DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
CAPR Capricor

Capricor Therapeutics [finance:Capricor Therapeutics, Inc.] is a clinical‑stage biotech focused on cell and exosome‑based therapeutics for rare diseases, with lead asset Deramiocel (CAP‑1002) targeting cardiomyopathy in Duchenne muscular dystrophy
PLRX Will PLRX have a merry Christmas?

"Will PLRX have a merry Christmas?
Very speculative play .
SMMT Summit Therapeutics

ummit Therapeutics Inc. (NASDAQ: SMMT) is a biopharmaceutical company focused on developing innovative oncology therapies. The primary pipeline is built around ivonescimab, a first-in-class bispecific antibody developed in partnership with Akeso Biopharma (China).
Anti-obesity drugs and weight-loss therapies
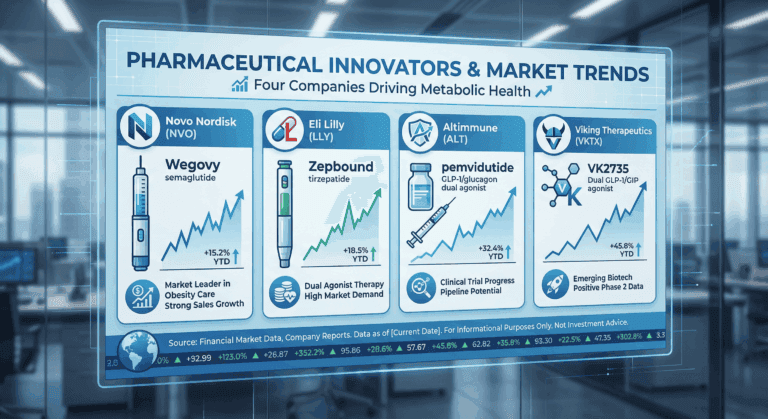
Scientific Comparison of Anti-Obesity Drug Efficacy 2025 – Complete Report Scientific Comparison of Anti-Obesity Drug Efficacy Semaglutide, Tirzepatide, Liraglutide, and Pemvidutide Complete Scientific Report – Updated: November 25, 2025 Table of Contents 1. Introduction 2. Mechanisms of Action and Pharmacological…
SYM Symbotic Inc
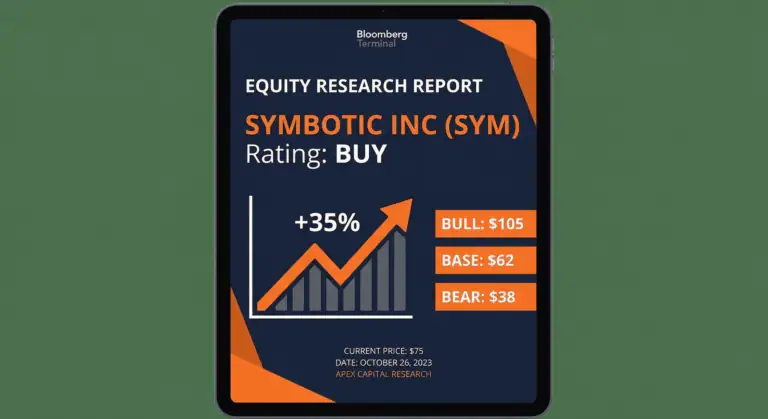
Symbotic surges +35% post-earnings; valuation tightens as growth investors price in upside
ADMA BIOLOGICS

ADMA Biologics is a rare biotech company that combines clinical success with financial profitability. Unlike pre-revenue biotech plays, ADMA operates a functioning immunoglobulin (plasma-derived therapy) business generating $426.5M in FY2024 revenue (+65% YoY) with actual net income of $197.7M. The company is well-managed, debt-reducing, and positioned in a high-growth market ($53.5B global plasma therapy market projected by 2033).
TNXP Tonix Pharmaceuticals
FDA APPROVED BUT HIGH DILUTION RISK
GME THE REDDIT REVOLUTION CONTINUES
GameStop [finance:GameStop Corp.] is not a typical stock—it is a cultural movement, a symbol of retail trader empowerment, and a battleground between Wall Street and Main Street. S
RIGL Rigel Pharmaceuticals Update Feb 2026
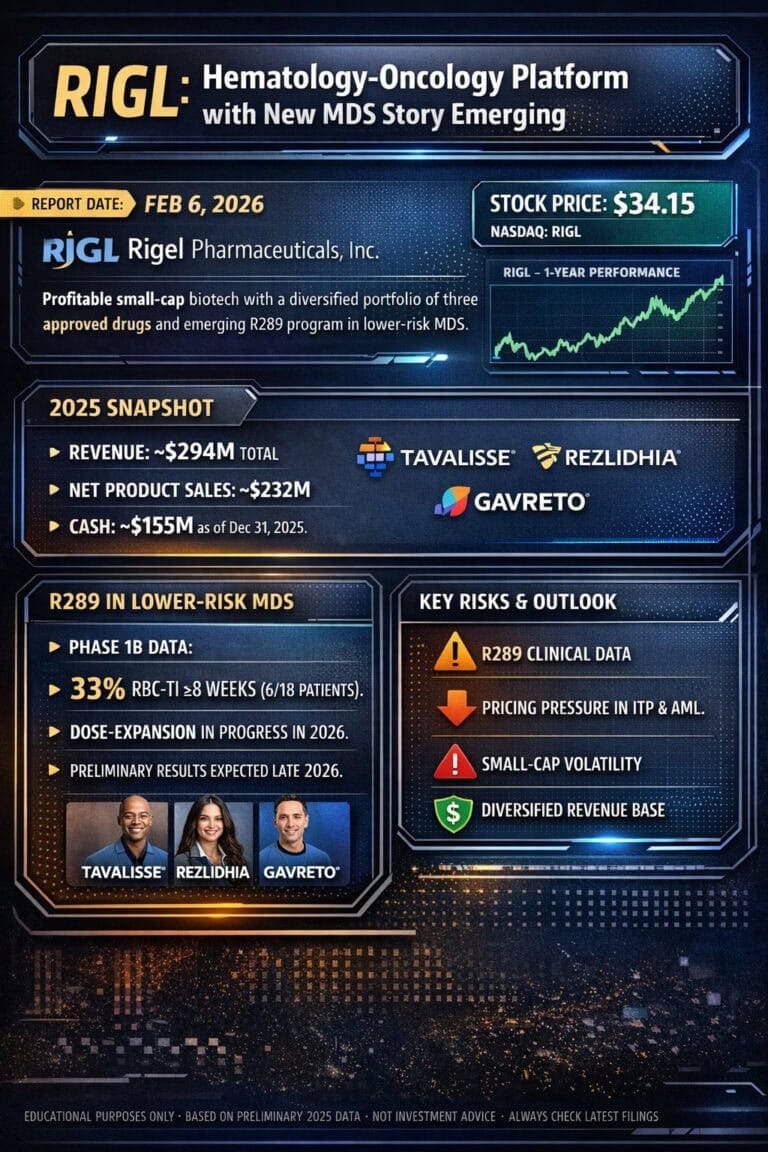
Rigel Pharmaceuticals [finance:Rigel Pharmaceuticals, Inc.] is a rare breed in biotech: profitable, cash-generative, and growth-oriented.
OSCR Oscar Health
oscr-aca-catalyst-report-nov24-2025.html OSCR Breaking News | Oscar Health | Merlintrader ?? ?? OSCR BREAKING CATALYST ACA SUBSIDY EXTENSION November 24, 2025 | Political Healthcare Policy Breakthrough OSCR CATALYST ACUTO ESTENSIONE SUSSIDI ACA 24 Novembre 2025 | Breakthrough Politica Healthcare Executive Summary…
ENLV Enlivex Therapeutics
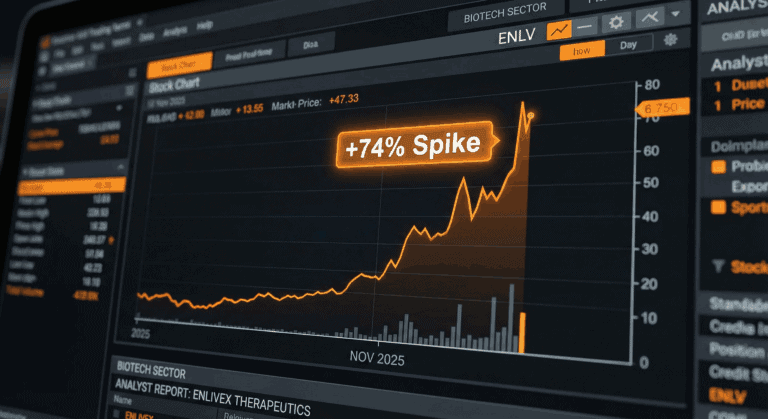
Enlivex Therapeutics [finance:Enlivex Therapeutics Ltd.] announced two simultaneous catalysts on November 23-24, 2025, triggering a 74% intraday spike to $1.59: (1) positive 6-month topline data from the Allocetra Phase IIa trial demonstrating durable pain reduction, and (2) a $212M PIPE financing at $1.00/share closing November 25. The clinical data supports Phase IIb progression, but the financing creates massive shareholder dilution (~99%) and introduces governance/crypto risks requiring careful assessment.
Weekly Market Analysis Nov 24

The major US stock indices closed the week of November 22 with significant losses, despite a Friday rebound.
ALT Altimmune, Inc.

Altimmune, Inc. (NASDAQ: ALT) is a late-stage clinical biotech company headquartered in Gaithersburg, Maryland, developing pemvidutide (ALT-801), a dual GLP-1/glucagon receptor agonist with a balanced 1:1 mechanism designed for metabolic diseases.
PLX Protalix Biotherapeutics

Protalix Biotherapeutics (NASDAQ: PLX) is an Israeli biotech company specializing in plant cell-expressed recombinant proteins using its proprietary ProCellEx platform.
OTLK Outlook Therapeutics, Inc.

Binary Event Analysis: Outlook Therapeutics (OLTK) faces a critical PDUFA decision on December 31, 2025, for ONS-5010 (LYTENAVA™), a bevacizumab biosimilar for wet age-related macular degeneration (wet AMD).
Palatin Technologies Inc.

Palatin Technologies (NYSE American: PTN) is a micro-cap biotech specializing in obesity treatment through melanocortin-4 receptor (MC4R) agonists.
BCRX BioCryst Pharmaceuticals nov 21
Positive Pediatric Catalyst:
FDA approval on December 12 could represent a doubling of TAM (Total Addressable Market) by adding the completely underserved pediatric segment (zero approved oral therapies).
RGNX REGENXBIO
Currently at channel support , technically a good entry point . REGENXBIO Inc. (RGNX) is facing a landmark FDA decision on February 8, 2026 regarding approval of RGX-121 (clemidsogene lanparvovec), a one-shot AAV gene therapy for MPS II (Hunter Syndrome).
Aldeyra Therapeutics dec 16 PDUFA

Aldeyra Therapeutics (ALDX) – Comprehensive PDUFA Report ?? ?? Aldeyra Therapeutics Ticker ALDX PDUFA Date December 16, 2025 Current Price ~$5.16 Report Date November 21, 2025 ? Executive Summary Aldeyra Therapeutics (ALDX) is approaching a pivotal catalyst: a binary FDA…
Top 10 Healthcare Small-Caps Stocks

Top 10 Healthcare Small-Caps Stocks Nov '25