DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
MBRX Moleculin Biotech
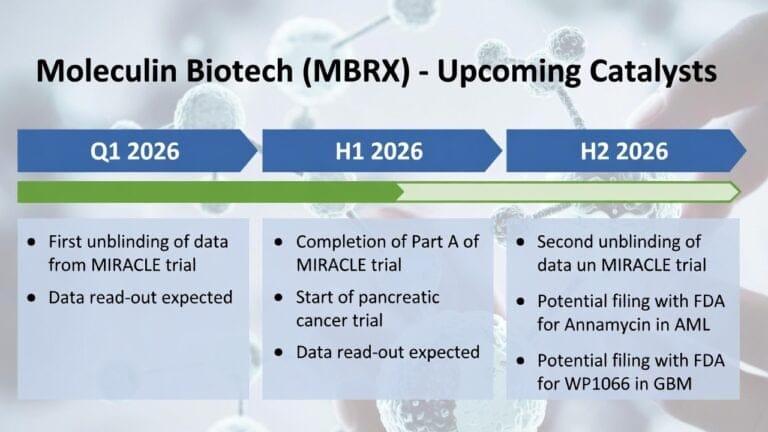
Moleculin Biotech, Inc. (NASDAQ: MBRX) is a Phase 3 clinical-stage pharmaceutical company dedicated to developing innovative therapies for hard-to-treat tumors and viruses. The company's lead candidate, Annamycin, is advancing in the pivotal Phase 3 MIRACLE trial for relapsed/refractory acute myeloid leukemia (AML), with the first unblinding of data anticipated in Q1 2026.
SER Serina Therapeutics Inc.

Serina Therapeutics is a clinical-stage biotechnology company based in Huntsville, Alabama, built around its proprietary POZ Platform, a drug optimization technology based on a controlled, water-soluble poly(2-oxazoline) polymer. The platform is designed to tune half-life, exposure and tissue distribution of small molecules, peptides and biologics, with the ambition of generating “best-in-class” dosing profiles versus conventional formulations.
TVTX Travere Therapeutics Inc

The FSGS decision remains a high-impact regulatory binary. The piece focuses on the run-up logic but does not remove the fundamental FDA risk from the picture.
CAPR Capricor Therapeutics Update feb08
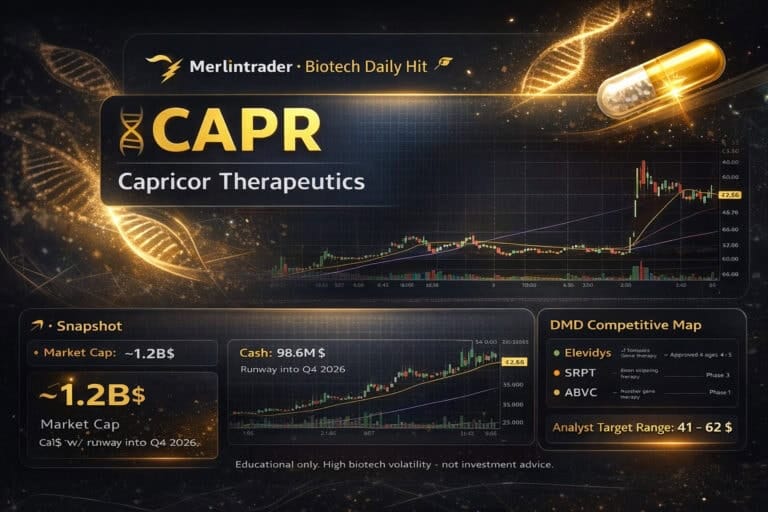
FDA asks for the full HOPE-3 clinical study report but does not require new trials, while Capricor prepares a February BLA update. The stock hovers around 23–24 $, with a market cap near 1.16 B$, after a 2025 rally fuelled by Duchenne data and aggressive retail positioning.
RGNX Regenxbio Inc Update Jan28
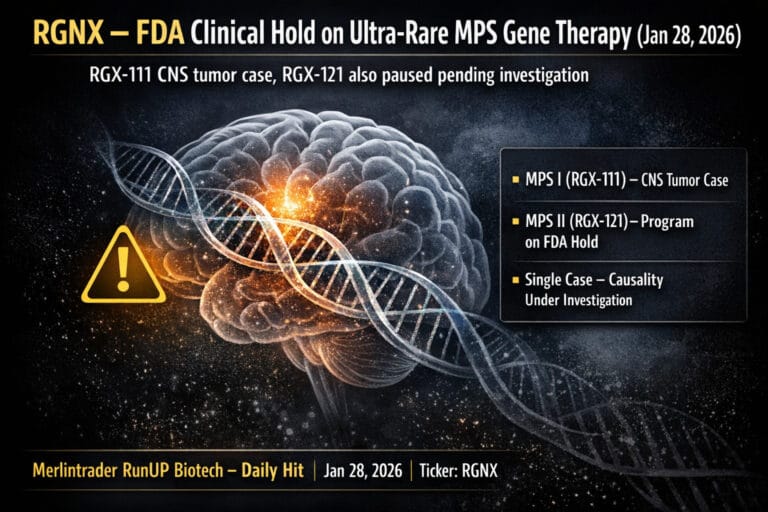
FDA places a clinical hold on RGX-111 (MPS I) and RGX-121 (MPS II) programs after a single intraventricular CNS tumor in an RGX-111 patient.
Preliminary analysis shows an AAV vector integration event near proto-oncogene PLAG1; causality is still under investigation.
NAVSUNLI (RGX-121) BLA for MPS II remains under priority review with a PDUFA date of February 8, 2026; no public change to that date has been announced as of this report.
CRDF Cardiff Oncology Inc
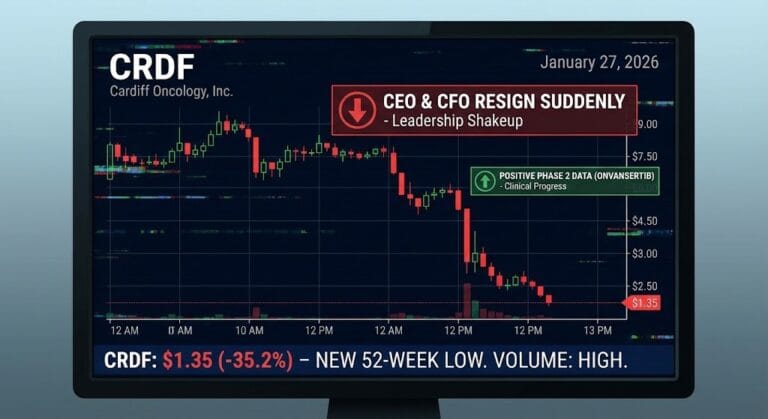
As of January 27, 2026, Cardiff Oncology (NASDAQ: CRDF) is trading around 2 dollars per share after a single-session loss of roughly thirty-two percent. The trigger was a GlobeNewswire press release announcing that long-time Chief Executive Officer Mark Erlander and Chief Financial Officer James Levine are both stepping down, with board member Mani Mohindru becoming interim CEO and Senior Vice President of Finance Brigitte Lindsay moving up as interim CFO.
NTLA Intellia Therapeutics Inc
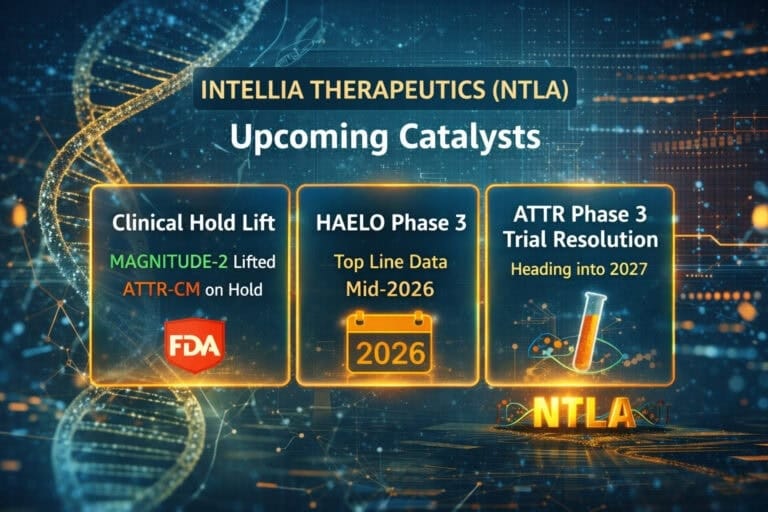
As of January 27, 2026, Intellia Therapeutics (NASDAQ: NTLA) is back in the spotlight. The FDA has lifted the clinical hold on MAGNITUDE-2, the Phase 3 study of its in vivo CRISPR therapy nexiguran ziclumeran (“nex-z”, formerly NTLA-2001) in hereditary transthyretin amyloidosis with polyneuropathy (ATTRv-PN), while a separate large cardiomyopathy trial (MAGNITUDE, ATTR-CM) remains on hold after a Grade 4 liver event and a subsequent patient death triggered intense scrutiny.
COYA Therapeutics Holdings Inc

COYA Therapeutics Holdings represents a high-risk opportunity in the biotech sector, focused on developing innovative therapies for neurodegenerative and autoimmune diseases through regulatory T-cell (Treg) modulation. With a pipeline including candidates for ALS, Alzheimer's, and other complex pathologies, the company positions itself in a therapeutic area with significant unmet medical needs but also with high clinical failure rates.
KURA Kura Oncology Inc

Kura Oncology is a precision oncology company built around one central thesis: targeting oncogenic signalling nodes such as menin and farnesyl transferase can reset the biology of hard-to-treat cancers, especially acute myeloid leukemia (AML).
ATYR Atyr Pharma Inc

aTyr Pharma ($ATYR) is a clinical-stage biotech developing first-in-class therapies from its tRNA synthetase platform, targeting fibrotic and inflammatory lung diseases. The company's lead asset, efzofitimod (a selective NRP2 modulator), suffered a major setback with the Phase 3 EFZO-FIT trial miss in pulmonary sarcoidosis (announced Sept 15, 2025), causing an ~80% stock drop from ~$4-5 to sub-$1 levels.
VNDA Vanda Pharmaceuticals Inc

Vanda is not a pre-revenue biotech: it is a commercial-stage CNS company with three marketed products (Fanapt, Hetlioz, Ponvory) and two late stage assets that matter for the next 12–24 months: Nereus (tradipitant) for motion sickness, now approved, and Bysanti (milsaperidone) with a PDUFA target action date on 21 February 2026 for schizophrenia and bipolar I disorder.
VRDN Viridian Therapeutics Inc

Viridian Therapeutics (VRDN) has started 2026 by reaffirming its "transformational" year narrative in the January 6 PR, which serves as a comprehensive roadmap update without any timeline shifts or negative surprises. This builds directly on the December 22, 2025 BLA acceptance for veligrotug (VRDN-001) in TED with Priority Review and PDUFA target of June 30, 2026 (as detailed in the previous report).
Celc Celcuity Inc
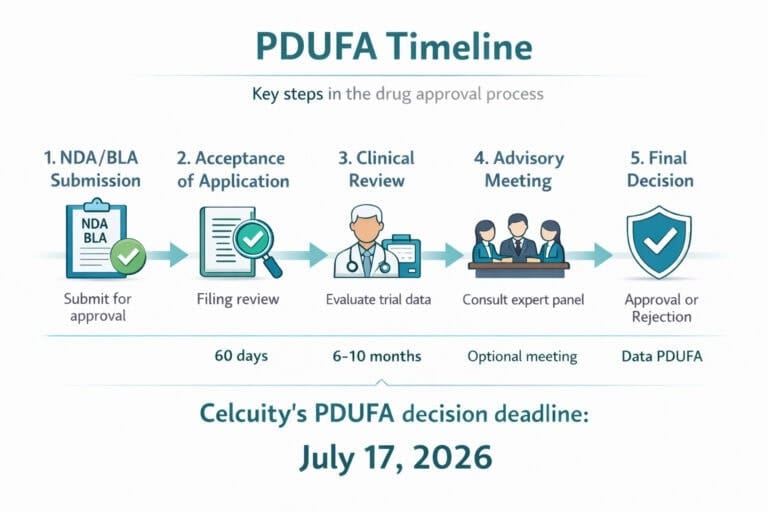
Celcuity (CELC) – Gedatolisib PDUFA July 17, 2026: Setup, Risks and RunUP Map | Merlintrader trading Blog English Italiano Celcuity (CELC) – Gedatolisib PDUFA July 17, 2026 PI3K/mTOR inhibitor in HR+/HER2- metastatic breast cancer – binary PDUFA, cash runway and…
RGNX Regenxbio

RGX-121 puts Regenxbio at the threshold of its first potential standalone approval, while the retina (AbbVie) and Duchenne programs build a second and third pillar for 2026–2027. The equity story is a classic “platform + royalty + binary PDUFA” mix, with a balance sheet extended into early 2027 via upfronts and royalty monetization.
CVKD Cadrenal Therapeutics Inc

Cadrenal Therapeutics (NASDAQ: CVKD) is a clinical-stage biopharma headquartered in Ponte Vedra, Florida, focused on anticoagulation solutions for high-risk patients who are poorly served by current therapies. The company was formed in 2022 around tecarfarin and, through two 2025 acquisitions, has expanded into a broader platform combining VKA, Factor XIa and 12-LOX mechanisms.
Weekly Briefing – Fed, Geopolitics, GLP-1 Obesity & Small Caps

Going into the 27–28 January FOMC, the message is basically “don’t expect fireworks”: after the 2025 cuts, the Fed keeps describing policy as “well positioned”, with inflation gliding down towards (but not yet at) target and real growth still positive. Futures now see a very high probability of a clean hold next week and push the bulk of any extra easing into late 2026.
NMRA Neumora Therapeutics

Deep dive on Neumora Therapeutics (NMRA): navacaprant reset, NMRA-511 Alzheimer’s agitation data, M4 franchise and a 2026–2027 catalyst map for biotech run-up traders. Educational only, not investment advice.
IOVA Iovance Biotherapeutics Inc

Iovance Biotherapeutics (IOVA) – From first TIL approval to industrial stress-test in solid tumors (Playbook 2026–2027) | Merlintrader trading Blog Merlintrader trading Blog – RunUP Biotech EN IT IOVA – Iovance Biotherapeutics (NASDAQ) Static daily chart from Finviz – click…
ETON Pharmaceuticals Inc

After years of being “just a pipeline story”, Eton has transitioned into a small but diversified rare-disease platform with multiple approved assets and a focused late-stage pipeline anchored by ET-400 and ET-600.
AQST & PHAR -Countdown Jan 31 PDUFA

We are on Friday, January 23, 2026 (Italy time). For January, in your catalyst tracker there are essentially two PDUFAs still “missing” that matter for this piece: Aquestive (AQST) with Anaphylm and Pharming (PHAR) with the pediatric Joenja sNDA (4–11 years, APDS). Both currently carry a PDUFA target action date of January 31, 2026.