DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Daily Hits
Hims & Hers Health ($HIMS): April 2026 deep dive after the GLP-1 reset, the Novo pivot, and the new peptide optionality

A full April 2026 Merlintrader-style update built on our prior HIMS work and focused on what changed since the last report: the Q4 and full-year 2025 numbers, the strategic reset in U.S. weight loss, the agreement with Novo Nordisk, the still-pending Eucalyptus acquisition, the slowing but still meaningful 2026 growth outlook, and the fresh peptide angle that has suddenly reopened part of the long-term story.
Planet Labs ( $PL ): Berlin expansion adds industrial depth to a stock already repriced by earnings

Today’s Berlin manufacturing update is not a “headline contract” event, but it matters because it fits the exact story the market has been rewarding in recent weeks: Planet is being re-rated less as a pure imagery vendor and more as a strategic geospatial infrastructure platform with sovereign, defense and AI-linked relevance.
Rocket Pharmaceuticals Inc ( $RCKT ) refreshes its ATM 17 days before the March 28 PDUFA

This was not a classic overnight equity raise. Rocket filed a new ATM framework with Cantor Fitzgerald, terminated the prior TD/Cowen agreement, and did so with the March 28, 2026 PDUFA for KRESLADI now close enough to matter in every financing read-through.
ImmunityBio (NASDAQ: IBRX) — FDA resubmission

What happened today
Today’s real headline is straightforward: ImmunityBio announced that it has resubmitted the supplemental biologics license application to the FDA for ANKTIVA plus BCG in patients with BCG-unresponsive non-muscle invasive bladder cancer with papillary disease.
According to the company, it had already submitted information requested by the FDA in February 2026, but in March the agency then requested updated efficacy information based on longer follow-up, which ImmunityBio says has now been included in the resubmitted package.
SeaStar Medical Holding Corp ( $ICU )

SeaStar Medical announced completion of the FDA-mandated enrollment requirement (50 patients) for the SAVE Surveillance Registry evaluating QUELIMMUNE safety in pediatric AKI with sepsis requiring continuous kidney replacement therapy (CKRT).
Ocugen Inc ( $OCGN )

Headline today: Ocugen will host its Q4 and full-year 2025 conference call and live webcast today at 8:30 a.m. ET (14:30 CET), giving investors a first chance to hear management on cash, strategy and the OCU400 program, just days after the company announced completion of enrollment in the Phase 3 liMeliGhT trial for retinitis pigmentosa.
Tonix Pharmaceuticals ( $TNXP ) uplisted to Nasdaq Global Select Market

Tonix Pharmaceuticals (TNXP) moves from Nasdaq Capital Market to the higher-tier Nasdaq Global Select Market and confirms a visible presence at TD Cowen and Barclays healthcare conferences in early March 2026.
uniQure N.V ( $QURE ) FDA says AMT-130 data not enough yet, shares collapse

uniQure’s stock is getting crushed after the company disclosed that the U.S. Food and Drug Administration does not consider current Phase I/II data for AMT-130 in Huntington’s disease sufficient to support a marketing application, and is instead recommending a fresh prospective, randomized, double-blind, sham surgery-controlled Phase III trial.
Rocket Pharmaceuticals ( $RCKT ) — Q4/FY 2025 results Road to March28-Pdufa

This update is anchored to the company’s latest filing that laid out (1) the reiterated March 28, 2026 FDA action date for KRESLADI, (2) the resumption plan for Danon dosing in 1H 2026, and (3) the year-end 2025 cash position and runway guidance. The goal is to keep it neutral: what was said, what it implies, and where the remaining uncertainty still lives.
Eton Pharmaceuticals Inc ( $ETON) Feb25 FDA OK

The U.S. Food and Drug Administration has approved DESMODA (desmopressin acetate) oral solution for the treatment of central diabetes insipidus, also known as arginine vasopressin deficiency (AVP-D), in adults and pediatric patients. This is the first and only FDA-approved ready-to-use oral solution of desmopressin, a meaningful upgrade in day-to-day management of this rare water-balance disorder.
Vanda Pharmaceuticals Inc ($VNDA)

The FDA has approved BYSANTI™ (milsaperidone) tablets as a first-line atypical antipsychotic for the acute treatment of manic or mixed episodes in bipolar I disorder and for the treatment of schizophrenia in adults. This is Vanda’s second FDA approval in under two months, following NEREUS™ for motion-induced vomiting prevention.
Geron Corp ($GERN) Feb 18 Update
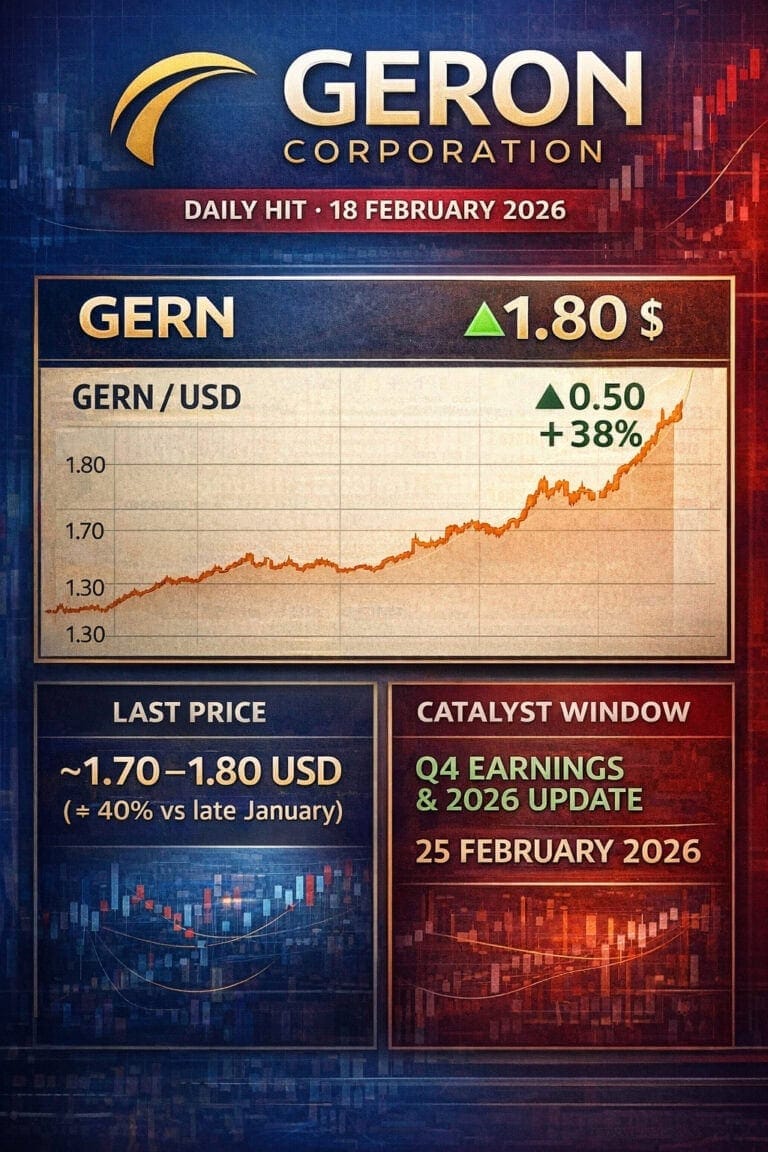
Almost one month after our long-form report at around $1.30, Geron is trading near $1.80. A slow but steady grind higher, driven more by positioning and expectations than by new hard data – with the first catalyst window now approaching: Q4 earnings and the first full-year RYTELO outlook under the new CEO.
Ocular Therapeutix Inc ($OCUL)

Positive 52-week SOL-1 data position AXPAXLI as a potential long-acting option versus aflibercept, yet the stock sells off as investors question trial design, real-world relevance and the regulatory path.
ImmunityBio (IBRX) — Saudi SFDA engagement

Two items in one tape-read: (1) a fresh BusinessWire update on Saudi regulatory engagement (rBCG dossier + ANKTIVA/CPI expansion discussions), and (2) a sell-side note in circulation reiterating a $24 price target from D. Boral Capital (note dated Jan 13, 2026 in the sources below).