DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
BFRI Biofrontera Inc

Photodynamic-therapy specialist with a single commercial franchise (Ameluz + RhodoLED) in U.S. dermatology, entering a dense 2026 data and regulatory window while revenues scale from the low–mid $40M range and the balance sheet remains tight.
PHAR Pharming Group Adr-CRL

On February 1, 2026, Pharming announced that the U.S. FDA issued a Complete Response Letter (CRL) to its supplemental New Drug Application (sNDA) for Joenja (leniolisib) in children aged 4 to 11 years with activated phosphoinositide 3-kinase delta syndrome (APDS). The letter does not question the fundamental clinical benefit of leniolisib in APDS but raises two specific technical concerns
AQST Aquestive Therapeutics Inc

From the January “deficiencies” shock to a Complete Response Letter focused on packaging and administration, why the market is suddenly relieved and why the story is still a multi-quarter grind.
CRMD CorMedix Inc

CorMedix is no longer the pre-revenue story that dialysis investors watched from a distance. After the U.S. launch of DefenCath in 2024, the company has delivered its first profitable quarter, acquired the Melinta anti-infective portfolio, built a meaningful cash position and, as of February 2026, added a $75 million share repurchase program on top.
ARTV Artiva Biotherapeutics Inc

Artiva Biotherapeutics is a clinical-stage biotech company focused on off-the-shelf allogeneic NK cell therapies built on a standardized, donor-derived, manufacturing platform. The lead asset, AlloNK (AB-101), is being developed in combination with anti-CD20 therapy both in B-cell non-Hodgkin lymphoma (NHL) and in autoimmune d
ICU SeaStar Medical Holding Corp
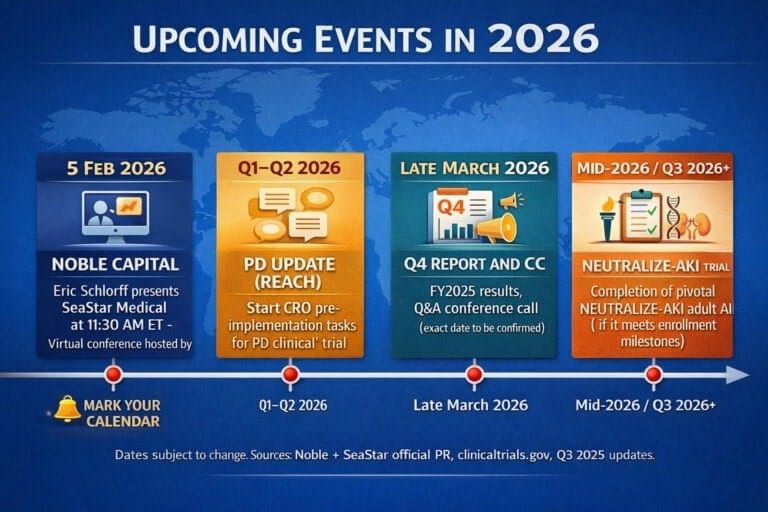
SeaStar Medical is not a typical “pre-clinical biotech on a slide deck”. The company has already crossed the line into real-world use: QUELIMMUNE (the pediatric implementation of the SCD) is approved by the US Food and Drug Administration via HDE and has been adopted by a growing group of leading US children’s hospitals. Early registry data suggest improved survival and kidney recovery in some of the sickest patients in the ICU, with no device-related safety signals reported so far
DRTS Alpha Tau Medical Ltd Jan Update

We wrote our original bullish editorial at the very end of December 2025, when the stock was still trading around five dollars and largely under the radar. January brought ASCO GI pancreatic data, a first PMA module to the FDA, a shareholder letter and a sharp re-rating. Here we revisit the thesis, check the fundamentals and map the next catalysts. Educational only, not investment advice.
ALLO Allogene Therapeutics Inc

Allogene Therapeutics is not the 2+ billion dollar hype story it was a few years ago. After the initial enthusiasm around off-the-shelf CAR T, the stock has been crushed by a sequence of events: an FDA clinical hold in 2021, years of execution risk, pipeline reshuffling and workforce reductions.
AMRN Amarin Corp ADR Update Jan 31

A few years ago, the Amarin story was dominated by a single word: generics. After losing key patent battles on Vascepa, the U.S. business was hit by an aggressive wave of skinny-label competitors, PBM exclusions and net price pressure, to the point where many investors simply filed AMRN under “broken story”. At the start of 2026, the picture is no longer that binary. The company is still far from the glory days, but the trajectory is clearly different: less bleeding, more structure, and a gradual shift from survival mode to controlled recovery.
SLS SELLAS Life Sciences Group Inc Update Jan 31

REGAL Survival Clock, SLS009 Momentum and the Waiting Game
Monthly recap on SELLAS Life Sciences: what changed since the December update, how the 72/80 events in the REGAL Phase 3 trial reshaped the narrative, and which catalysts traders are watching next.
ALT Altimmune Inc
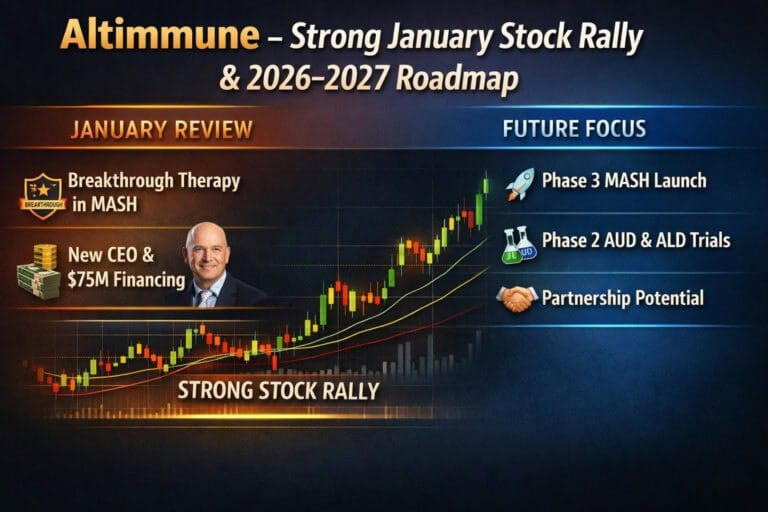
From 48-week IMPACT data and a CEO switch to FDA Breakthrough status, a 75M$ raise and a +50% month: how the Altimmune story and the tape have changed in a few weeks
IBRX ImmunityBio Inc Update Jan 31

Since the last Merlintrader pieces on ImmunityBio, the story has accelerated on several fronts. Anktiva net product revenue exploded to a preliminary 113 million dollars for 2025, a roughly 700 percent year-over-year jump with 38.3 million in the fourth quarter alone, backed by strong unit growth and broader coverage in the United States and abroad, as detailed in the company’s mid-January 2026 update and echoed across financial coverage.
Week ahead and recap jan 31 2026

Week Ahead and Recap – January 31, 2026
Gold near record highs, AI shockwaves in gaming, and a cluster of high-grade biotech catalysts from DF-003 in ROSAH syndrome to camizestrant in ESR1-mutant breast cancer. This is your condensed map for the week.
PLX Protalix BioTherapeutics Inc

The core of the Protalix model today is Elfabrio (pegunigalsidase alfa) for Fabry disease, commercialized globally by Chiesi under the two Chiesi agreements described in the FY 2024 release and 10-K. :contentReference[oaicite:2]{index=2} Protalix manufactures the drug substance in Carmiel and sells finished product to Chiesi, receiving tiered payments in the 15–35% ex-US and 15–40% US range on average selling price, linked to purchased volumes, as outlined in the “Chiesi Agreements” and “Revenues from selling goods” sections.
KYTX Kyverna Therapeutics Inc

Deep dive (as of January 30, 2026) on Kyverna Therapeutics (KYTX): autologous CD19 CAR-T mivocabtagene (miv-cel, KYV-101) in autoimmune diseases, positive registrational KYSA-8 data in stiff person syndrome (SPS), RMAT/Fast Track designations, 2026 BLA ambitions and cash runway into 2027. Educational only, not investment advice
TLSA Tiziana Life Sciences Ltd

Tiziana Life Sciences is building an entire company around one idea: that intranasal delivery of a fully human anti-CD3 antibody, foralumab, can re-educate the immune system and cool down chronic neuroinflammation without broad systemic immunosuppression.
QNCX Quince Therapeutics Inc

Quince Therapeutics (QNCX) released topline results from its pivotal Phase 3 NEAT clinical trial of eDSP (EryDex) in Ataxia-Telangiectasia, a rare pediatric neurodegenerative and immunodeficiency disorder. The press release confirms that the study did not meet its primary endpoint on the Rescored modified International Cooperative Ataxia Rating Scale (RmICARS), with insufficient separation versus placebo despite prior Phase 2 and ATTeST signals
AARD Aardvark Therapeutics Inc

Aardvark Therapeutics (AARD) is one of the more interesting new names in the rare-disease/obesity intersection: a clinical-stage biotech built around a gut-restricted small-molecule (ARD-101) targeting bitter taste receptors (TAS2Rs) to modulate hunger signaling. The core story is very focused: Prader–Willi syndrome (PWS), an ultra-rare genetic disease dominated by uncontrolled hyperphagia and weight gain, with ARD-101 in a pivotal Phase 3 trial (HERO) that has aligned with the FDA and is intended to support an NDA filing if successful.
GUTS Fractyl Health Inc

“Good headline, bad read” session
Fractyl reported 6-month randomized midpoint data from a blinded, sham-controlled cohort in REMAIN-1. The release highlights less weight regain vs sham, better cardiometabolic profile, and a De Novo pathway discussion. The stock is getting hit because traders are discounting the quality of the signal and the timeline.
KALV KalVista Pharmaceuticals Inc

From “great science” to “great delivery”: Ekterly’s launch is now the purest real-world test of whether KalVista can turn first-in-class oral HAE convenience into durable cash flow before competition closes the gap.