DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
Humacyte $HUMA

From FDA approval of Symvess for vascular trauma to Ukrainian war-zone data and fresh U.S. support for biologic vascular repair, Humacyte is trying to turn a once “sci-fi” idea – off-the-shelf human vessels grown in a factory – into something routine in emergency rooms and military hospitals.
ImmunityBio Inc $IBRX

Potential resubmission of the supplemental BLA for BCG-unresponsive papillary NMIBC following the January 20 FDA meeting. The company committed to deliver additional analyses within 30 days, without new clinical trials. If the resubmission is accepted, the review clock could define the next major run-up window.
Biofrontera Inc $BFRI
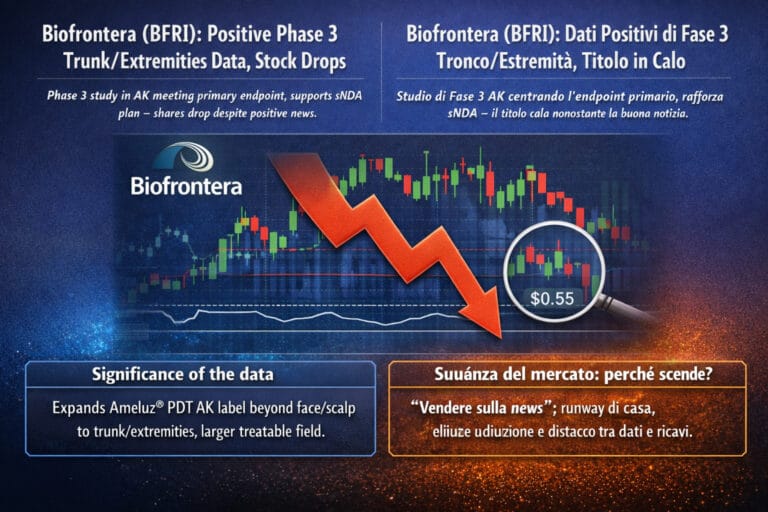
Biofrontera (BFRI) – Positive Phase 3 trunk/extremities data for Ameluz PDT, sharp micro-cap sell-off
Biofrontera reports that its Phase 3 study of Ameluz® PDT for actinic keratoses on the extremities, neck and trunk met the primary endpoint, strengthening the case for a 2026 sNDA – but the stock trades lower intraday, reminding investors how noisy tiny-cap price action can be.
Protara Therapeutics $TARA

Protara Therapeutics (TARA) — TARA-002 Phase 2 NMIBC catalyst at the end of February 2026
Protara Therapeutics is a small-cap biotech that has quietly moved from “option on survival” to a fully funded late-stage story built around a single, high-leverage asset: TARA-002. The drug is a TLR2/NOD2 agonist derived from the same cell bank as the long-used immunotherapy OK-432 (Picibanil) and is being developed both for non-muscle invasive bladder cancer (NMIBC) and for pediatric lymphatic malformations. The near-term catalyst now staring at the tape is an updated Phase 2 read-out in BCG-unresponsive NMIBC that will be presented on 27 February 2026 and that the company itself frames as aligned with the registrational pathway for this setting.
Cognition Therapeutics $CGTX

Cognition Therapeutics, Inc. is a clinical-stage biopharmaceutical company focused on developing innovative, small-molecule oligomer antagonists for age-related degenerative disorders of the central nervous system, primarily Alzheimer's disease (AD) and Dementia with Lewy Bodies (DLB).
Compass Therapeutics (CMPX)

Clinical-stage oncology company focused on bispecific antibodies that couple angiogenesis and immune modulation.
KOD Kodiak Sciences Feb 08 2026

KOD
Kodiak Sciences Inc.
Late-stage retina biotech riding a multi-hundred-percent run-up into a dense 2026–2027 catalyst cluster (tarcocimab, KSI-501 and KSI-101). Chart from Finviz, always double-check the latest quote and news.
OCUL Ocular Therapeutix run-up Updated Feb 16 2026

Deep dive on Ocular Therapeutix: from DEXTENZA to the late-stage AXPAXLI retina franchise, with SOL-1 topline data in wet AMD coming in late February 2026 and a balance sheet funded into 2028.
Weekly Briefing Feb 9 2026
Week of 9–15 February 2026 – the setup is unusual: the U.S. government shutdown pushed key releases into the same week, so the market gets a jobs + CPI combo while earnings keep dictating leadership.
AGIO Agios Pharmaceuticals Inc

A year ago, Agios was still mostly a “promise stock”: one approved niche drug for pyruvate kinase deficiency and a lot of hope around sickle cell disease. Today the picture is more complex but also more concrete: mitapivat now has two U.S. approvals, a full rare-disease haematology franchise is forming, and 2026 has a surprisingly busy catalyst map.
PLSE Pulse Biosciences Inc

First-in-human data for the nPulse™ Cardiac Catheter Ablation System show high pulmonary vein isolation durability and low SAE rate in atrial fibrillation – but this is still a pre-commercial, single-platform story.
RCKT Rocket Pharmaceuticals
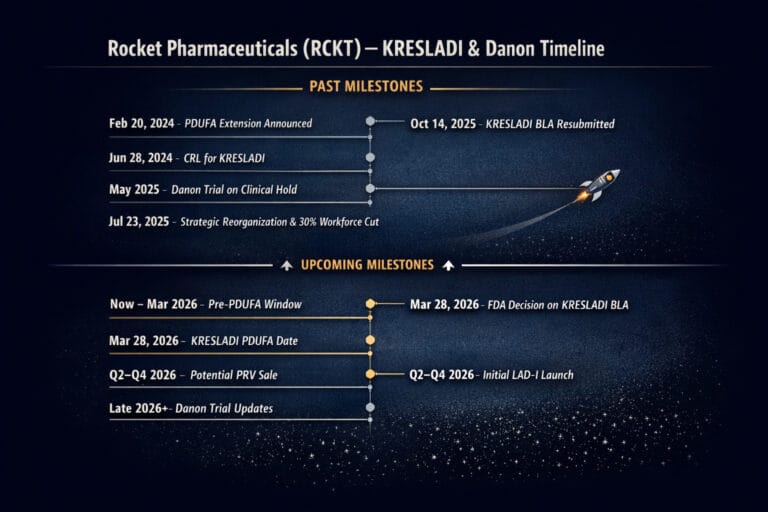
Gene therapy for ultra-rare immunodeficiency (LAD-I), a hard reset after last year’s CRL and the Danon shock, and a March 28, 2026 PDUFA that could flip sentiment in a name now trading near multi-year lows. This is a classic “platform vs. execution” test in gene therapy – with an additional potential upside lever in the form of a Rare Pediatric Disease Priority Review Voucher (PRV) if approval comes through.
ALVO Alvotech Feb 2026

Alvotech is not a typical development-stage biotech: it is a vertically integrated biosimilar platform that already generates hundreds of millions of dollars in revenue from launched products like AVT02 (adalimumab) and AVT04 (ustekinumab), while also running a crowded late-stage pipeline. The stock sits in that awkward middle ground between “early growth” and “heavy balance sheet”, with over 1B$ of debt but also positive adjusted EBITDA and operating profit in the latest 9-month period.
CRMD Thinking After Hours

This after-hours note starts from a simple, honest message in the comments. A reader who first bought CRMD around 11 USD, then averaged down at 9 USD and again at 6.5 USD, with one clear idea: one day, selling “north of 15” if the CorMedix story plays out.
RNXT RenovoRx Inc
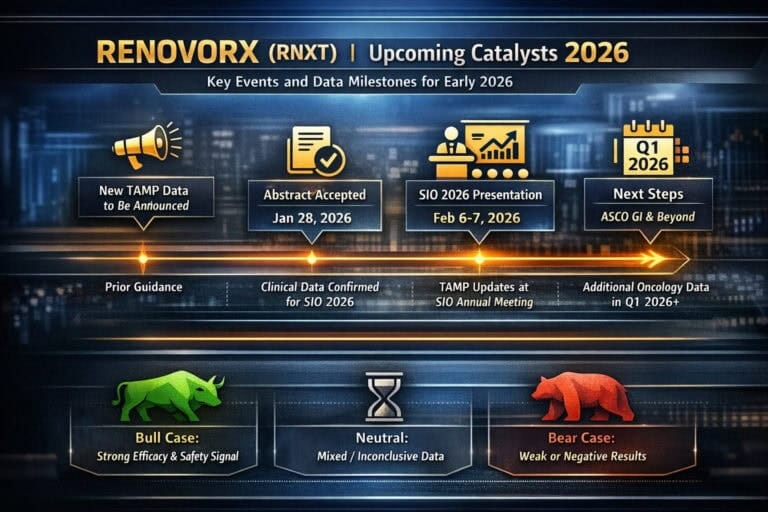
Quick take: RenovoRx brings fresh clinical data on its TAMP™ therapy platform to SIO 2026 (February 6). The event is not a registrational readout, but it is a real-world test of how much the market still cares about this targeted intra-arterial delivery story as the company starts to generate device revenues.
DFTX Definium Therapeutics Inc.

Definium Therapeutics (DFTX) is not a mature commercial pharma story; it is a clinical‑stage CNS and mental health company whose equity behaves like a classic high‑beta biotech: the tape is dominated by expectations around clinical data, regulatory interactions, and the company’s ability to fund its programs through to value‑defining milestones.
ONCY Oncolytics Biotech, Inc

On February 4, 2026, Oncolytics announced that the FDA granted Fast Track designation to pelareorep combined with bevacizumab and FOLFIRI for adults with KRAS-mutant, microsatellite-stable metastatic colorectal cancer (MSS mCRC) in the second-line setting, after progression on a first-line oxaliplatin-based regimen. The designation is based on clinical signals from the REO 022 study and prior pelareorep experience across GI tumors and breast cance
VSTM Verastem Inc

Verastem Oncology enters 2026 as a very different story compared with two years ago. The company now has a commercial product, Avmapki Fakzynja (avutometinib + defactinib), approved under the FDA’s accelerated approval pathway for adult patients with KRAS-mutant recurrent low-grade serous ovarian cancer (LGSOC) after at least one prior systemic therapy.
ADMA Adma Biologics Inc

Commercial IG platform with three FDA-approved products, a yield-enhanced manufacturing process, and a revenue ramp from <$200M to >$500M in a handful of years. Not a binary biotech story, but an execution and capacity game that the market is finally treating like a growth compounder.
OMER Omeros Corporation

What happened in January: Omeros successfully transitioned from pre-commercial biotech to commercial-stage rare disease company with the FDA approval of YARTEMLEA on December 23, 2025 and first commercial sales announced January 27, 2026.