DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Biotech Deep Dive – Pre-PDUFA Update
Omeros Corporation (OMER) – Updated 2025 Deep Dive
Narsoplimab’s Dec 26, 2025 FDA decision, the Novo Nordisk MASP-3 licensing deal, and a small-cap biotech trading at the center of a binary risk setup.
Binary setup: FDA PDUFA Dec 26, 2025 for TA-TMA
Company Snapshot & Key Numbers
Small-cap complement-pathway biotech
Quick view
Omeros Corporation is a US-based biotechnology company focused on complement-related diseases. Its lead asset, narsoplimab (OMS721), targets transplant-associated thrombotic microangiopathy (TA-TMA), with a PDUFA target action date of December 26, 2025 for its resubmitted BLA. This is the core binary catalyst currently driving the stock.
Shares have rebounded strongly from sub-$3 levels earlier in 2025 to trade near recent double-digit highs ahead of the FDA decision, amplified by a transformative licensing deal with Novo Nordisk for its MASP-3 inhibitor Zaltenibart (OMS906).
Key idea: OMER is effectively a high-volatility, catalyst-driven name where
the Dec 26 decision on narsoplimab can re-rate the entire equity, positively or negatively.
Key figures (latest available)
| Market Cap | ≈ $800M |
|---|---|
| Price (Dec 8, 2025) | $11.40 |
| 52-week performance | From sub-$3 lows to near $13+ |
| Short Float | ≈ 19.6% |
| Institutional Ownership | ≈ 46–50% |
| Q3 2025 Net Loss | $30.9M |
| Q3 Cash (9/30/25) | $36.1M |
| Upfront from Novo | $240M (Dec 1, 2025) |
| Total potential from deal | Up to $2.1B + royalties |
Market cap, ownership and short float based on Finviz/MarketBeat; cash and losses from Omeros Q3 2025 results; Novo deal terms from company releases and transaction press coverage.
Executive Summary – Why OMER Matters Now
High risk, high sensitivity to one FDA event
OMER has become one of the most closely watched small-cap biotech names into year-end 2025. The stock sits at the intersection of:
- A binary regulatory event – the FDA decision on narsoplimab for TA-TMA on Dec 26, 2025.
- A validation and financing deal – Novo Nordisk’s licensing of Zaltenibart (OMS906) for up to $2.1B, with $240M in cash already received.
- High short interest and mixed analyst views – consensus is “Moderate Buy”, with targets ranging from single digits to the mid-$40s.
The pivotal trial for narsoplimab in TA-TMA showed a hazard ratio of 0.32 (95% CI 0.23–0.44, p<0.00001), corresponding to an over three-fold reduction in mortality versus an external registry control. These data underpinned FDA’s Breakthrough Therapy designation and the BLA resubmission.
This report focuses on the updated picture as of early December 2025: the current
risk/reward profile, the Novo transaction and cash runway, the technical picture and
short interest, and how the bull and bear cases line up ahead of the PDUFA date.
It is an educational, non-promotional analysis based on verifiable,
primary sources.
Pipeline Overview – Complement Franchise and Beyond
MASP-2, MASP-3, and earlier programs
Narsoplimab (OMS721 – MASP-2, TA-TMA)
Narsoplimab is a fully human monoclonal antibody targeting MASP-2, a key enzyme in the lectin pathway of complement activation. In the pivotal TA-TMA study, narsoplimab-treated patients showed a statistically significant and clinically meaningful survival benefit, with a hazard ratio of 0.32 (95% CI 0.23–0.44, p<0.00001) versus registry controls. This translates into more than a three-fold reduction in mortality risk in a setting where historical mortality can reach 60–80 percent.
The BLA was resubmitted in March 2025 and accepted as a Class 2 resubmission. The PDUFA date, initially set for September 25, 2025, was extended to December 26, 2025 after Omeros responded to an FDA information request, with the agency noting that all requested analyses continue to support the drug’s benefit. An MAA has been submitted to the EMA, with a CHMP opinion expected around mid-2026.
If approved, narsoplimab (to be marketed as Yartemlia) would be the first approved therapy for TA-TMA, giving Omeros a potential monopoly in a rare, life-threatening indication.
Zaltenibart (OMS906 – MASP-3, Novo Nordisk)
Zaltenibart is a MASP-3 antibody in development for rare hematologic and renal disorders. In October 2025, Omeros announced an exclusive global license with Novo Nordisk, worth up to $2.1B in potential payments. The deal includes $240M upfront in cash (received at closing on December 1, 2025) and up to $100M in near-term milestones, plus further development and commercial milestones and tiered royalties on net sales.
Novo now leads development and commercialization, while Omeros retains certain rights to small-molecule MASP-3 programs. The transaction both validates the complement platform and materially extends the cash runway.
Other pipeline components
- OMS1029 (long-acting MASP-2) – Subcutaneous, long-acting MASP-2 inhibitor in Phase 1. Management has highlighted this as a follow-on opportunity building on any success with narsoplimab.
- OMS527 (PDE7 inhibitor, addiction) – In March 2025 the company disclosed a commitment from the US NIDA to fund early clinical work in cocaine use disorder, providing non-dilutive support.
- Oncology / GPCR / other complement programs – A broader set of preclinical efforts (including T-CAT oncology and antimicrobial complement approaches) that are longer-dated but could become more visible if the balance sheet and core franchise are secured.
Efficacy statistics for narsoplimab are taken from Omeros’ pivotal TA-TMA analyses and subsequent updates; deal terms for Zaltenibart are based on Omeros and Novo Nordisk press releases and transaction summaries.
Recent Developments, Cash Burn and Runway
Q3 2025 results and Novo cash impact
Q3 2025 snapshot
- Net loss: $30.9M (Q3 2025), compared with $32.2M a year earlier.
- Nine-month 2025 net loss: $89.8M.
- Cash and equivalents (9/30/25): $36.1M.
- Approximate cash burn: around low-$20M per quarter (before financing inflows).
To bridge to catalysts, Omeros raised capital during 2025 through a premium-priced direct offering and an at-the-market program. In parallel, the company restructured a portion of its convertible notes, extending maturities to 2029 in exchange for a higher coupon and conversion premium, which reduces near-term refinancing pressure.
Novo cash and debt pay-down
The closing of the Zaltenibart deal on December 1, 2025 is a major balance-sheet event. Omeros receives $240M upfront in cash, plus eligibility for up to $100M of near-term milestones and much larger longer-term milestones and royalties.
Management has indicated that the upfront will be used to:
- Repay approximately $67M of secured term debt.
- Retire around $17M of remaining 2026 convertible notes.
- Fund at least 12+ months of operations post-closing, including commercial preparations for a potential US launch of narsoplimab/Yartemlia.
The Novo proceeds do not eliminate all future financing risk, but they reduce
near-term default risk and allow Omeros to approach the PDUFA date from a
significantly stronger cash position compared with mid-2025.
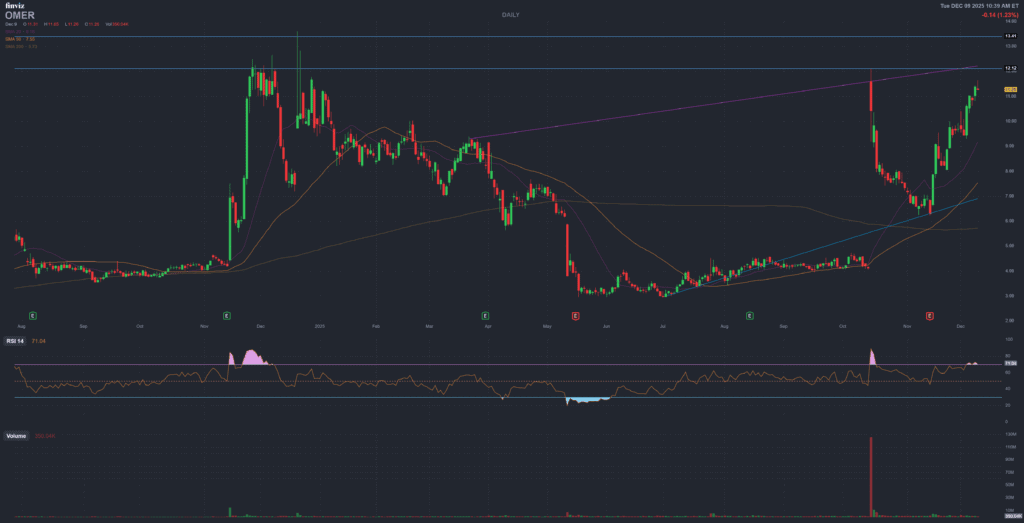
Technical Picture & Short Interest
Momentum vs. positioning
Price trend and momentum
OMER’s chart reflects the binary narrative. After trading in the low single digits for much of early 2025, the stock broke out sharply following the announcement and later closure of the Novo Nordisk deal. It has:
- Broken through prior resistance in the $7–8 range.
- Moved significantly above its 50-day and 200-day moving averages, which are still clustered in the mid-single-digit area.
- Reached a recent 52-week high in the low-teens before consolidating around $11–12 ahead of the PDUFA date.
Standard momentum indicators (RSI, MACD) place the stock near or slightly above typical “overbought” thresholds, consistent with a crowded, catalyst-driven move.
Short interest and positioning
- Short float: roughly 19–20 percent of free float, with over 13M shares sold short as of the latest settlement date.
- Institutional ownership: around 46–50 percent, with several funds holding mid-single-digit percentage stakes.
- Insider ownership: low-single-digit percentage of shares outstanding.
High short interest into a binary event cuts both ways. A positive FDA outcome could force covering and potentially amplify upside moves, while a negative decision would likely see shorts press their position into weakness.
Technically, OMER is in a strong uptrend with stretched momentum and elevated
short interest – a setup that can produce very large moves in either direction
around news.
Analyst Views & Valuation Context
Moderate Buy with very wide targets
Street snapshot (MarketBeat, TipRanks and others)
- Consensus rating: “Moderate Buy”, with a mix of Strong Buy, Buy, Hold and at least one Sell rating.
- Average price target (MarketBeat): around the mid-$30s (≈ $33–34), implying material upside versus the current share price.
- Average price target (TipRanks): lower double digits, closer to current levels, based on a smaller analyst set.
- Notable calls: HC Wainwright lifting its target from $9 to $20 (Buy), WBB Securities reiterating a Strong Buy with a $45 target, and more cautious firms placing single-digit targets or Sell ratings.
The spread between the lowest and highest published targets (roughly $9 to $45) underscores how dependent the valuation is on:
- Whether narsoplimab is approved on the current timelines.
- How quickly TA-TMA adoption ramps and what real-world pricing looks like.
- How investors discount the broader complement pipeline and Novo partnership.
The Street as a whole is optimistic but far from unanimous. Analyst targets should be
viewed as scenario markers rather than guarantees, especially with a single regulatory
decision driving most of the near-term value.
Management, Governance & Capital History
Founder-led with an active use of capital markets
CEO profile – Gregory A. Demopulos, M.D.
Omeros is led by Dr. Gregory Demopulos, who founded the company in 1994 and has served as president, CEO and chairman ever since. Prior to founding Omeros, he trained as an orthopedic surgeon at Stanford University and completed fellowship training in hand and microvascular surgery at Duke University. He has also previously served in interim roles as chief financial officer and chief medical officer.
This long tenure creates continuity and deep familiarity with the science, but also concentrates strategic control in a single individual. Investors often look closely at long-term capital allocation decisions when assessing founder-led biotech companies.
Dilution, debt and shareholder treatment
- Omeros has historically relied on a combination of equity offerings and convertible debt to fund operations, including a 2025 premium-priced direct offering and regular at-the-market issuance.
- The company completed a reverse stock split in its earlier years and, more recently, restructured a portion of its 2026 convertible notes into higher-coupon 2029 notes, trading near-term dilution risk for extra time.
- The Novo Nordisk transaction is a rare example where a large amount of non-dilutive cash replaces what might otherwise have required further equity issuance at lower prices.
For readers, it is important to review historical SEC filings, note offerings and stock-based compensation trends to understand the cumulative impact on per-share ownership over time.
Bull vs Bear Case – Scenario Framework
What could go right, and what could go wrong
High-level framework (not a forecast, not advice)
Bull case (upside scenario)
- FDA approves narsoplimab on Dec 26, 2025 (or shortly after), confirming the survival benefit seen in the pivotal trial and recognizing the unmet need in TA-TMA.
- Omeros executes a focused launch, supported by established diagnosis/reimbursement codes and potential Medicare NTAP support, allowing relatively rapid adoption in transplant centers.
- Real-world data reinforce durability of effect and acceptable safety, enabling peak sales toward the higher end of current estimates for a rare disease asset.
- The Novo partnership progresses with positive data from MASP-3 programs, and at least some early milestones are reached, adding non-dilutive cash and validating the platform further.
- Equity markets reward the combined franchise with a valuation more in line with other commercial-stage rare-disease companies, re-rating OMER above current levels and toward bullish analyst targets.
Bear case (downside scenario)
- FDA does not approve narsoplimab on the current resubmission, issues a Complete Response Letter, or requests additional data and analyses that significantly delay any potential market entry.
- Even if approval is ultimately granted, real-world adoption proves slower than modeled, due to payer scrutiny, logistical challenges in TA-TMA diagnosis, or competition from other complement-targeting strategies.
- Cash burn remains elevated versus incoming non-dilutive cash, forcing additional equity issuance at weaker share prices, further diluting existing shareholders.
- The Novo collaboration progresses more slowly than expected, with key milestones pushed to the right, making the effective value of the deal lower than headline potential payouts suggest.
- Market sentiment turns against high-risk small-cap biotech broadly, compressing multiples even for assets with apparently solid data.
The ranges often quoted in commentary (for example, downside scenarios in the low-single-digit share price versus upside scenarios above $20) come from Street target spreads and historical trading reactions to comparable binary events, not from this report as a recommendation.
Key Catalyst Timeline (Next 12–18 Months)
Regulatory and clinical milestones to watch
December 26, 2025
PDUFA – US
FDA target action date for narsoplimab in TA-TMA. This is the pivotal binary event.Late 2025 – early 2026
Debt reduction
Application of Novo upfront proceeds to repay term debt and remaining 2026
convertible notes, leaving a cleaner balance sheet.Mid 2026 (indicative)
EMA / CHMP – EU
Expected EMA/CHMP opinion on narsoplimab MAA for TA-TMA. A positive outcome would
open the European market.2026+
Novo MASP-3 milestones
Potential clinical updates and milestone events from the Zaltenibart (OMS906) program
under Novo Nordisk’s leadership.Exact timings are based on Omeros guidance and regulatory process norms; all dates and expectations remain subject to change by regulators and the company.
Sentiment – Analysts, Funds and Retail
Binary narratives on both sides
On the professional side, OMER is widely treated as an event-driven name. Funds and analysts focused on rare-disease and complement biology tend to focus on the strength of the survival data, the Breakthrough designation and the Novo deal. Skeptics emphasize the prior Complete Response Letter, small target market size and the company’s historical capital raises.
Retail sentiment on social platforms (Stocktwits, Reddit, X) is similarly polarized. Bullish commentary usually highlights:
- The apparent scale of the survival benefit and the lack of approved alternatives in TA-TMA.
- The credibility boost from Novo Nordisk’s decision to commit billions in potential payments to the MASP-3 program.
- The possibility of a short squeeze given nearly 20% of the float sold short.
Bearish or cautious voices tend to point out:
- The prior BLA rejection and the risk that FDA may still not be satisfied with external control comparisons.
- Ongoing losses and the need for careful cash management even after the Novo cash is deployed.
- The fact that “first-in-class” does not always guarantee rapid adoption, especially in complex transplant settings.
Sentiment observations here combine analyst notes, fund commentary and typical themes visible in public discussions; they are descriptive only and should not be interpreted as recommendations.
Useful Tools & Partners for Independent Research
Always cross-check with primary sources
- Finviz – OMER technicals, short interest and ownership .
- ChartsWatcher – real-time US market scanner for intraday setups . ChartsWatcher is a real-time, next-generation scanner for the US stock market.
- Seeking Alpha – earnings call transcripts, deep-dive articles and quant data .
- Stocktwits – real-time stream of trader sentiment on OMER .
- Medved Trader – advanced desktop trading platform for detailed charting, level 2 and tape reading around catalyst dates (no referral link).
- Merlintrader trading Blog – hub for biotech catalyst reports, timelines and educational content .
Some of the links above are affiliate or referral links for the site. If you choose to open an account or subscribe through them, a small commission may be paid to support the Merlintrader project at no extra cost to you. See the full legal disclaimer for details.
Support Merlintrader trading Blog
If this kind of long-form research is useful to you and you would like to help keep the site and reports free for everyone, you can support the project with a one-time PayPal donation.
 Donate to keep content free
Donate to keep content free
Important Disclaimer
Educational content – not investment advice
This report is provided strictly for informational and educational purposes only. It is not and must not be interpreted as an offer to buy or sell any financial instrument, nor as individualized investment advice, portfolio management, legal advice or tax advice. All opinions expressed here reflect the author’s personal interpretation of publicly available information at the time of writing and may change without notice.
The analysis relies on sources believed to be reliable (including official company filings, press releases, conference call transcripts, and recognized financial data providers), but no guarantee is given as to their completeness or accuracy. Numbers may contain errors, may be revised by companies and regulators, and may become outdated quickly, especially around binary regulatory events.
Trading in biotech stocks, particularly around FDA decisions and other binary catalysts, is inherently risky and can lead to rapid and substantial losses of capital. Anyone considering an investment or trade should carry out their own independent research, verify all key figures from primary sources (for example, SEC filings and official company communications), and consult with a qualified financial professional where appropriate. Past performance and analyst price targets are not indicative of future results.
For full legal information and disclosures for Merlintrader, please refer to:
Author Note & Methodology
Authors: Merlintrader and Jane
Key Source Links (for independent verification)
Always go back to the primary documents
- Omeros Corporation – news releases and financial results (including Q3 2025 and Novo deal closing).
- Omeros historical price lookup – official daily closing prices for OMER.
- Omeros Q3 2025 results press release and earnings call transcript.
- Omeros and Novo Nordisk Zaltenibart (OMS906) licensing deal announcements and related coverage (upfront, milestones, and royalty structure).
- Narsoplimab TA-TMA pivotal trial publications and Omeros updates on survival analyses and PDUFA date extensions.
- MarketBeat and TipRanks – analyst ratings, price targets and consensus views for OMER.
- Finviz – OMER overview, technicals, short interest and ownership breakdown.
This list is not exhaustive; readers are encouraged to review SEC filings, clinical trial registries and peer-reviewed journals for the most detailed information.
FDA Pillar + OMER Articles
PDUFA – December 2025
Published: 2025-12-04
-
OMER – Path of the Force
Published: 2025-12-09
-
OMER – The Biotech Force Awakens (Analysis)
Published: 2025-11-19
-
OMER – A Sleeping Giant
Published: 2025-11-11
-
OMER – Update #2
Published: 2025-11-04
-
OMER – Early Article
Publication date not available
Scanner for active traders
Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


