DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Biotech Deep Dive – Parkinson’s Disease
ACI-7104.056 – Active Immunotherapy in Early Parkinson’s Disease
VacSYn Phase 2 interim data suggest the first credible signal that targeting misfolded α-synuclein with an active vaccine could slow Parkinson’s progression, with robust biomarker effects and a clean safety profile.
Executive Summary
High level view
AC Immune’s ACI-7104.056 is an active peptide vaccine designed to raise antibodies against misfolded α-synuclein in early Parkinson’s disease (PD). The ongoing Phase 2 VacSYn trial is the first randomized, double-blind study to show that such an approach can consistently drive very high antibody titers that reach the CNS and are associated with stabilization of multiple disease-relevant biomarkers.
In Part 1 of VacSYn (n≈34), 100% of treated patients became immunologic responders by week 76, with serum and CSF IgG titers more than 500-fold higher than placebo and tightly correlated across compartments. CSF total α-synuclein and neurofilament light (NfL) remained stable on therapy while declining or rising, respectively, in the placebo group, alongside preserved DaT-SPECT signal and essentially flat MDS-UPDRS Part III motor scores over roughly 18 months.
Safety to date is favorable, with no drug-related serious adverse events and mostly mild, transient injection-site reactions, headache and fatigue. Against a backdrop where multiple passive α-syn antibodies (e.g. Biogen’s cinpanemab) failed to show benefit in Phase 2, VacSYn’s active immunization approach looks differentiated and, at least on biomarkers, disease-modifying.
From a market perspective, PD affects more than 1.1 million people in the US and over 10 million worldwide; nearly 90,000 Americans are newly diagnosed each year, and the annual US economic burden is estimated around 52–61 billion dollars. A safe vaccine that can even modestly slow progression in early PD would likely be a blockbuster-scale opportunity.
Today’s interim Phase 2 update has shifted the ACIU story from a long-shot PD science project to a program with genuine disease-modification potential, but clinical proof of slower disability progression in larger, longer trials remains the key missing piece.
1. Company and Scientific Background
AC Immune SA is a Swiss clinical-stage biotech focused on precision immunotherapy and small molecules for neurodegenerative diseases, particularly Alzheimer’s and Parkinson’s. Its platform has generated programs against misfolded amyloid-β, Tau and α-synuclein, several of which have been partnered historically with large pharma (for example, Eli Lilly and others) while ACI-7104.056 remains wholly owned.
The company has deliberately sharpened its pipeline focus and cut approximately 30% of its workforce to concentrate resources on later-stage assets, while extending its cash runway to at least Q3 2027. That strategic discipline matters because PD development is lengthy and capital-intensive; any credible disease-modifying vaccine must be supported through large, multi-year Phase 3 programs.
ACI-7104.056 builds directly on earlier active α-syn vaccines such as PD01A/PD03A, which demonstrated safety and α-syn-specific antibody responses in Phase 1 but were not developed through to robust randomized Phase 2 experiments. ACI-7104 is best thought of as an optimized second-generation formulation, now being tested with biomarker-driven endpoints and modern PD trial design.
2. Mechanism of Action: Active α-Synuclein Immunotherapy
Pathological aggregation of α-synuclein in neurons and glia is a central feature of Parkinson’s disease and related synucleinopathies. Active immunotherapy aims to train a patient’s immune system to recognize and clear toxic α-syn species, particularly soluble oligomers believed to drive synaptic dysfunction and neurodegeneration.
ACI-7104.056 is a peptide-based vaccine that presents selected α-syn epitopes in a way that elicits high-affinity antibodies against misfolded forms, while minimizing off-target T-cell responses. In preclinical models and early clinical work, such vaccines have generated antibody titers in both plasma and CSF, with evidence that antibodies bind aggregated α-syn and can reduce oligomeric species in the CSF.
Mechanistically, active vaccination differs from monoclonal antibodies (mAbs) in several ways:
- Broader immune engagement: the polyclonal antibody response can recognize multiple conformations/epitopes of α-syn, potentially improving clearance versus a single mAb epitope.
- Pharmacokinetics: once a robust immune memory is established, booster doses can maintain high antibody levels over years with relatively infrequent injections.
- Cost of goods: peptide vaccines are typically cheaper to manufacture and distribute than chronic high-dose IV mAbs, an important consideration in an aging PD population.
These advantages are theoretical until confirmed clinically, but they help explain why ACI-7104.056 is attracting attention after mAb failures in the field.
3. VacSYn Phase 2 Trial Design
VacSYn (NCT06015841) is a prospective, multicenter, randomized, double-blind Phase 2 trial of ACI-7104.056 in early Parkinson’s disease. Key aspects:
- Population: newly diagnosed PD with motor symptoms ≤2 years, Hoehn–Yahr stage I–II, on stable low-dose L-Dopa.
- Design: two seamless parts (up to ~150 patients total). Part 1 enrolls ~34 subjects randomized 3:1 to ACI-7104.056 vs placebo, followed up to 74 weeks; Part 2 is an adaptive expansion that may enroll up to 150 additional patients based on interim biomarker data.
- Dosing: intramuscular injections of the vaccine or placebo, six doses spread over approximately 18 months.
- Core objectives: safety and tolerability, immunogenicity (serum/CSF antibody titers), and pharmacodynamics on α-syn-related biomarkers (CSF α-syn, NfL, DaT-SPECT).
- Exploratory endpoints: MDS-UPDRS motor and non-motor scores, “OFF” L-Dopa motor assessments, digital endpoints, and composite biomarker outcomes.
The design is explicitly biomarker-driven: Part 2 and any subsequent pivotal trials will be guided by the relationship between antibody levels, CSF markers and clinical trajectories observed in Part 1.
4. Interim Clinical and Biomarker Results
4.1 Immunogenicity and CNS Target Engagement
The latest interim analysis from Part 1 shows ACI-7104.056 achieves exactly what an active vaccine is supposed to do:
- 100% responder rate: by week 76 (two weeks after the 6th dose), all treated patients met predefined responder criteria across immunogenicity measures.
- Very high titers: mean serum IgG titers against the immunizing peptide and native α-syn exceeded placebo by more than 500-fold at week 76, with earlier time points already showing ~20-fold increases after four immunizations.
- CSF penetration: CSF IgG levels in the vaccine arm were also ~500-fold above placebo, with strong serum–CSF correlations (Spearman ρ around 0.85–0.92, p<0.05), demonstrating CNS target engagement.
Simply put, ACI-7104.056 reliably generates “boostable” antibodies that reach the brain at levels far above placebo and bind the pathological target.
4.2 Biomarker Trends: Signs of Disease Modification?
Several independent biomarker readouts point in the same direction:
- CSF total α-synuclein: in treated patients, CSF α-syn levels remained stable over the 74-week observation period, whereas placebo patients showed the expected decline, interpreted as increased brain trapping and pathological aggregation (post-hoc p≈0.018).
- CSF neurofilament light (NfL): a marker of axonal damage and neurodegeneration, NfL stayed essentially flat on ACI-7104.056 but increased in the placebo group, again consistent with slowed neuronal injury in the active arm.
- DaT-SPECT: striatal dopamine transporter binding (a proxy for nigrostriatal neuron integrity) was preserved in the vaccine arm compared with decline on placebo, albeit in a small sample.
- Plasma GFAP and peripheral markers: glial fibrillary acidic protein (GFAP) and other peripheral markers showed stabilization trends with ACI-7104 relative to placebo.
While each of these endpoints is exploratory, the overall picture (stable α-syn, stable NfL, stable DaT-SPECT vs deterioration on placebo) is exactly what one would hope to see if a therapy is slowing underlying PD pathology rather than just symptoms.
4.3 Clinical Outcomes
Clinical readouts from Part 1 are still early and not powered for definitive efficacy, but they are directionally encouraging:
- MDS-UPDRS Part III (motor): vaccine-treated patients showed essentially flat motor scores through week 74, whereas placebo patients worsened in line with typical early-PD progression (rising mean UPDRS motor score).
- “OFF” L-Dopa analysis: in assessments performed after withholding L-Dopa, the gap between ACI-7104 and placebo widened further in favor of active therapy, suggesting that the signal is not purely symptomatic.
- Non-motor and composite measures: exploratory non-motor scales and digital endpoints trend towards stabilization but remain too underpowered for strong claims at this stage.
Regulators will ultimately care about clinically meaningful slowing of motor and functional decline. The current data suggest that ACI-7104.056 is at least aligned with that goal, but larger Phase 2 and Phase 3 cohorts will be needed to confirm.
4.4 Safety and Tolerability
Thus far, ACI-7104.056 has a clean safety profile:
- No drug-related serious adverse events reported.
- Most common adverse events are mild, transient injection-site reactions (around half of vaccine-treated patients), headache and fatigue at rates comparable to other peptide vaccines.
- No signal of encephalitis, autoimmune neurologic complications or systemic immune toxicity has emerged to date.
These are interim findings in a relatively small cohort and longer-term safety in larger populations is still untested. But compared to past experience with some Alzheimer’s immunotherapies, the absence of inflammatory CNS events so far is reassuring.
5. Clinical Significance in the Parkinson’s Landscape
Parkinson’s disease remains entirely without an approved disease-modifying therapy. Current standards of care – L-Dopa, dopamine agonists, MAO-B inhibitors, deep brain stimulation and others – treat symptoms but do not alter the underlying neurodegenerative process. Against that backdrop, even a modest, well-documented slowing of motor decline would represent a major shift in practice.
Epidemiology and economic burden underlines the stakes:
- More than 1.1 million people in the US and over 10 million worldwide are estimated to be living with PD.
- Nearly 90,000 new PD diagnoses occur annually in the US, about 50% higher than previous estimates.
- The total annual economic burden in the US alone is roughly 52 billion dollars, rising towards 61.5 billion when updated for inflation.
On that scale, delaying loss of independence by even 6–12 months in early PD – the type of population VacSYn targets – would be clinically and economically meaningful. The alignment between biomarker stability (α-syn, NfL, DaT-SPECT) and early clinical trends in VacSYn is therefore noteworthy.
Importantly, regulators have started to accept biomarker-clinical combinations as supportive evidence in other neurodegenerative diseases (e.g. amyloid-targeting drugs in Alzheimer’s). Whether a similar path emerges for α-syn-targeting vaccines is an open question, but these interim data give AC Immune a credible basis to engage with agencies on adaptive, biomarker-driven development.
6. Competitive and Scientific Context
6.1 ACI-7104.056 vs Earlier Active Vaccines (PD01A / PD03A)
ACI-7104.056 is the successor to AFFiRiS’s PD01A (and related PD03A), early α-syn peptide vaccines that demonstrated:
- Acceptable safety and tolerability in Phase 1.
- Induction of α-syn-specific antibodies in a majority of vaccinated patients.
- Up to ~50% reductions in oligomeric α-syn in CSF in high responders, with largely stable clinical ratings over follow-up, although the trials were small and not powered for efficacy.
PD01A’s signal was intriguing but ultimately too early and under-powered to shift the field. ACI-7104.056 effectively takes the same biological concept into a more rigorous Phase 2 program, with:
- Higher and more consistent antibody titers (100% responder rate, 500-fold titer increases vs placebo).
- Demonstrated robust CSF penetration and biomarker correlations.
- Randomized, double-blind design with prespecified biomarker and clinical endpoints.
6.2 Passive α-Syn Antibodies: Lessons from Cinpanemab
Biogen’s cinpanemab (BIIB054) is the most relevant cautionary tale. In the Phase 2 SPARK trial in early PD, cinpanemab failed to separate from placebo on the primary endpoint (change in MDS-UPDRS total score at week 52) and did not show meaningful benefit on imaging or quality-of-life measures. Biogen discontinued development after a futility-driven interim analysis.
Reviews of α-syn immunotherapy trials have noted that both cinpanemab and other mAbs have so far produced no convincing clinical benefit despite target engagement, raising questions about timing, epitope choice, dosing and the fundamental hypothesis. ACI-7104.056 must therefore do more than just move biomarkers – it has to demonstrate tangible slowing of disability in larger cohorts to change skeptical minds.
6.3 Other Modality Competitors (e.g. Blarcamesine)
Outside α-syn targeting, several neuroprotective or network-modulating approaches are in development:
- Anavex blarcamesine (ANAVEX 2-73): an oral sigma-1 receptor agonist tested in Parkinson’s disease dementia (PDD). Phase 2 studies have reported statistically significant improvements in cognitive and motor scales (including MDS-UPDRS) over 14–48 weeks versus placebo, along with biomarker changes in sigma-1-regulated pathways.
- Other immunotherapies: additional active and passive α-syn programs (e.g. UB-312) have shown CSF antibody responses and reductions in aggregated α-syn, but clinical effects remain exploratory or inconclusive.
Blarcamesine and similar small molecules operate via cellular stress and survival pathways rather than directly clearing α-syn aggregates. If both approaches ultimately succeed, they may be complementary rather than directly competitive.
7. Financial Snapshot and Runway
AC Immune’s latest disclosures indicate:
- Cash, cash equivalents and short-term financial assets of roughly CHF 108–127 million across mid-2025, depending on the quarter referenced.
- Operational streamlining and a 30% workforce reduction aimed at focusing on key late-stage programs and extending cash runway.
- Projected runway into the end of Q3 2027, excluding any additional partnership or milestone income.
For an early-commercial-stage biotech, a roughly two-year plus runway from today offers a reasonable window to:
- Deliver full Part 1 VacSYn data in mid-2026.
- Initiate and begin enrolling Part 2 during 2026.
- Secure potential strategic partnerships or non-dilutive capital tied to positive PD data or progress in other programs (e.g. Alzheimer’s).
However, a full-scale Phase 3 PD program will almost certainly require significant additional funding or a large-pharma partner, and the stock has historically been volatile around data events.
8. Market Opportunity for a PD Vaccine
The potential addressable market for ACI-7104.056 is large:
- Over 1.1 million people living with PD in the US today, expected to reach around 1.2 million by 2030.
- More than 10 million patients worldwide.
- Roughly 90,000 new diagnoses per year in the US, with incidence rising as the population ages.
- A current total US economic burden around 52 billion dollars annually, predicted to increase towards 79 billion by 2037 as prevalence nears 1.6 million patients.
ACI-7104.056 is initially aimed at early-stage PD – the very segment where disease-modifying therapy would logically be deployed and where DaT-SPECT and clinical criteria allow relatively clean enrollment. If efficacy and safety are confirmed, expansion into prodromal PD and other synucleinopathies (e.g. dementia with Lewy bodies, multiple system atrophy) could further enlarge the opportunity.
Pricing assumptions will ultimately depend on magnitude of benefit and duration of treatment (finite vaccine series vs chronic boosters), but any therapy that demonstrably slows progression could justify premium neurodegenerative-class pricing in the US and major markets.
9. Upcoming Catalysts and Development Timeline
| Timing (approx.) | Event | Potential Impact |
|---|---|---|
| Mid-2026 | Full Part 1 VacSYn readout (74-week data) including comprehensive safety, immunogenicity, biomarker and exploratory clinical results. | Key value inflection; will confirm whether interim trends hold and refine effect size estimates. |
| 2026 | Decision and potential launch of Part 2 (up to ~150 additional patients) with refined biomarker and digital endpoints. | De-risking step; shows regulatory confidence in the biomarker-led design and supports eventual Phase 3 strategy. |
| 2026–2027 | Regulatory interactions (FDA/EMA) on potential Breakthrough/Fast Track and Phase 3 endpoint strategy. | Critical for defining whether biomarker-enriched paths or conventional clinical endpoints will be required for approval. |
| Ongoing (2025–2027) | Conference presentations, peer-reviewed publications and KOL commentary on VacSYn data. | Shapes scientific consensus on whether α-syn vaccination is a viable disease-modifying strategy. |
| Post-Part 2 (earliest around 2027–2029) | Potential pivotal Phase 3 study launch in early PD if data justify it; NDA/MAA submissions around decade’s end in an optimistic scenario. | Long-term upside but highly contingent on consistent efficacy and safety across all phases. |
All timelines are approximate and based on current disclosures; they can shift materially with new data, regulatory feedback, or strategic decisions.
10. Scenario Framework (Illustrative, Not Investment Advice)
The ranges below are qualitative scenarios, not price targets or recommendations. They highlight the main drivers that could move ACI-7104.056 closer to or further from eventual approval.
Bull case
Consistent disease-modifying signal
- Full Part 1 readout in 2026 confirms robust biomarker separation and statistically meaningful motor slowing.
- Part 2 replicates clinical benefits in a larger cohort with supportive digital endpoints.
- Regulators accept a biomarker-plus-clinical package as sufficient for accelerated development.
- AC Immune secures a strong global partner, reducing financing risk for Phase 3.
Base case
Biomarker success, modest clinical effect
- Biomarker trends remain clearly positive, but motor/functional differences are modest and require large samples.
- Program progresses into a substantial, conventional Phase 3 trial focused on clinical endpoints.
- Funding comes from a mix of equity and smaller partnerships, with moderate dilution.
- Timeline to any potential filing extends into the early 2030s.
Bear case
Effect fades or safety issues emerge
- Updated data show inconsistent biomarker effects or no clinically meaningful separation from placebo.
- Rare immune-mediated adverse events appear as patient numbers grow.
- Field attention shifts to alternative mechanisms (e.g. GLP-1 agonists, cell/gene therapies).
- Program is discontinued or deprioritized; AC Immune pivots back to other pipeline assets.
11. Key Risks and Red Flags
- Small sample size: Part 1 involves only a few dozen patients. Apparent differences can vanish in larger, more heterogeneous Phase 2/3 cohorts.
- Field track record: prior α-syn immunotherapies, notably Biogen’s cinpanemab, failed to show clinical benefit despite target engagement, and programs were discontinued.
- Regulatory uncertainty: there is no established regulatory precedent for approving a PD vaccine based on biomarker stabilization. Agencies may insist on large, long-duration clinical trials with conventional disability endpoints.
- Safety over time: long-term effects of sustained high anti-α-syn antibody levels in the CNS are unknown. Rare neuroinflammatory or autoimmune events could emerge in larger populations.
- Financing and dilution: even with a runway into Q3 2027, pivotal trials will likely require substantial additional capital or partnering. Negative or ambiguous data could force equity raises at unfavorable valuations.
- Competitive evolution: other modalities (e.g. GLP-1 agonists, neuroprotective small molecules, cell or gene therapies) may deliver more compelling clinical effects and capture the “disease-modifying” narrative if ACI-7104.056 stalls.
12. Sentiment Snapshot (Retail and Analysts)
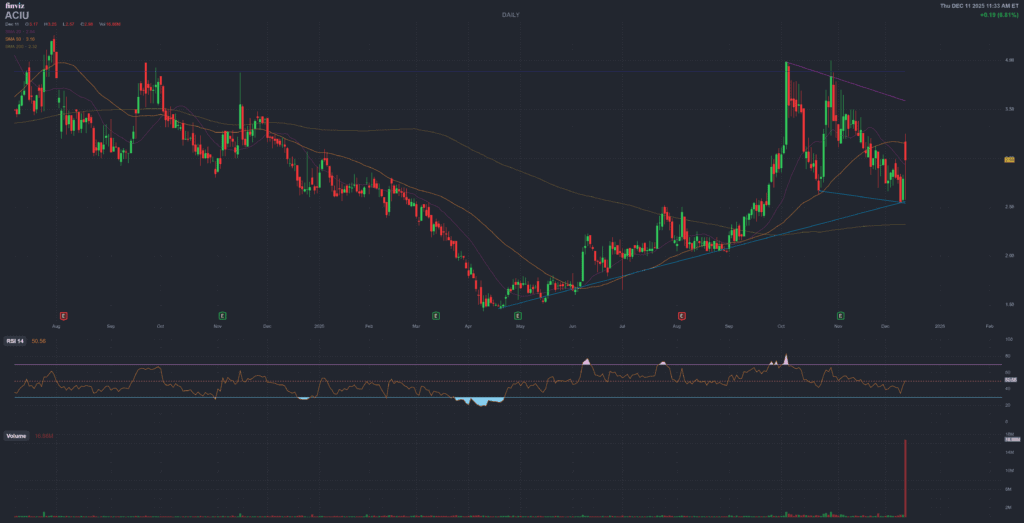
Immediately after the December 11, 2025 interim Phase 2 update, ACIU shares jumped more than 20% in pre-market trading and traded near the 3 USD level intraday, with market cap in the low-300 million USD range. Stocktwits data show message volume spiking into “extremely high” territory and sentiment flipping from bearish to strongly bullish over 24 hours as retail traders reacted to the “first signals” of disease modification.
On the sell-side, a small analyst pool currently rates AC Immune as a “Strong Buy,” with an average 12-month price target near 10 USD and a range roughly 8–12 USD. These are independent third-party opinions and should not be taken as recommendations here; they do, however, illustrate how sensitive valuation is to perceived probabilities of success in VacSYn.
Outside of these pockets, coverage on Reddit and X (Twitter) remains relatively light but is trending more positive as news spreads through neurology and biotech circles. This is typical for a small-cap neuro company that has just produced a high-impact readout.
Sentiment can reverse quickly if future data are weaker than expected or if the broader market sells off. It should be viewed as a short-term indicator of crowd positioning, not as a fundamental thesis.
13. Tools, Data Sources and Research Partners
Research and Screening Tools
- Finviz – charting, fundamentals and sector comparison for ACIU and PD peers.
- ChartsWatcher – real-time scanner; ChartsWatcher is a real-time, next-generation scanner for the US stock market.
- Seeking Alpha – earnings call transcripts, analyst commentary and factor grades.
- Stocktwits – live retail sentiment and message flow around ACIU.
- Medved Trader – professional-grade trading and charting platform used alongside the research in this report.
- Merlintrader trading Blog – home base for this and other deep-dive biotech reports.
Some of the links above are affiliate or referral links. If you choose to register or subscribe through them, they may generate a commission for Merlintrader at no additional cost to you. This does not influence the data used or the conclusions drawn in this report.
Support Merlintrader trading Blog
All reports on Merlintrader are based on primary sources, SEC filings and official trial data, and remain free to read. If this analysis on AC Immune and ACI-7104.056 helped you, you can support future work with a small one-time donation.
 Donate via PayPal
Donate via PayPal
14. Sources and Further Reading
This report is based exclusively on primary and high-quality sources (company filings and press releases, peer-reviewed articles, major medical/neurology journals and leading PD foundations). A non-exhaustive list:
- AC Immune SA – investor presentations, Q2/Q3 2025 financial results and VacSYn interim updates.
- December 11, 2025 AC Immune press release and associated news flow on interim Phase 2 data for ACI-7104.056.
- Peer-reviewed and review articles on α-syn immunotherapy, including trials of PD01A/PD03A and monoclonal antibodies.
- Parkinson’s Foundation – epidemiology, incidence, prevalence and economic burden studies.
- Michael J. Fox Foundation and academic cost-of-illness analyses for PD.
- Published Phase 2 data and company communications from Anavex on blarcamesine in PDD.
- Public market data (NASDAQ, Yahoo Finance, Stockanalysis, Stocktwits, Investing.com) for ACIU price history and market capitalization.
Important Disclaimer – Not Investment Advice
This report is provided strictly for educational and informational purposes. It is not, and must not be interpreted as, an offer, solicitation, recommendation or invitation to buy or sell any financial instrument, nor as individualized investment advice. The analysis reflects the personal views of the author at the time of writing, based on publicly available information, and may change without notice.
Although the data and sources used (including SEC filings, official company communications, clinical trial registries and reputable medical organizations) have been cross-checked where possible, errors or omissions can still occur. Clinical development outcomes are inherently uncertain; past or interim results do not guarantee future performance. Any forward-looking statements, scenarios or probabilities discussed here are illustrative only and may turn out to be incorrect.
Readers should perform their own independent research, consider their individual financial situation and risk tolerance, and, where appropriate, consult a licensed financial advisor before making any investment decisions. Trading and investing in biotech and small-cap equities involves substantial risk, including the possible loss of the entire capital invested.
This content is not produced by, nor does it represent the views of, any broker-dealer, asset manager or regulated financial institution. It is not intended to satisfy the regulatory requirements for investment research in any jurisdiction.
For full legal information, risk warnings and privacy terms, please refer to: https://merlintrader.com/disclaimer/ and https://merlintrader.com/condizioni-duso-e-info-privacy/.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


