 2025, and provided recent corporate highlights.
ALT Altimmune Update dec 3
Altimmune is entering a pivotal phase with several bullish drivers converging through Q4 2025 and into 2026
ADCT ADC Therapeutics
ADC Therapeutics announced updated LOTIS-7 Phase 1b trial results on December 3, 2025, showing highly encouraging efficacy and manageable safety for ZYNLONTA + glofitamab combination in relapsed/refractory diffuse large B-cell lymphoma (DLBCL)
JANX

Janux Therapeutics has gone from market darling to high-risk story stock in 2025:
52-Week High (early 2025): around US$71–72 per share (after strong early JANX007 data and sector momentum).
Price after December 2–3, 2025 update: mid-teens (≈US$16–17), following a two-day collapse of ~50% after the latest Phase 1 JANX007 update.
CAPR Capricor Therapeutics (CAPR)

Positive Phase 3 HOPE-3 topline results in Duchenne muscular dystrophy cardiomyopathy with statistically significant skeletal and cardiac benefit, reopening the regulatory path after the 2025 CRL.