DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
Greenwich LifeSciences ( $GLSI ): FLAMINGO-01 Reorders the Story, but the Trial Is Still Binary

GLSI is exactly the kind of name that can look much cleaner in a press release than it does in real portfolio construction. The company has one lead asset, one pivotal clinical thesis, limited cash, ongoing need for financing, and a story that can produce violent stock reactions because the float is relatively small and the clinical promise is easy to narrate. That is why the most useful way to rebuild the article in the new format is to put the scientific signal and the financing reality in the same frame from the first page.
Olema Pharmaceuticals Inc ( $OLMA ) deep dive march 17 2026

Olema sits in one of the most interesting transition zones in clinical-stage biotech. It is no longer a company that can be dismissed as little more than a speculative preclinical story, because it now has a late-stage development strategy centered on palazestrant, a second clinical engine in OP-3136, and a balance sheet that looks strong enough to bridge the company through several major milestones. At the same time, Olema is not yet a diversified oncology company with multiple independent late-stage assets that can absorb a major failure without a violent valuation reset. That is why the stock remains so fascinating. It offers a genuine setup for a re-rating if execution continues, but it also remains highly exposed to a narrow band of high-information events.
Structure Therapeutics Inc ADR ( $GPCR ) DD march 16 2026

Structure Therapeutics was founded with a singular mission: to build a structure-based GPCR (G-protein-coupled receptor) drug discovery platform. The company's name literally reflects its core intellectual property—the belief that understanding the three-dimensional structure of GPCR proteins could enable design of highly selective, potent small molecule drugs that traditional high-throughput screening couldn't easily find.
Annovis Bio ( $ANVS ) – Deep Dive on Buntanetap 2026

Annovis is one of those biotech names that looks tiny on the screen but can become very large in narrative terms whenever the market wants a fresh Alzheimer’s angle. The company’s March 16, 2026 fiscal-year update did not announce an approval, an acquisition or a phase-changing headline readout. What it did provide was something more practical: a clean status check on the one thing that matters most for Annovis today, namely whether its pivotal Alzheimer’s program is moving forward fast enough to keep the investment story alive before the cash window narrows too much.
NRx Pharmaceuticals ( $NRXP ) – FDA Minutes Lock In the NRX-100 NDA Path

NRx Pharmaceuticals finally got the kind of news biotech traders usually wait for: not just another optimistic corporate interpretation, but formal written FDA minutes that document the regulatory path. In today’s release, the company said it received the confirmatory written minutes from its February 17, 2026 in-person Type C meeting with the FDA. According to NRx, those minutes support a path to file an NDA for NRX-100 by June 2026 using existing adequate and well-controlled clinical trial data together with confirmatory Real World Evidence. The company also said the FDA confirmed that no additional clinical trials and no bridging studies are required for the preservative-free formulation
Vertex Pharmaceuticals ( $VRTX ) no longer just a cystic fibrosis machine

Vertex Pharmaceuticals remains one of the highest-quality large-cap biotech stories in the market, but the real reason the name deserves a first full Merlintrader deep dive now is not simply scale, profitability or balance-sheet strength. The more important point is that the company has reached a stage where investors can begin to judge whether the long-promised diversification beyond cystic fibrosis is becoming economically meaningful rather than merely aspirational.
Weekly Market Recap & Next Week Prep — March 15, 2026
The week that ended on Friday, March 13, did not deliver a clean “risk-on” or “risk-off” message. It delivered something more difficult and, for active traders, more important: a selective tape in which macro stress was real, energy shock risk was impossible to ignore, but not every pocket of the market reacted in the same way. That distinction matters. A market that falls together can often bounce together. A market that starts separating winners from losers under pressure is usually telling you that stock-picking, catalyst timing and sector choice are starting to matter more than headline beta.
Aldeyra Therapeutics ( $ALDX ): past, present and future around reproxalap and the March 2026 FDA decision

A long-form bilingual deep dive built around the full regulatory arc of Aldeyra, from the first dry-eye filing to the 2025 collapse and the current March 2026 binary setup. The focus here is not only on what happened, but on why the market still cares, what could realistically change if reproxalap is approved, what the damage would look like if it is rejected again, and how retail traders are framing the setup in the final stretch before the decision.
ImmunityBio Inc ( $IBRX ) today’s NK-cell manufacturing milestone is real

Today’s official release from ImmunityBio was about cell-manufacturing execution, not about ANKTIVA sales, not about a fresh FDA approval, and not about a pivotal top-line readout. The company said it has completed manufacturing engineering programs that established a reproducible leukapheresis-to-manufacturing pathway for its autologous M-ceNK platform. In simple English, ImmunityBio is telling the market that it has moved another step from “interesting immunology concept” toward “repeatable cell-therapy process that can be run at scale.”
5 energy stocks under $2 billion to watch in march ( $PUMP $EGY $RNGR $GEOS $KLXE )

This article compares five U.S.-listed energy names below $2 billion in market value that could benefit, directly or indirectly, from a stronger oil tape, firmer field activity, or renewed investor appetite for smaller-cap energy exposure: ProPetro (PUMP), VAALCO Energy (EGY), Ranger Energy Services (RNGR), Geospace Technologies (GEOS) and KLX Energy Services (KLXE).
Immutep Limited ( Nasdaq $IMMP ) after TACTI-004 futility

Core fact
On March 13, 2026 Immutep said the Independent Data Monitoring Committee recommended discontinuing the registrational Phase III TACTI-004 study following the planned futility analysis.
Artiva Biotherapeutics Inc ( $ARTV ) March 11th

Artiva Biotherapeutics rallied sharply after its full-year 2025 results and business update put the spotlight back where biotech traders care most: near-term clinical readouts, FDA path visibility, analyst support, and whether the balance sheet is strong enough to carry the story into the next real value-inflection point.
Hims & Hers ( $HIMS ) from GLP-1 backlash to a big strategic reset

A few weeks ago the core question was whether Hims had pushed the GLP-1 trade too far and invited a regulatory and legal backlash that could damage one of the market’s favorite growth stories. That question has not disappeared. But the story has changed meaningfully. The company now has a formal strategic shift in U.S. weight loss, a public collaboration with Novo Nordisk, access to branded Wegovy and Ozempic on the platform, a dropped lawsuit, and still the pending Eucalyptus acquisition that could reshape the company from a fast U.S. telehealth operator into a broader global consumer-health platform.
Ocugen Inc ( $OCGN ) why the stock exploded, what the dual analyst boost really means

Ocugen came back to life on a violent session after a stacked sell-side day: one firm initiated with an Outperform and a $10 target, while another raised its target to $22 from $15 and kept a Buy rating. But the move does not sit on analyst language alone. It lands on top of a Phase 3 enrollment-complete lead program, a visible BLA path, multiple ocular follow-on catalysts, a high-beta retail base, meaningful short interest, and a balance sheet that is improved from panic but still nowhere near bulletproof.
Capricor Therapeutics Inc ( $CAPR ) FDA PDUFA Approval Target (August 22, 2026)

On March 10, 2026, Capricor Therapeutics announced that the FDA has lifted its Complete Response Letter (CRL) and resumed review of the Biologics License Application (BLA) for Deramiocel. The agency assigned a new PDUFA target action date of August 22, 2026—a pivotal moment after months of regulatory uncertainty.
ImmunityBio (NASDAQ: IBRX) — FDA resubmission

What happened today
Today’s real headline is straightforward: ImmunityBio announced that it has resubmitted the supplemental biologics license application to the FDA for ANKTIVA plus BCG in patients with BCG-unresponsive non-muscle invasive bladder cancer with papillary disease.
According to the company, it had already submitted information requested by the FDA in February 2026, but in March the agency then requested updated efficacy information based on longer follow-up, which ImmunityBio says has now been included in the resubmitted package.
IMMP under halt: what may really be behind it ( $IMMP )

Immutep’s Nasdaq-listed ADRs were halted on Friday, March 6, 2026 under code T1 / News Pending. On the Australian side, the public halt notice points to an announcement regarding the outcome of the pre-specified interim futility analysis for the Phase III TACTI-004 trial. That single detail matters a lot: it shifts the center of gravity from wild speculation to a very specific clinical event. What follows is a structured look at the facts, the company, the pipeline, and the full range of plausible scenarios.
Weekly Recap & Briefing – Oil Shock, Weak Jobs, Iran War and a More Selective Tape (Mar 2–6, 2026)

The week that closed on Friday 6 March was the first full trading week in which markets had to process the new Middle East war regime rather than just react to a weekend headline. The U.S.–Israeli campaign against Iran and the retaliation that followed kept energy, shipping and inflation risk at the centre of the tape, while the S&P 500 ended the week lower, the VIX climbed to its highest level since April 2025 and Brent crude pushed above 90 dollars a barrel. On top of that, Friday’s U.S. jobs report added a second layer of stress: payrolls unexpectedly fell and the unemployment rate moved up, pushing investors into an awkward mix of growth fear and inflation fear at the same time.
PMV Pharmaceuticals Inc ( $PMVP )
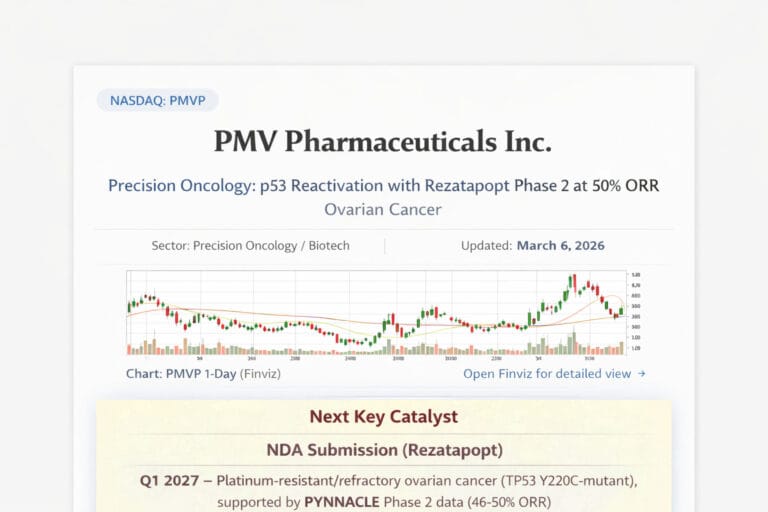
PMV Pharmaceuticals is a precision oncology company built around rezatapopt (PC14586), an oral small-molecule reactivator of p53 designed for tumors harboring the TP53 Y220C mutation. The story matters because TP53 is one of the most important tumor suppressor genes in cancer biology, and because PMV has now moved from mechanism-only interest into clinically visible efficacy data. The company’s registrational PYNNACLE study has produced an overall 34% ORR across evaluable Phase 2 patients and a 46% ORR in the ovarian cohort at the September 4, 2025 cutoff, with the company later reporting a 50% ORR in the same ovarian cohort after that cutoff. FDA Orphan Drug Designation granted on March 2, 2026 adds meaningful regulatory support. PMV says it plans to submit an NDA in Q1 2027 for platinum-resistant/refractory ovarian cancer with TP53 Y220C mutation.
Obesity Medicines 2026 — Part 1: sector map for investors

A stock-market overview built around the names that matter most in the public conversation today: Novo Nordisk (NVO), Eli Lilly (LLY), Viking Therapeutics (VKTX), Structure Therapeutics (GPCR), Altimmune (ALT), Fractyl Health (GUTS) and Zealand Pharma (ZEAL). This first part is not a medical explainer. It is a market map: who already has real commercial power, who is still trying to earn strategic relevance, and which catalysts actually matter for the shares.