DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
GUTS Fractyl Health Inc

Fractyl Health (NASDAQ: GUTS) is emerging as one of the most interesting names in the metabolic space thanks to its flagship device Revita®, a duodenal mucosal resurfacing (DMR) procedure designed to maintain weight loss after stopping GLP-1 drugs.
VERA Vera therapeutics

VERA Therapeutics is a biopharmaceutical company in advanced clinical stage focused on serious immunological diseases. The company achieved its most important milestone in November 2025 with the submission of a Biologics License Application (BLA) to the FDA for atacicept, its lead candidate for the treatment of IgA Nephropathy (IgAN).
VSTM Verastem Inc

Verastem Oncology is transitioning from a late-stage developer into a commercial oncology company focused on RAS/MAPK pathway-driven cancers.
PMCB PharmaCyte Biotech
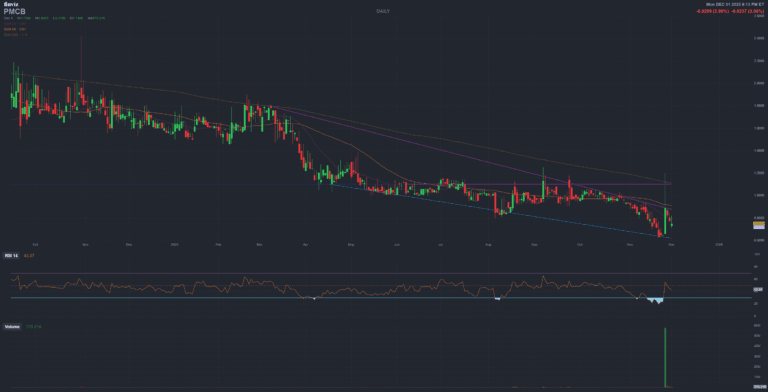
PharmaCyte has successfully monetized its equity stake in Femasys Inc. (FEMY), increasing its cash and marketable securities to roughly ~20 million USD, up from about 13.3 M USD as of July 31, 2025, according to the company’s press release and BusinessWire distribution.
CTOR Citius Oncology Inc (ctxr)
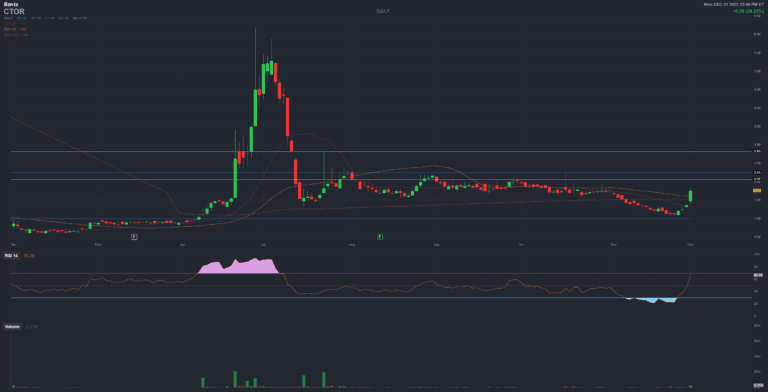
FDA approval secured June 2024 with orphan drug exclusivity through 2031
$8.27M inventory ready for immediate commercialization
Strong clinical efficacy: 36.2% ORR, 84.4% tumor burden reduction
RVPH Reviva Pharmaceuticals

FDA Pre-NDA Meeting (Schizophrenia)
TIMEFRAME: Q4 2025 (IMMINENT)
The Event: Reviva has a scheduled meeting with the FDA in Q4 2025 to discuss submitting a New Drug Application (NDA) for Brilaroxazine based solely on existing data (RECOVER-1 Phase 3 + OLE Long-term data).
AGIO BCRX MIST December 7-13, 2025: Three Biotech PDUFA Catalysts – Complete Analysis

AGIO, BCRX, MIST: December 2025 PDUFA Deep Dive | Analyst Targets & Timeline
Weekly Market Analysis & Trading Outlook
The week of November 24-29 closed with continued volatility as investors digested stretched AI valuations, mixed macro signals, and elevated geopolitical tensions. Major indexes ended modestly lower: S&P 500 [finance:S&P 500] -1.9%, Nasdaq [finance:Nasdaq Composite] -2.7%, Dow [finance:Dow Jones Industrial Average] -1.9%.
LXRX Lexicon Pharmaceuticals Inc
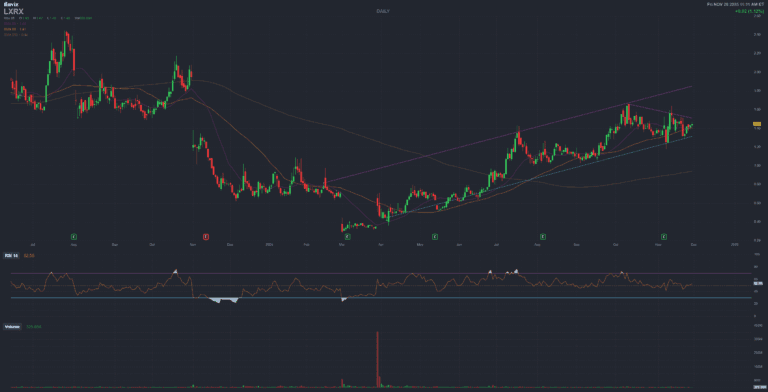
Lexicon Pharmaceuticals, Inc. (NASDAQ: LXRX) is a biopharmaceutical company focused on the discovery, development, and commercialization of pharmaceutical products using its proprietary gene science approach. The company operates in two segments: Product (INPEFA® commercialization) and Licensing (Novo Nordisk partnership for LX9851 obesity program).
ONMD OneMedNet Corporation
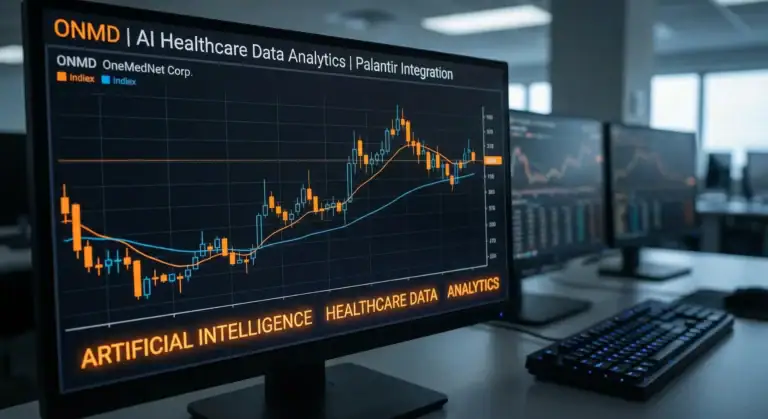
OneMedNet Corporation (NASDAQ: ONMD) is a healthcare technology company specializing in curating and providing regulatory-grade Real-World Data (RWD) through its proprietary iRWD™ platform. The company connects life sciences organizations, medical device companies, and AI researchers with de-identified medical imaging, electronic health records, and clinical data from over 2,130 healthcare provider sites spanning 47+ million unique patient lives and 186+ million clinical exams.
MBOT Microbot Medical Inc.
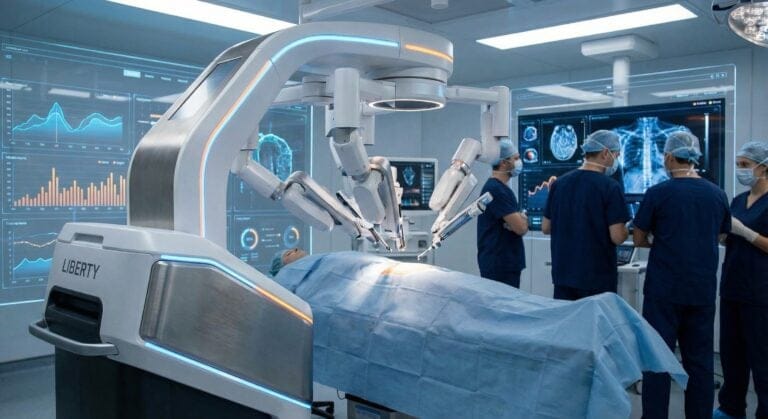
Microbot Medical is a commercial-stage medical device company specializing in endovascular robotic systems. The company achieved a transformational milestone in September 2025 with FDA 510(k) clearance for its flagship LIBERTY® Endovascular Robotic System - the world's first FDA-cleared single-use, remotely operated robotic platform for peripheral endovascular procedures.
VRCA Verrica Pharmaceuticals Inc
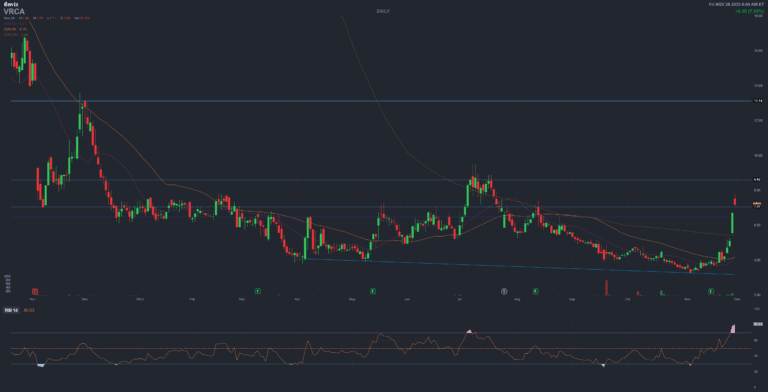
Verrica Pharmaceuticals [finance:Verrica Pharmaceuticals Inc.] (NASDAQ: VRCA) is a commercial-stage dermatology biopharmaceutical company focused on developing and commercializing innovative medical aesthetic solutions for skin conditions including molluscum contagiosum, basal cell carcinoma, and aesthetic dermatology indications. The company operates through a partnership model, leveraging strategic alliances with larger pharmaceutical companies (Torii Pharmaceutical [finance:Torii Pharmaceutical Co., Ltd.]) for commercialization and geographic expansion.
ACRS Aclaris Therapeutics Inc

Aclaris Therapeutics, Inc. (NASDAQ: ACRS) is a clinical-stage dermatology-focused biopharmaceutical company developing innovative immunological therapies for inflammatory and immune-mediated skin diseases. The company transitioned from a commercial-stage company with FDA-approved products to a clinical-stage organization, strategically acquiring promising pipeline assets to build a competitive portfolio in atopic dermatitis and related indications.
FLGT Fulgent Genetics, Inc.

Diagnostics + Therapeutics | Genetic Testing & Cancer Drug Development | Q3 2025 Report
XLO Xilio Therapeutics Inc Report

Xilio Therapeutics, Inc. (NASDAQ: XLO) is a clinical-stage biopharmaceutical company pioneering the development of masked tumor-activated immunotherapies
AMRN Amarin Corp

? Amarin Corporation (AMRN) – Company History & Analysis ⚠️ IMPORTANT NOTICE / AVVISO IMPORTANTE: This document is a company analysis and financial summary compiled from publicly available sources (SEC filings, press releases, clinical publications). It is NOT investment advice,…
AVXL Anavex Life Sciences

What happened: The Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (EMA) informed Anavex of a negative trend vote on the Marketing Authorisation Application (MAA) for blarcamesine following its oral explanation on November 11, 2025.
NRXP

NRx Pharmaceuticals (NRXP) | Investment Research Report ⚠️ IMPORTANT NOTICE / AVVISO IMPORTANTE: This document is a news digest and company profile compiled from publicly available sources (SEC filings, press releases, official company announcements). It is NOT investment advice, NOT…
CUE Biopharma
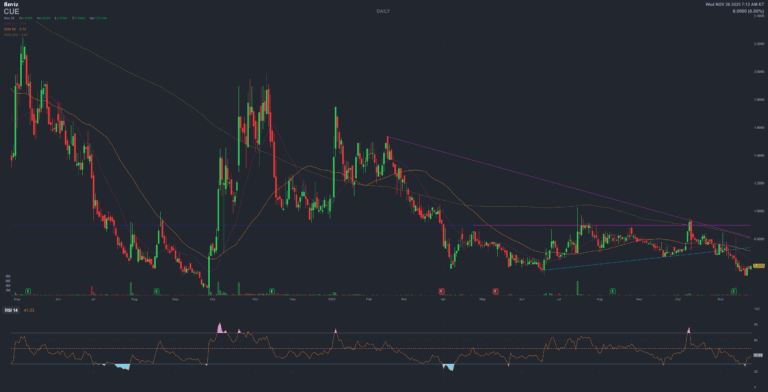
Cue Biopharma (CUE) | Investment Research Report ?? English Version ? Cue Biopharma (CUE) Investment Research Report — Platform Play: Immuno-STAT T-Cell Modulators Update: November 26, 2025 ⚠️ GOING CONCERN WARNING ? STRATEGIC PIVOT TO AUTOIMMUNE NASDAQ: CUE | Price:…
KALA Bio Caution risk 100%
KALA Bio was a clinical-stage biotech developing KPI-012, a mesenchymal stem cell secretome (MSC-S) therapy for Persistent Corneal Epithelial Defect (PCED), an orphan eye disease with ~100,000 US patients and no FDA-approved treatments.