DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
? Technical Analysis: Biotech Stocks Ahead of December PDUFA
⚠️ **Important Disclaimer:** Biotech PDUFA events are fundamental catalysts that often override technical analysis. Technical levels are only theoretical targets based on chart patterns and price action *in the absence of fundamental news*.
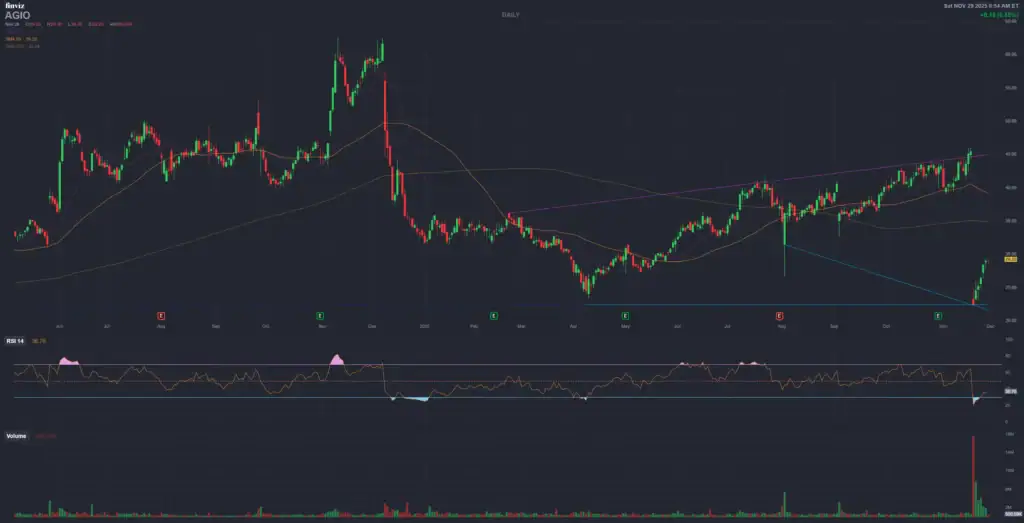
1. ? Agios Pharmaceuticals, Inc. (AGIO)
Catalyst Event: PDUFA for **PYRUKYND® (mitapivat)** in thalassemia (label expansion) – **December 7, 2025**.
Technical Context (Daily/Weekly Chart): The stock experienced a **sharp decline** after mixed results in another study, breaking key supports. The price is **significantly below** the 200-day Moving Average (around $40.79), indicating a strong long-term bearish trend. The RSI is likely in or emerging from the **oversold** area, suggesting a potential technical bounce or that the primary selling pressure has subsided.
| Technical Level | Theoretical Price (USD) | Role |
|---|---|---|
| **Key Support** | $26.00 – $27.00 | Recent low/historical support level that could serve as a base for pre-PDUFA buying pressure. |
| **Immediate Resistance** | **$32.00 – $33.00** | Psychological and former support level that the price must overcome to reverse short-term sentiment. |
| **Bullish Target (Approval)** | **$40.00 – $42.00** | Corresponds to the **200-day Moving Average** and previous resistance/pre-fall high; a realistic target on a positive outcome. |
| **Bearish Target (Non-Approval)** | $20.00 – $22.00 | Subsequent historical low/support level in case of a strong sell-off. |
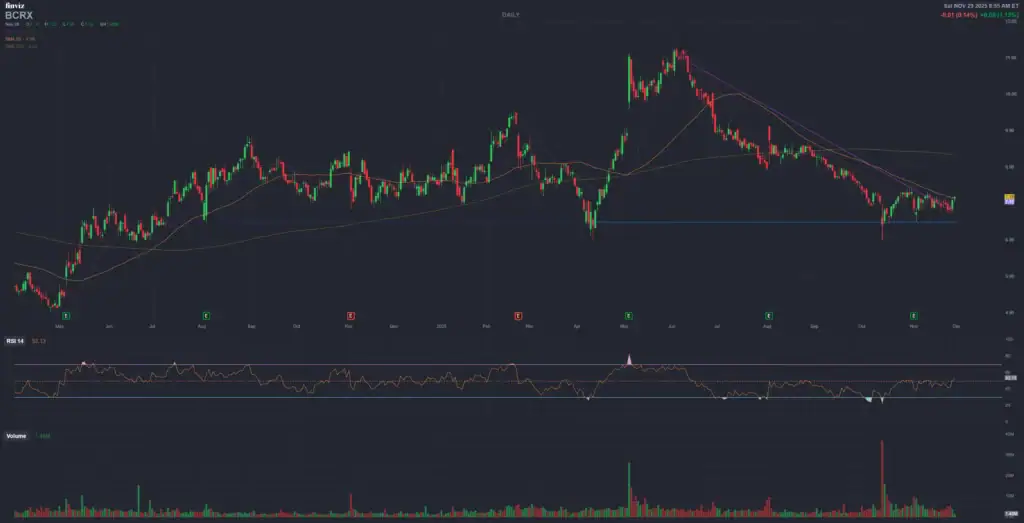
2. ? BioCryst Pharmaceuticals, Inc. (BCRX)
Catalyst Event: PDUFA for **ORLADEYO®** (berotralstat) in pediatric HAE (label expansion) – **December 12, 2025**.
Technical Context (Daily Chart): The stock is trading in a tight sideways range or a **mild short-term uptrend**, generally holding **near or above** key Moving Averages (50 and 200 days, with the 200-day MA around $7.064). Indicators like RSI and MACD are likely showing mild buy signals, suggesting increasing **accumulative pressure** ahead of the news. Analyst consensus remains strong (“Strong Buy”) with a 12-month average target significantly above the current price.
| Technical Level | Theoretical Price (USD) | Role |
|---|---|---|
| **Key Support** | $6.50 – $6.70 | Base of the recent trading range and strong support in case of pre-PDUFA profit-taking. |
| **Immediate Resistance** | $7.20 – $7.40 | Recent resistance level that, if broken, could trigger a move in anticipation of the event. |
| **Bullish Target (Approval)** | **$10.00 – $12.00** | Significant psychological level and previous historical peak/resistance. Breaking this opens the path to analyst targets. |
| **Bearish Target (Non-Approval)** | $5.00 – $5.50 | Significant historical support; a drop below the 200-day MA (around $7.06) would indicate a rapid move toward these lows. |
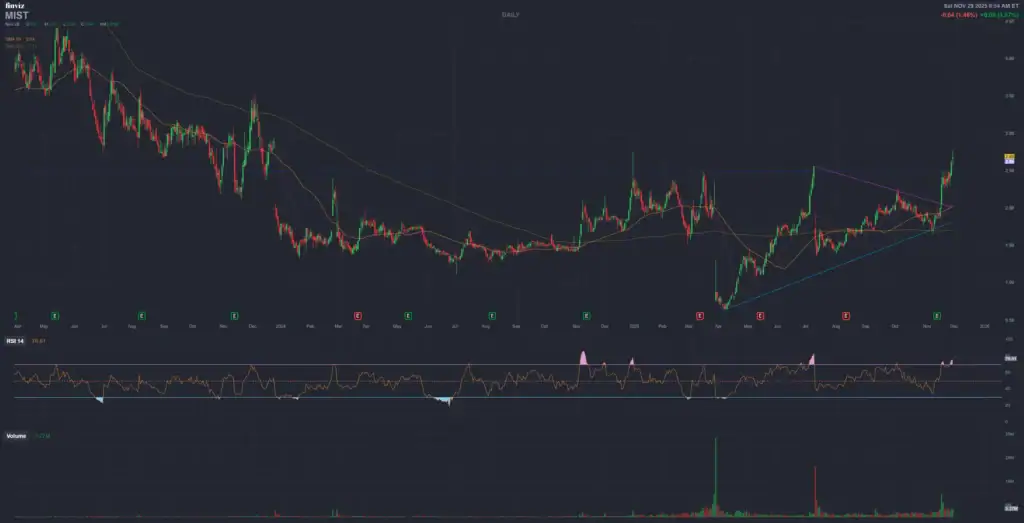
3. ✨ Milestone Pharmaceuticals Inc. (MIST)
Catalyst Event: PDUFA for **CARDAMYST™** (etripamil) nasal spray (after CRL) – **December 13, 2025**.
Technical Context (Daily/Weekly Chart): The stock has shown **high volatility**, especially after a public offering caused a crash, followed by a strong recovery (52-week high indicated at $2.77). The price is in a **recovery trend** from historical lows, indicating buyers are dominating the short-term trend. Breaking the 52-week high resistance ($2.77) is crucial to confirm sustained bullish momentum (buy-in) before the event.
| Technical Level | Theoretical Price (USD) | Role |
|---|---|---|
| **Key Support** | $2.00 – $2.20 | Crucial psychological level that acted as a base during the recent recovery. |
| **Immediate Resistance** | **$2.75 – $3.00** | 52-week high and key psychological resistance. A break indicates strong pre-PDUFA momentum. |
| **Bullish Target (Approval)** | **$3.75 – $4.50** | Aligns with the **Average Analyst Price Target** ($3.75) and a previous trading zone before the crash. |
| **Bearish Target (Non-Approval)** | $1.00 – $1.50 | In case of a negative outcome (another CRL), the stock would likely fall back to or near historical lows with a potentially violent sell-off. |
? December 2025 FDA Catalysts Deep Dive
AGIO · BCRX · MIST | PDUFA Dates, Analyst Targets, SEC Filings, News Timeline
$AGIO – Agios Pharmaceuticals | Dec 7 PDUFA
PDUFA Dec 7 Thalassemia Small-CapCatalyst Overview
Drug: PYRUKYND® (mitapivat)
Indication: Supplemental New Drug Application (sNDA) for treatment of adult patients with alpha- or beta-thalassemia (both transfusion-dependent and non-transfusion-dependent)
Mechanism: First-in-class oral pyruvate kinase (PK) activator
PDUFA Date: December 7, 2025 (extended from September 7, 2025)
Background & Timeline
Mitapivat is an oral, allosteric activator of the red blood cell pyruvate kinase enzyme. Phase 3 ENERGIZE and ENERGIZE-T trials demonstrated significant hemoglobin improvements and reduced transfusion burden in thalassemia patients.
FDA Extended PDUFA: On September 4, 2025, Agios announced the FDA extended the PDUFA goal date by three months (from September 7 to December 7, 2025) after the company submitted a proposed Risk Evaluation and Mitigation Strategy (REMS) to address hepatocellular injury risk observed in clinical trials. [Globe Newswire, Sept 4, 2025]
Recent Clinical Data (ASH 2025)
At the 67th American Society of Hematology (ASH) Annual Meeting (December 6-9, 2025 in Orlando), Agios presented multiple abstracts highlighting mitapivat’s potential to:
- Significantly reduce transfusion needs in thalassemia patients
- Provide early evidence of protection against heart complications (cardiomyopathy risk)
- Demonstrate sustained efficacy in both transfusion-dependent and non-transfusion-dependent populations
[Source: Simply Wall St, Nov 24, 2025]
Sickle Cell Disease Data (Mixed Results)
On November 20, 2025, Agios reported mixed topline results from its Phase 3 RISE UP trial evaluating mitapivat in sickle cell disease (SCD):
- ✅ Primary Endpoint Met: 40.6% of patients receiving mitapivat showed ≥1.0 g/dL increase in hemoglobin levels vs 2.9% placebo
- ❌ Pain Crisis Endpoint Missed: Did not demonstrate significant reduction in annualized rate of sickle cell pain crises vs placebo (weak trend only)
[Source: Investing.com, Nov 20, 2025]
Analyst Ratings & Price Targets
| Firm | Rating | Price Target | Date |
|---|---|---|---|
| Leerink Partners | Outperform (Upgrade from Market Perform) | $34.00 | Nov 20, 2025 |
| Consensus (Multiple Analysts) | Moderate Buy | $25.00 – $48.00 range | Nov 2025 |
Leerink’s Rationale: The firm identified the December 7 thalassemia PDUFA as a near-term catalyst not reflected in current share prices. They noted potential approval in a broader population than competitor luspatercept and possible commercialization partnership in key markets.
SEC Filings & Recent Activity
- Form 10-Q (Q3 2025): Filed October 30, 2025 – Quarterly results for period ended September 30, 2025 [SEC EDGAR]
- Form 8-K: September 4, 2025 – PDUFA extension announcement [Stock Titan]
- Insider Activity: November 11, 2025 – Chief Medical Officer exercised options at $25.01, sold 2,454 shares at $43.78 under 10b5-1 plan [Form 4, Stock Titan]
Market Position & Competition
If approved, PYRUKYND would compete with luspatercept (Reblozyl, Bristol Myers Squibb) in the thalassemia market. Analysts note mitapivat’s oral administration (vs subcutaneous injection for luspatercept) as a potential differentiator, along with broader population eligibility.
⚠️ Key Risk: Regulatory delay risk persists given the FDA’s REMS requirement related to hepatocellular injury risk. Third PDUFA extension after September delay would significantly impact timeline and investor sentiment.
$BCRX – BioCryst Pharmaceuticals | Dec 12 PDUFA
PDUFA Dec 12 Pediatric HAE Commercial StageCatalyst Overview
Drug: ORLADEYO® (berotralstat) oral granules
Indication: Long-term prophylaxis of hereditary angioedema (HAE) attacks in pediatric patients aged 2 to under 12 years
Mechanism: Oral plasma kallikrein inhibitor
PDUFA Date: December 12, 2025 (extended from earlier 2025 date)
Background & Clinical Data
ORLADEYO is already approved and commercially available for HAE prophylaxis in adults and pediatric patients 12 years and older. The current PDUFA is for label expansion into younger pediatric patients (ages 2-11) using an oral granule formulation suitable for this age group.
APeX-P Trial (Phase 2): At the American College of Allergy, Asthma & Immunology (ACAAI) Annual Scientific Meeting (November 6-10, 2025 in Orlando), BioCryst presented a second interim analysis from the APeX-P trial showing:
- Positive long-term prophylaxis results with oral granule formulation
- Reduction in HAE attack rates in children aged 2-11
- Favorable safety profile consistent with adult data
[Source: Globe Newswire, Oct 23, 2025]
Revenue & Commercial Performance
Q3 2025 Results (Reported Nov 3, 2025):
- ORLADEYO net revenue: $159.1 million (+37% year-over-year)
- Operating profit: $29.6 million (+285% y-o-y)
- Non-GAAP operating profit: $51.7 million (+107% y-o-y)
- FY 2025 guidance raised: $590-600 million (from prior guidance)
[Source: BioCryst Q3 2025 Earnings, Globe Newswire, Nov 3, 2025]
Strategic Transactions
- European Business Sale: Completed sale of European ORLADEYO business; proceeds used to retire all Pharmakon term debt (now debt-free)
- Astria Therapeutics Acquisition: Definitive agreement signed to acquire Astria Therapeutics; transaction expected to close Q1 2026. This acquisition targets expanding HAE portfolio and driving sustainable growth.
Analyst Ratings & Price Targets
| Firm | Rating | Price Target | Date |
|---|---|---|---|
| Consensus (13 Analysts) | Moderate Buy | $19.18 | Nov 2025 |
| Price Target Range | – | $8.00 – $30.00 | Nov 2025 |
| JMP Securities | Market Outperform | $16.00 (raised from $15) | Aug 6, 2025 |
| Barclays | Hold | $7.00 (raised from $6) | Aug 6, 2025 |
[Sources: MarketBeat, CNN Business]
Analyst Perspective: The November 6 positive pediatric data announcement and upcoming FDA decision have increased confidence in near-term approval. Pediatric expansion addresses a significant unmet need and extends ORLADEYO’s addressable market without cannibalization risk.
SEC Filings & Recent Activity
- Form 8-K: November 2, 2025 – Q3 2025 earnings report and business update [Stock Titan]
- FDA Review Status: FDA extended PDUFA by 3 months to review additional formulation data and final reports for pediatric dosing
Market Opportunity
Pediatric HAE patients (ages 2-11) represent a significant underserved population. Current treatment options for this age group are limited, primarily consisting of on-demand therapies or injectable prophylaxis. ORLADEYO oral granules would be the first oral prophylactic option for this population.
Commercial Advantage: BioCryst is a commercial-stage company with existing ORLADEYO infrastructure, reducing execution risk vs pure pipeline plays. Pediatric approval is an incremental expansion, not a make-or-break event.
$MIST – Milestone Pharmaceuticals | Dec 13 PDUFA
PDUFA Dec 13 PSVT ResubmissionCatalyst Overview
Drug: CARDAMYST™ (etripamil) nasal spray
Indication: Treatment of paroxysmal supraventricular tachycardia (PSVT) – rapid heart rhythm condition
Mechanism: Novel, self-administered calcium channel blocker nasal spray
PDUFA Date: December 13, 2025
Background & Regulatory History
This is a resubmission following a Complete Response Letter (CRL). The FDA accepted Milestone’s response to the CRL on July 11, 2025, setting the new PDUFA target date of December 13, 2025.
Previous CRL (March 2025): The CRL cited chemistry, manufacturing, and controls (CMC) issues – notably, NO efficacy or safety concerns were raised. This is a critical distinction, as CMC-only CRLs typically have higher approval probability upon resubmission compared to efficacy/safety-related CRLs.
[Source: Globe Newswire, July 11, 2025]
Recent Clinical Data (AHA 2025)
At the American Heart Association (AHA) Scientific Sessions 2025 (November 10, 2025), Milestone presented new analysis of etripamil clinical data from >600 unique enrolled patients showing:
- Aligned efficacy and safety across multiple study phases
- Consistent performance across different trial designs (controlled vs open-label)
- Consistent results across multiple geographic regions
- Etripamil treatment achieved consistently greater conversion rates for symptomatic PSVT termination vs placebo
[Source: Milestone Q3 2025 Earnings, Globe Newswire, Nov 12, 2025]
Q3 2025 Financial Results (Nov 12, 2025)
- EPS: ($0.12) vs ($0.17) consensus estimate (beat by $0.05)
- Cash Position: $82.6 million at September 30, 2025
- Financing: Successful equity offering completed in Q3; amended $75 million royalty purchase agreement with RTW (payment upon FDA approval)
- Burn Rate: FY2025 estimated EPS: ($0.62) per share
Commercial Launch Readiness
CEO Joseph Oliveto stated: “We are approaching our December 13 PDUFA date with optimism and excitement. Our team has recently increased pre-launch activities to better enable market readiness. We strengthened our balance sheet, and together with the $75 million royalty payment upon FDA approval, we believe we have resources in place to drive a successful commercial launch.”
Launch Strategy: Milestone has promotional launch plans set with “quick-start capability” upon potential FDA approval, indicating readiness to commercialize immediately if approved.
Analyst Ratings & Price Targets
| Firm | Rating | Price Target | Date |
|---|---|---|---|
| Consensus (3 Analysts) | Strong Buy | $4.50 | Nov 2025 |
| Price Target Range | – | $4.00 – $5.00 | Nov 2025 |
| H.C. Wainwright | Buy (Reiterated) | $5.00 | Sept 17, 2025 |
[Sources: TipRanks, Stock Analysis]
SEC Filings & Recent Activity
- Q3 2025 Earnings: November 12, 2025 – Financial results and regulatory update [Globe Newswire]
- Inducement Grants: November 3, 2025 – Granted 65,000 stock options at $1.85 exercise price under 2021 Inducement Plan (Nasdaq Rule 5635(c)(4)) [Stock Titan]
Market Opportunity
PSVT affects an estimated 2-3 per 1,000 individuals in the U.S. Current standard of care involves emergency room visits and IV adenosine administration. Etripamil nasal spray would be the first self-administered treatment option, potentially avoiding ER visits and empowering patients to manage episodes at home.
Peak Sales Potential: Analysts project peak sales potential exceeding $700 million if approved, based on addressable U.S. PSVT population and pricing assumptions.
⚠️ Key Risk: While the prior CRL was CMC-only (not efficacy/safety), this is still the second submission attempt. Any additional FDA concerns could result in another CRL or approval with restrictions. The company’s cash position ($82.6M + $75M upon approval) provides runway, but extended delay would impact viability.
? Comparative Summary: AGIO vs BCRX vs MIST
| Factor | AGIO (Dec 7) | BCRX (Dec 12) | MIST (Dec 13) |
|---|---|---|---|
| Event Type | sNDA (label expansion) | sNDA (pediatric expansion) | NDA (resubmission) |
| Approval Probability | Moderate (REMS requirement concern) | High (commercial drug + positive data) | Moderate-High (CMC-only CRL) |
| Revenue Status | Commercial (PYRUKYND for PKD) | Commercial ($590-600M FY25 guidance) | Pre-revenue |
| Market Cap (approx) | ~$450M | ~$1.5B | ~$200M |
| Analyst Consensus Target | $34 (Leerink) | $19.18 | $4.50 |
| Volatility Risk | High (binary PDUFA, small-cap) | Low-Moderate (existing revenue cushion) | High (pre-revenue, binary approval) |
| Recent Catalyst | ASH 2025 positive thalassemia data | ACAAI 2025 positive pediatric data | AHA 2025 consistent efficacy data |
Timeline Concentration Risk
All three PDUFA dates fall within a 7-day window (Dec 7-13, 2025). This creates potential for:
- Momentum scenarios: Early approvals (AGIO Dec 7) could create positive sentiment for later decisions
- Sector rotation risk: Multiple CRLs in short succession could dampen biotech small-cap sentiment
- Portfolio concentration: Traders holding multiple positions face compounded binary risk exposure
? Sources & References
Official SEC Filings
- Agios Pharmaceuticals (AGIO): SEC EDGAR – AGIO Filings
- BioCryst Pharmaceuticals (BCRX): SEC EDGAR – BCRX Filings
- Milestone Pharmaceuticals (MIST): SEC EDGAR – MIST Filings
Company Press Releases (Globe Newswire)
- Agios – PDUFA Extension Announcement (Sept 4, 2025)
- BioCryst – Q3 2025 Earnings (Nov 3, 2025)
- Milestone – Q3 2025 Earnings & Regulatory Update (Nov 12, 2025)
- Milestone – FDA CRL Response Acceptance (July 11, 2025)
- BioCryst – ACAAI 2025 Pediatric Data Presentation (Oct 23, 2025)
Financial Data & Analysis
- Investing.com – Leerink Upgrades AGIO (Nov 20, 2025)
- MarketBeat – BCRX Stock Analysis
- TipRanks – MIST Stock Forecast
- Simply Wall St – AGIO Analysis
- CNN Business – BCRX Financial Data
- Stock Analysis – MIST Overview
Medical & Scientific Sources
?️ Professional Trading Tools
We use these professional tools for biotech research, catalyst tracking, and market analysis. Links below include affiliate partnerships that support our work.
? Stock Screening & Financial Data
Finviz Elite – Advanced stock screeners, real-time market data, pre-market scanners for small-cap biotech. Earn: 30% recurring commission on subscriptions.
Seeking Alpha Premium – Comprehensive biotech coverage, earnings transcripts, analyst ratings. Essential for PDUFA tracking and catalyst monitoring. Earn: Unlimited commissions.
ChartsWatcher – Professional stock scanner with pattern recognition and real-time alerts. Earn: 30% commission on up to 6 billing intervals.
? Trading Platform
Medved Trader – Lightning-fast charting & trading platform designed by traders for traders. Real-time alerts, Level 2 data, direct broker integration for PDUFA event preparation.
? Support Merlintrader
If you find value in our catalyst reports and biotech analysis, consider supporting our work:
- PayPal Donations: Donate via PayPal
Your support helps us continue delivering free, ad-free, unbiased biotech research. ?
Transparency: Affiliate links support our research. We only recommend services we personally use and trust.
⚠️ Important Disclaimers
Educational Purpose Only: This report is provided for educational and informational purposes only. It does not constitute financial advice, investment recommendation, or an invitation to buy or sell securities.
No Guarantees: FDA decisions are binary regulatory events that can result in significant volatility. Each user is solely responsible for their own research and trading decisions.
Sources: All information compiled from publicly available sources including SEC filings, company press releases via Globe Newswire, analyst reports, and financial news outlets. While we strive for accuracy, errors or omissions may exist. Always verify information independently.
Regulatory Compliance: Not registered investment advice under SEC, CONSOB, or MiFID II regulations. For full disclaimers, visit merlintrader.com/disclaimer/
FDA Pillar + BCRX & AGIO Articles
PDUFA – December 2025 (Global Calendar)
Published: 2025-12-04
-
BCRX – BioCryst Pharmaceuticals (Main Report)
Published: 2025-11-21
-
AGIO – Agios Pharmaceuticals (Thalassemia Deep Dive)
Published: 2025-11-19
Scanner for active traders
Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


