DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
ImmunityBio Inc ($IBRX) ANKTIVA in EU Ok!

European Commission Grants Final Conditional Marketing Authorization for ANKTIVA® in EU
Decision C(2026)1197: Final, binding authorization allowing commercial launch across 27 EU member states + Iceland, Norway, Liechtenstein for BCG-unresponsive non-muscle invasive bladder cancer (NMIBC).
ImmunityBio (IBRX) — Saudi SFDA engagement

Two items in one tape-read: (1) a fresh BusinessWire update on Saudi regulatory engagement (rBCG dossier + ANKTIVA/CPI expansion discussions), and (2) a sell-side note in circulation reiterating a $24 price target from D. Boral Capital (note dated Jan 13, 2026 in the sources below).
Compass Pathways Plc ADR ($CMPS) Updated post CC

Release of new clinical data from two Phase 3 trials (COMP005 and COMP006) on COMP360 in TRD + management webcast
Tradable short-term event: additional clinical data (specific portions of studies) and official management narrative. This is the type of event that changes the perception of probability/quality of clinical evidence, and thus volatility too.
AI in Biotech 2026 $RXRX $TEM $EXAI

Recursion, Tempus AI and Exscientia are three “pure-play” examples of what it really means to put artificial intelligence at the centre of a biotech business model. Used properly, AI can compress timelines, surface non-obvious biology and make better use of messy data. Used badly, it can amplify errors, burn cash faster and create stories the clinic will never confirm.
Precision Biosciences Inc ($DTIL)

Next catalyst
H1 2026 – Updated ELIMINATE-B Phase 1/2 data in chronic hepatitis B (PBGENE-HBV), including higher-dose biopsy readouts.
Secondary 2026 catalyst: first sites activated and initial multi-patient data from the FUNCTION-DMD Phase 1/2 trial of PBGENE-DMD after FDA IND clearance (Study May Proceed) in February 2026.
Aldeyra Therapeutics (ALDX) PDUFA March 2026

Aldeyra Therapeutics (NASDAQ: ALDX) is a clinical-stage biotechnology company developing modulators of reactive aldehyde species (RASP) for immune-mediated and metabolic diseases. The key late-stage asset is reproxalap, an ophthalmic RASP modulator for dry eye disease (DED) and allergic conjunctiviti
2026 Decisive year for three cult biotech stocks $IOVA $IBRX $ALT

A parallel “year of truth” roadmap for Iovance Biotherapeutics (IOVA), ImmunityBio (IBRX) and Altimmune (ALT), three names with huge retail followings, real science on the table and very little room for execution mistakes.
Weekly Market Briefing (February 16–20, 2026)

Wall Street just closed a choppy week where enthusiasm about AI and data-center spend ran into valuation nerves, while inflation data kept the “soft landing” narrative alive. The coming days will be dominated by U.S. GDP, the Fed’s preferred inflation gauge (core PCE), fresh PMIs and a cluster of sector-specific catalysts across biotech, space, defense and AI small/mid caps.
Outlook Therapeutics Inc ($OTLK)

A post-CRL, post-Type A meeting request deep dive on ONS-5010/LYTENAVA, EU/UK launch reality, funding pressure and how much option value is left in a sub-$35M market cap.
Top 10 Biotech Stocks to Watch in H1 2026 — PDUFA Dates & Catalysts

Big Pharma is heading into a new patent cliff while its in-house pipelines struggle to keep up. That makes small and mid-cap biotech one of the most fertile hunting grounds for both acquisitions and high-beta “run-up” trades ahead of binary events like FDA decisions and pivotal clinical readouts. In this note we highlight ten US-listed biotech names under roughly $2 billion in market cap that all have a clearly defined catalyst between now and the end of June 2026.
Neumora Therapeutics Inc ($NMRA) Feb 12 2026

Key idea. In January we framed Neumora as a post-crash special situation: a neuroscience platform that had just blown up its lead asset navacaprant in Phase 3 MDD, wiping out about 80% of the market cap in one session, but still sitting on a sizeable cash pile and a broad pipeline. The weeks since then have added three crucial pieces: (1) full Phase 1b signal-seeking data for NMRA-511 in Alzheimer’s agitation, (2) a more detailed 2026 pipeline strategy, and (3) better visibility on ownership, institutional support and analyst appetite post-crash. This second deep dive doesn’t repeat the full story — it extends it and tries to answer a simple question: is NMRA slowly rebuilding a credible equity story, or just trading on residual hope and short-term speculation?
Viking Therapeutics Inc (VKTX)
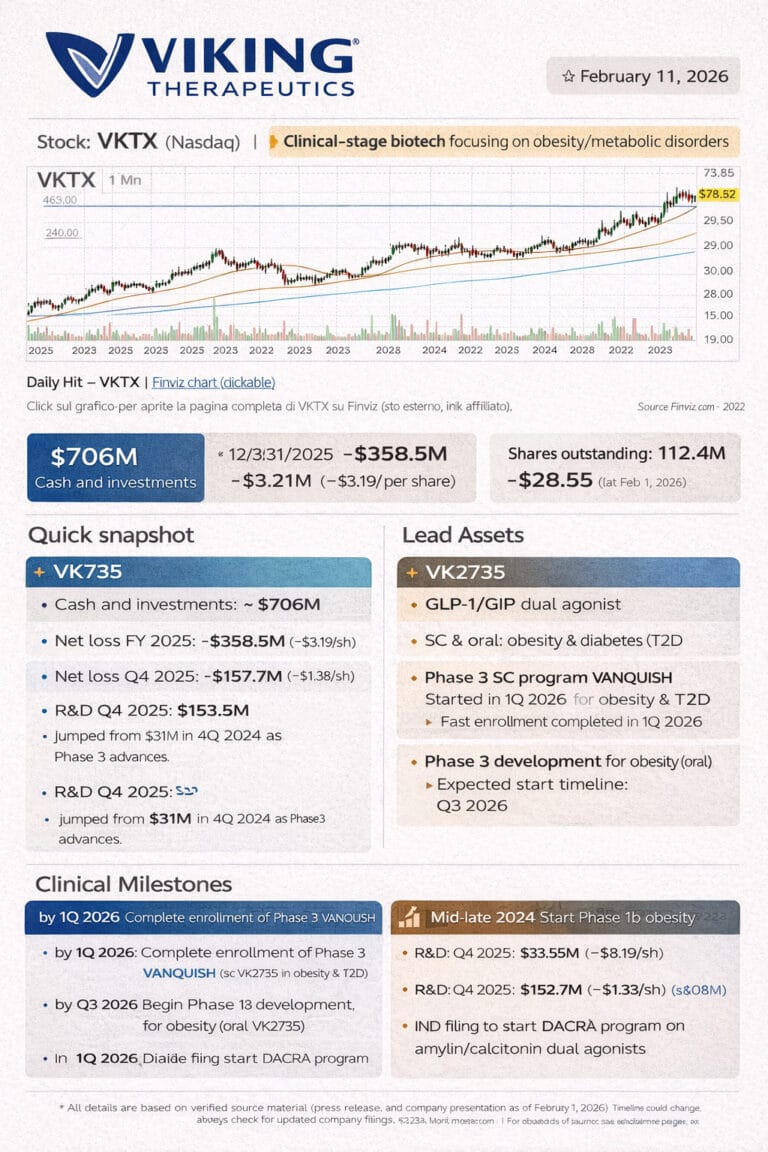
Viking has just reported fourth-quarter and full-year 2025 numbers and given a dense update on its obesity and metabolic pipeline. The picture is clear: spending is ramping, the cash pile is still very strong, and the company is steering both injectable and oral VK2735 toward the next wave of pivotal trials while preparing a new amylin program and keeping VK2809/VK0214 alive in the background.
Moderna Inc ($MRNA) Flu Shot? No Shot

Why this Daily Hit, if Moderna is not a small biotech?
Moderna is obviously not a small-cap biotech living hand-to-mouth. Yet the refusal-to-file (RTF) letter that the U.S. Food and Drug Administration (FDA) just sent for its seasonal flu vaccine mRNA-1010 is such an outlier in terms of process and signalling that it deserves a dedicated “hit” article.
Denali Therapeutics Inc ($DNLI)

Denali Therapeutics is moving into the most delicate phase of its evolution: from a blood–brain-barrier platform story to a commercial-stage rare-disease company. The centre of gravity is tividenofusp alfa (DNL310), a TransportVehicle-enabled enzyme replacement therapy for Hunter syndrome (MPS II) designed to address both systemic and central nervous system manifestations, something that current standard ERTs cannot do.
TuHURA Biosciences ($HURA)

TuHURA announces that Kintara’s REM-001 trial in metastatic cutaneous breast cancer has met its primary safety endpoint in ten patients with early signs of clinical efficacy. This triggers the contractual milestone under the Kintara CVR Agreement and unlocks the distribution of 1,539,958 HURA shares to legacy Kintara shareholders holding CVRs.
uniQure N.V ($QURE)

Updated preliminary Phase I/IIa data in Fabry disease show sustained, dose-dependent α-Gal A activity far above normal and allow enzyme-replacement withdrawal in more than half of treated patients, while safety findings trigger a pause in higher-dose cohorts. At the same time, AMT-130 for Huntington’s disease faces a tougher regulatory path after FDA feedback that current Phase I/II data are unlikely to support a BLA as primary evidence. This report connects Fabry, Huntington and the broader AAV5 platform story under the lens of balance sheet, catalyst map, key risks and retail sentiment.
Maze Therapeutics Inc ($MAZE)

Maze trades as a high-expectations play on APOL1-mediated kidney disease: the stock has rerated into a roughly mid-cap valuation on the back of a very strong balance sheet and the upcoming proof-of-concept data from the HORIZON Phase 2 trial in a broad AMKD population. The key tension now is simple: can the first substantive MZE829 dataset justify how far the share price has already come.
REGENXBIO (RGNX)

The FDA has sent a Complete Response Letter (CRL) for RGX-121 (clemidsogene lanparvovec) in MPS II, rejecting the BLA under the accelerated approval pathway and questioning the external control, the neuronopathic eligibility criteria and the CSF HS D2S6 biomarker. Management plans a Type A meeting and aims to resubmit, but the ultra-rare MPS II path is now longer and more complex, against a backdrop of recent clinical holds on the company’s ultra-rare MPS programs.
Quince Therapeutics Inc ($QNCX)

Quince Therapeutics has announced that it has engaged LifeSci Capital as its exclusive financial advisor to assist with a restructuring process and a formal review of “strategic alternatives” with the stated goal of maximizing shareholder value. The menu of options explicitly listed by the company includes partnerships, joint ventures, mergers, acquisitions, licensing deals or other strategic transactions, and LifeSci will also support any restructuring of the company’s liabilities.
Ensysce Biosciences Inc ($ENSC)

Ensysce Biosciences has provided a new operational update on its pivotal Phase 3 trial PF614-301, which evaluates PF614, a next-generation oxycodone prodrug, in patients with moderate to severe acute pain following abdominoplasty surgery. The company reports that the study has now enrolled 50% of the subjects targeted for the planned interim review, a meaningful operational milestone for a small clinical-stage pain company trying to move an abuse- and overdose-resistant opioid toward the market.