DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
Alpha Tau Medical Ltd ($DRTS)

Alpha Tau Medical is trying to build an entire platform around one idea: using very short-range, high-energy alpha particles to burn tumors from inside the lesion, instead of bathing a large region of the body in external radiation. With the Japanese Ministry of Health, Labour and Welfare (MHLW) now granting full Shonin marketing approval for Alpha DaRT in unresectable, locally advanced or locally recurrent head and neck cancer, the company has just obtained its first real proof that regulators outside Israel are willing to treat this as a commercial therapy, not only an experimental one.
Edesa Biotech ($EDSA)

Edesa Biotech (NASDAQ: EDSA) is a clinical-stage biopharmaceutical company developing host-directed therapeutics (HDTs) – drugs that modulate the body's own immune response rather than targeting a specific pathogen. The company's lead asset, paridiprubart (EB05), is a first-in-class anti-TLR4 monoclonal antibody being evaluated as a treatment for Acute Respiratory Distress Syndrome (ARDS).
Iovance Biotherapeutics Inc ($IOVA)

As of February 24, 2026, Iovance Biotherapeutics is a small-cap oncology company (~$1.5B market cap) focused on TIL cell therapies for solid tumors, with AMTAGVI (lifileucel) already approved in the U.S. for advanced melanoma and a growing pipeline exploring TIL in other indications, including soft-tissue sarcomas.
The stock trades around $3.80 with roughly 364M shares outstanding, after a volatile path that has seen heavy dilution in past years but now combines commercial revenues (~$264M FY25) with a cash runway into Q3 2027 thanks to cost controls and better gross margins on AMTAGVI. The new 50% objective response rate (ORR) in the very difficult setting of advanced UPS/DDLPS sarcomas is what the market is reacting to right now.
Fortress Biotech Inc ($FBIO)

Fortress Biotech (NASDAQ: FBIO) is a diversified biopharmaceutical company with a unique portfolio acquisition and value-creation model. Rather than pursuing a single blockbuster drug, Fortress systematically acquires, advances and partners assets across oncology, dermatology and rare diseases, generating returns through product revenue, equity holdings, royalties and strategic asset sales.
SELLAS Life Sciences Group Inc ($SLS) Updated feb 23

SELLAS Life Sciences Group (NASDAQ: SLS) is a clinical‑stage biotechnology company focused on cancer immunotherapies and targeted therapies. The current story is driven by the cancer vaccine galinpepimut‑S (GPS), a Wilms Tumor 1 (WT1)–targeting immunotherapy, and by a CDK9 inhibitor program for hematologic and solid tumors.
ImmunityBio Inc Logbook Feb 23 / It Could Not Be Missing

The latest BusinessWire release finally puts hard numbers and structure around what we had already been tracking on IBRX: ~113 million USD of ANKTIVA net product revenue in 2025 (~700% year-over-year), ~750% unit growth, a regulatory footprint in 33 countries across four major jurisdictions, the first lung cancer approval in Saudi Arabia in combination with checkpoint inhibitors, and explicit patent protection for ANKTIVA combinations beyond 2035. On top of that, management now frames a clear three-year strategy with ANKTIVA as a “backbone” immunotherapy across multiple tumor types.
ADMA Biologics ($ADMA) meeting or exceeding guidance Feb25?

ADMA Biologics (NASDAQ: ADMA) is a commercial-stage biopharmaceutical company specializing in plasma-derived immunoglobulins for the treatment of patients with primary humoral immunodeficiency (PI) and others at risk of certain infections. The company operates a fully integrated business model encompassing plasma collection, fractionation, manufacturing and commercialization.
Ardelyx Inc (ARDX) Growth is real, panic is optional: what Q4 2025 earnings really say

The market reaction to Ardelyx’s Q4 2025 earnings was simple and brutal: the stock lost roughly fifteen percent in the days after the print, sending a message that “something is wrong”. If you stop at the chart, it looks like a broken story. If you read the filings, the picture is very different: this is a company that is clearly growing, but still paying a high price in operating expenses to sustain that growth.
Weekly Recap & Briefing – Biotech, Space, Defense & AI Feb 21 2026

This week’s recap walks through a classic “binary” market: indices inch higher while gold hits new records and oil creeps up on tariff and Iran risk. In biotech we see clean wins (VNDA, Telix) versus brutal setbacks (GRAIL, RGNX), while defense and space keep riding record military spending and drone/missile contracts. The briefing for February 23–28 closes with what really matters next: Nvidia’s earnings, the U.S. PPI print, and the first concrete signs from the Pentagon’s USD 1.1bn drone program.
Vanda Pharmaceuticals Inc ($VNDA)

The FDA has approved BYSANTI™ (milsaperidone) tablets as a first-line atypical antipsychotic for the acute treatment of manic or mixed episodes in bipolar I disorder and for the treatment of schizophrenia in adults. This is Vanda’s second FDA approval in under two months, following NEREUS™ for motion-induced vomiting prevention.
Oscar Health Inc ($OSCR) Update 20 Feb

Tech-enabled health insurer focused on Affordable Care Act individual and small-group markets, trying to recover from a difficult 2025 with a 2026 guidance that points back toward operating profitability, under the shadow of U.S. healthcare policy and subsidy decisions.
ImmunityBio Inc ($IBRX) Update Feb 20

ImmunityBio announced a commercial partnership with Biopharma and Cigalah Healthcare to launch ANKTIVA in Saudi Arabia and, over time, across the wider Middle East and North Africa, backed by an SFDA registration certificate with pricing. It’s not a new label approval, but it is a concrete step in the global monetization of the ANKTIVA franchise.
Tandem Diabetes Care Inc ($TNDM)

After crossing 1 billion dollars in annual revenue and reporting record gross margins, Tandem enters 2026 in a delicate transition phase: rolling out a pay-as-you-go pharmacy model, expanding direct international operations and pushing its Control-IQ+ automation to more patients, while still carrying net losses and a sizeable convertible note stack.
Solid Biosciences ($SLDB)

Solid Biosciences Inc. (NASDAQ: SLDB) is a precision genetic medicines company developing AAV‑based gene therapies for rare neuromuscular and cardiac diseases, with a lead focus on Duchenne muscular dystrophy (DMD), Friedreich’s ataxia (FA), and catecholaminergic polymorphic ventricular tachycardia (CPVT)
Corcept Therapeutics Inc ($CORT) Feb 19 Update
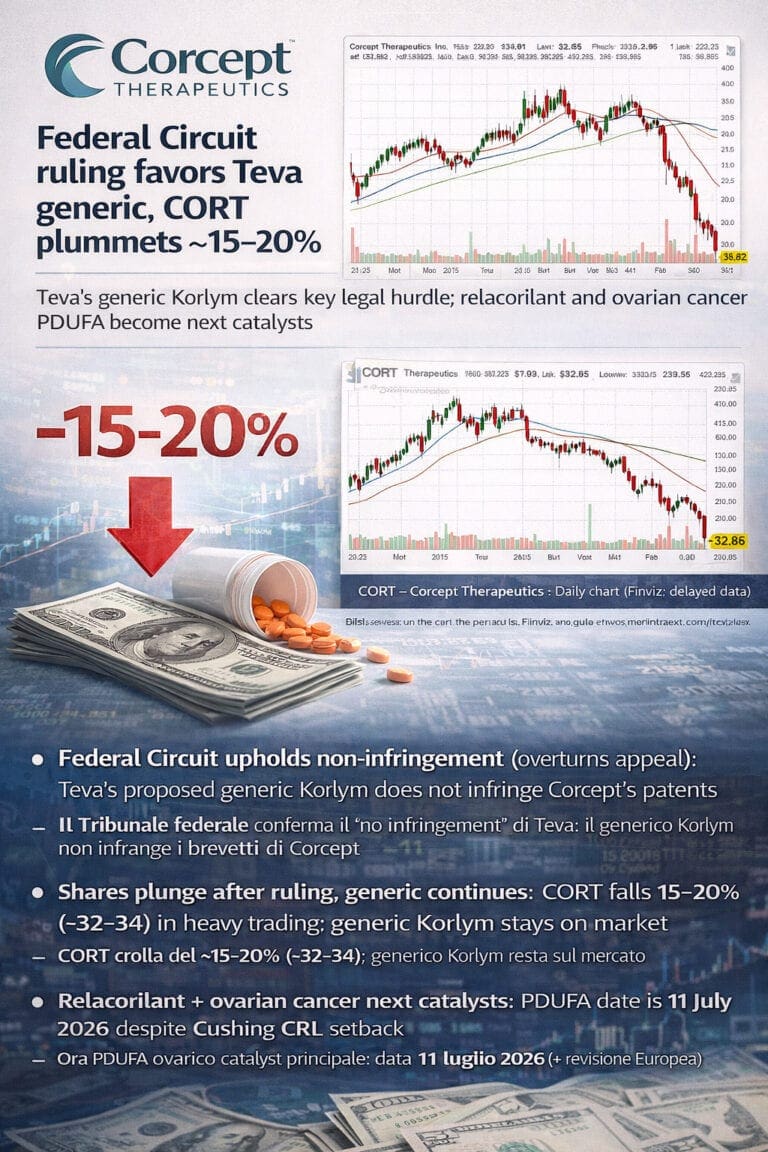
Quick EN/IT follow-up after the U.S. Court of Appeals for the Federal Circuit upheld the New Jersey decision that Teva’s proposed generic Korlym does not infringe two Corcept patents, clearing a key legal hurdle for generic competition. We look at what this means for Korlym cash flows, how it interacts with the recent relacorilant CRL and why the ovarian cancer PDUFA remains the central medium-term catalyst.
Hims & Hers Health Inc ($HIMS)

A deep dive into Hims & Hers after the GLP-1 “pill gambit”, the Novo Nordisk lawsuit, and the transformative acquisition of Eucalyptus: business model, financials, balance sheet, insiders, retail crowd and key catalysts for 2026–2027
Grace Therapeutics (GRCE) April 23 Fda Decision

NASDAQ: GRCE · Biotech · aSAH / nimodipine IV Grace Therapeutics (GRCE) – GTx-104 and the 2026 FDA Decision Deep dive on Grace Therapeutics ahead of the April 23, 2026 PDUFA date for GTx-104, the company’s IV nimodipine for aneurysmal…
Geron Corp ($GERN) Feb 18 Update
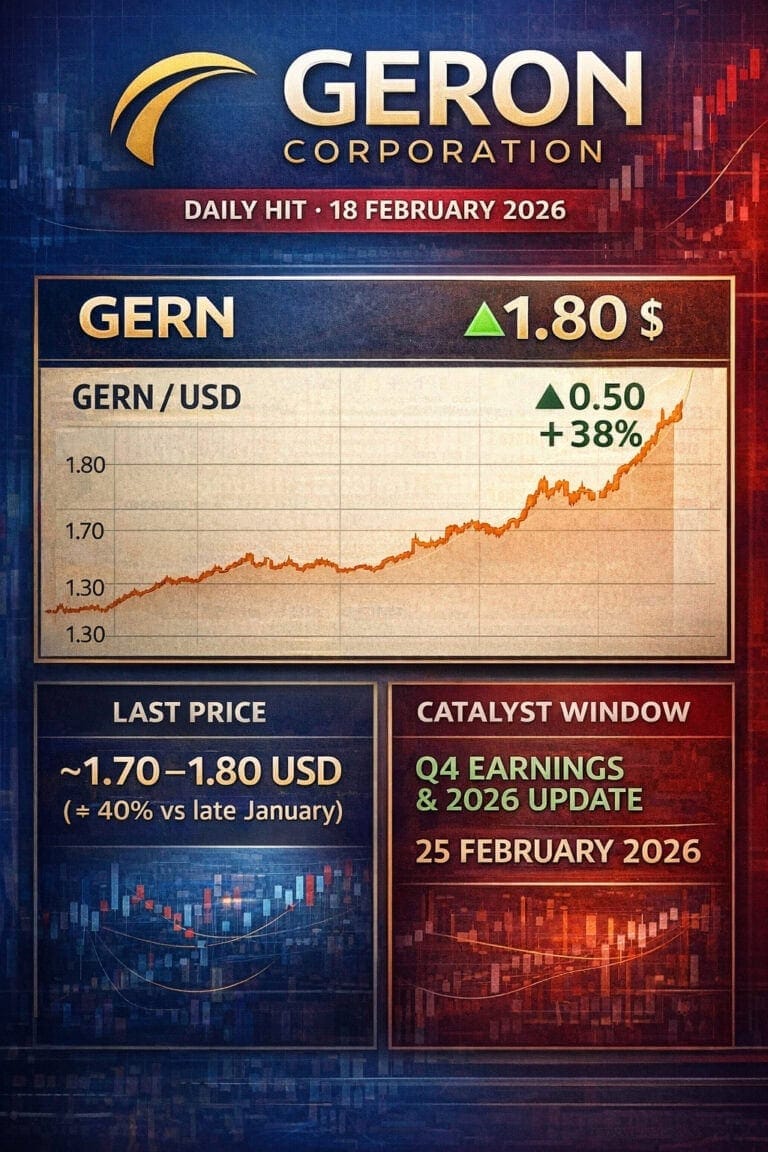
Almost one month after our long-form report at around $1.30, Geron is trading near $1.80. A slow but steady grind higher, driven more by positioning and expectations than by new hard data – with the first catalyst window now approaching: Q4 earnings and the first full-year RYTELO outlook under the new CEO.
NRX Pharmaceuticals Inc ($NRXP)

NRx Pharmaceuticals is a micro-cap biotech focused on severe psychiatric conditions: suicidal depression, bipolar depression with suicidality, PTSD and related disorders. The core of the story is NRX-100, a preservative-free intravenous ketamine program supported by a large Real-World Evidence dataset, and NRX-101, an oral D-cycloserine + lurasidone combination with Breakthrough designation. Around those assets, the company is building HOPE Therapeutics, a network of interventional psychiatry clinics that should both validate the care model and generate revenue.
Ocular Therapeutix Inc ($OCUL)

Positive 52-week SOL-1 data position AXPAXLI as a potential long-acting option versus aflibercept, yet the stock sells off as investors question trial design, real-world relevance and the regulatory path.