DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Category Reports Biotech
Tickers reports and analysis
IBIO iBio Inc

iBio (IBIO) has turned into a small but noisy name in the obesity-drug corner of biotech. It’s a micro-cap (~42–45M $) that has radically repositioned itself as an AI-driven antibody company for obesity and cardiometabolic disease, with a clear—if high-risk—angle: “post-GLP-1” biology focused on Activin E, myostatin/Activin A and the amylin receptor.
NBIX Neurocrine Biosciences, Inc

Companion EN/IT deep dive on Neurocrine Biosciences: INGREZZA as the main cash engine in movement disorders, CRENESSITY as the first new therapy for classic CAH in 70 years, and an expanding psychiatry pipeline (NBI-1117568 and related muscarinic programmes). Focus on 2025–2026 catalysts, risk map and how NBIX fits in a catalyst-driven biotech watchlist.
INCY Incyte Corp

Incyte is a classic mid-cap biotech with a single dominant product (Jakafi) and a long history of oncology R&D. After several high-profile pipeline disappointments, the company has shifted from aggressive expansion to a more selective, risk-controlled development strategy.
VRTX Vertex Pharmaceuticals- CRISPR-Therapeutics

ASGEVY hits primary endpoints in children 5–11 with SCD and TDT – global paediatric filings planned for 1H 2026
Context: ASH 2025 & pre-JPM 2026 – CLIMB-151 (SCD) and CLIMB-141 (TDT) paediatric cohorts (5–11 years).
Vertex has presented the first pivotal data for CASGEVY in children aged 5–11 with sickle cell disease and transfusion-dependent beta-thalassemia. In both Phase 3 trials, all evaluable patients with sufficient follow-up achieved the primary endpoints (absence of vaso-occlusive crises for SCD; sustained transfusion independence for TDT) with VOC-free and transfusion-free periods approaching two years in some cases. Safety so far remains consistent with the adult programme.
JPM Healthcare Conference 2026

The 44th Annual J.P. Morgan Healthcare Conference is widely considered the most influential healthcare investment meeting in the world. Often described as the “Super Bowl of Healthcare”, it brings together more than 8,000 participants, including C-suite executives, institutional investors, policymakers, analysts and media.
KALA Kala Bio Inc

Deep dive on KALA BIO after the CHASE Phase 2b collapse, the Oxford Finance default, David Lazar’s $6M rescue, and the December 26, 2025 completion of the $2M debt settlement that wipes out ~$10.6M in obligations.
AUPH Aurinia Pharmaceuticals Inc

AUPH has been on every biotech retail watchlist since the January 22, 2021 FDA approval of LUPKYNIS (voclosporin) for lupus nephritis. The buyout never came, but the company did the one thing that really matters: it built a profitable, growing LN franchise.
TARA Protara Therapeutics Inc

Key message: the FDA has granted Breakthrough Therapy and Fast Track designations to TARA-002 for the treatment of pediatric macrocystic and mixed cystic lymphatic malformations (LMs), on top of the existing Rare Pediatric Disease designation and inclusion in the FDA’s CMC Development and Readiness Pilot program.
This turns TARA-002 into a rare triple-designation asset in a high-need pediatric space, and gives Protara a clearly accelerated regulatory lane while the stock is still trading in single digits.
ALT Altimmune

Key message: the FDA has granted Breakthrough Therapy Designation (BTD) to pemvidutide for the treatment of MASH, based on 24-week data from the Phase 2b IMPACT trial (company press release, Jan 5 2026 and IMPACT publication).
The stock immediately reacts with an intraday move around +15–20% (as reported by outlets like Seeking Alpha and Investing.com). This is not a random bounce, it is a repricing of regulatory risk.
Weekly Recap and next week outlook dec 29-jan 02 jan 05-jan 09

From PDUFA shocks to early 2026 catalysts: what just happened and what traders should watch next week.
BNGO Bionano Genomics Inc

Bionano Genomics is one of those names where the technology and the share price seem to live in different universes. On the lab side, optical genome mapping (OGM) keeps racking up publications, clinical use-cases and even a Category I CPT code with a 47% reimbursement boost for blood cancers.
AXSM Axsome Therapeutics Inc

Axsome Therapeutics is no longer just “the Auvelity company”. In 2025 it quietly turned into a multi-product CNS platform with three marketed drugs, a late-stage pipeline and, now, a Priority Review from the FDA for AXS-05 in agitation associated with Alzheimer’s disease – with a PDUFA action date set for April 30, 2026.
NVAX Novavax, Inc

From going concern to platform story with a cult retail base
For a long stretch of 2023, Novavax was treated as a failed Covid trade. A late vaccine, production issues, a balance sheet that flirted with collapse and a market that moved on to other themes. On paper, the story should have ended there.
CORT Corcept Therapeutics Inc
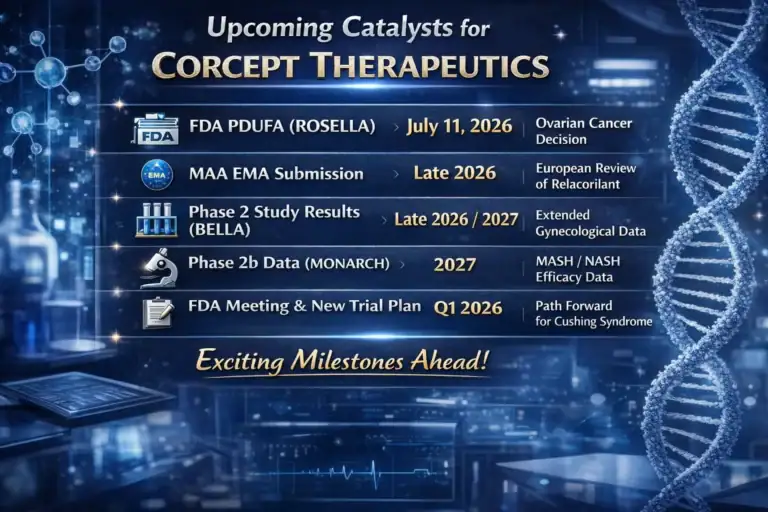
CRL in one paragraph:
The FDA acknowledged that one of Corcept’s Phase 3 trials in Cushing was successful, but another failed its primary endpoint. Given this mixed picture, the agency said it could not reach a favorable benefit-risk assessment without additional evidence of effectiveness and therefore rejected the NDA via CRL. No insurmountable safety issues were cited; the focus is efficacy and consistency.
SLS http://www.sellaslifesciences.com/

News of the day (REGAL): On 29 December 2025, SELLAS reported that the contract research organization running the Phase 3 REGAL trial notified the company that 72 of the 80 required events (deaths) had occurred as of 26 December 2025. REGAL is an overall survival study; the final analysis will be triggered once 80 events are reached. SELLAS remains fully blinded, and this one-time update does not change the statistical plan. Source: SELLAS REGAL update PR (Dec 29, 2025) .
IRWD Ironwood Pharmaceuticals Inc

News of the day: Ironwood maintained FY2025 guidance and issued FY2026 guidance projecting LINZESS U.S. net sales of $1.125–$1.175B, total revenue $450–$475M, and adjusted EBITDA >$300M, citing improved net pricing driven by a January 1, 2026 Linzess list-price reduction. It also reaffirmed that a confirmatory Phase 3 trial for apraglutide is expected to begin in H1 2026, and that the strategic alternatives review is progressing. Source: BusinessWire (Jan 2, 2026).
ONCY Oncolytics Biotech, Inc

Oncolytics Biotech (ONCY) — Pelareorep, GI focus, catalysts into Q1–H1 2026 | Oncolytics Biotech (ONCY) — Pelareorep, focus GI, catalyst Q1–H1 2026 EN IT Oncolytics Biotech (ONCY) Panel-style report (EN/IT): news-first, catalyst timelines, fundamentals/cash runway, pipeline, management/ownership, retail sentiment, risk…
VNDA Vanda Pharmaceuticals Inc

The FDA approved NEREUS (tradipitant) for the prevention of vomiting induced by motion in adults — described as the first new FDA-approved pharmacologic treatment in this area in over 40 years. The company expects a commercial launch “in the coming months.”
DRTS Alpha Tau Medical Ltd Updated jan 8

eep dive on Alpha Tau Medical (DRTS) from a bullish angle: differentiated alpha-radiation platform, multiple late and early-stage trials, solid cash runway and heavy insider ownership. Educational only, not investment advice.
EKSO Ekso Bionics Holdings Inc

On December 29, 2025, Applied Digital (APLD) and Ekso Bionics (EKSO) announced a non-binding term sheet for a proposed business combination between Applied Digital’s cloud computing arm, Applied Digital Cloud, and EKSO.