DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Biotech Deep Dive – Neurodegeneration & Rare Disease
Denali Therapeutics (DNLI) – DNL310 PDUFA April 5, 2026
Data review and scenario analysis for Denali Therapeutics ahead of the accelerated approval decision on tividenofusp alfa (DNL310) in Hunter syndrome (MPS II).
Overall stance: data-driven neutral with positive bias (not a recommendation)
DNL310: strong clinical data, Priority Review, PDUFA 5 Apr 2026
High burn rate, dilution, dependence on a single lead asset
Quick data
Market snapshot – denominated in USD
Last closing price
$19.66
9 Dec 2025 – regular session
After-hours move
$17.75 (-9.72%)
Post-offering reaction
Market cap
$2.88B
Small/mid-cap biotech
52-week range
$10.57 – $26.18
Price near upper part of the range
30-day volatility (annualised)
≈ 64%
Very high for most portfolios
RSI (approx.)
High 60s
Close to overbought before the offering
Strong TV platform and late-stage rare-disease asset
Momentum positive but strongly event-driven
Post-offering pressure and dilution risk
Executive summary
Where Denali stands going into 2026
Denali Therapeutics is a late-stage biotech focused on neurodegenerative and lysosomal storage diseases, with a proprietary Transport Vehicle (TV) platform to move biologics across the blood–brain barrier. The investment case in the next 12–18 months is dominated by the accelerated approval decision for DNL310 (tividenofusp alfa) in Hunter syndrome (MPS II), alongside the company’s ability to fund and execute a first commercial launch while advancing a broader pipeline.
The picture is intentionally presented here in a balanced way:
- Positive elements: strong biomarker-driven DNL310 data with sustained reduction of CSF heparan sulfate, multiple regulatory designations, a PDUFA date of 5 April 2026, and a 275M USD synthetic royalty funding agreement with Royalty Pharma that validates the asset and the TV platform if approval is achieved.
- Areas of attention: a very high burn rate (around 500M USD per year), a pre-revenue profile, and a new 200M USD public offering announced shortly after the Royalty Pharma deal, which extends runway but dilutes existing shareholders.
- Key vulnerabilities: strong dependence on one main near-term catalyst, with past setbacks in other programmes (such as DNL343 in ALS), and the structural volatility typical of small/mid-cap biotech.
The external analyst community is broadly constructive on DNLI with a consensus “Buy” / “Strong Buy” stance and a 12-month target range typically around the high-20s to low-30s in USD. Those targets and all scenario ranges mentioned here should be seen as illustrative expectations based on current public information, not as promises or personalised advice.
DNL310 and DNL126: platform-defining rare-disease assets
Royalty deal improves funding quality, equity offering adds dilution
Binary regulatory event and high cash burn remain central risks
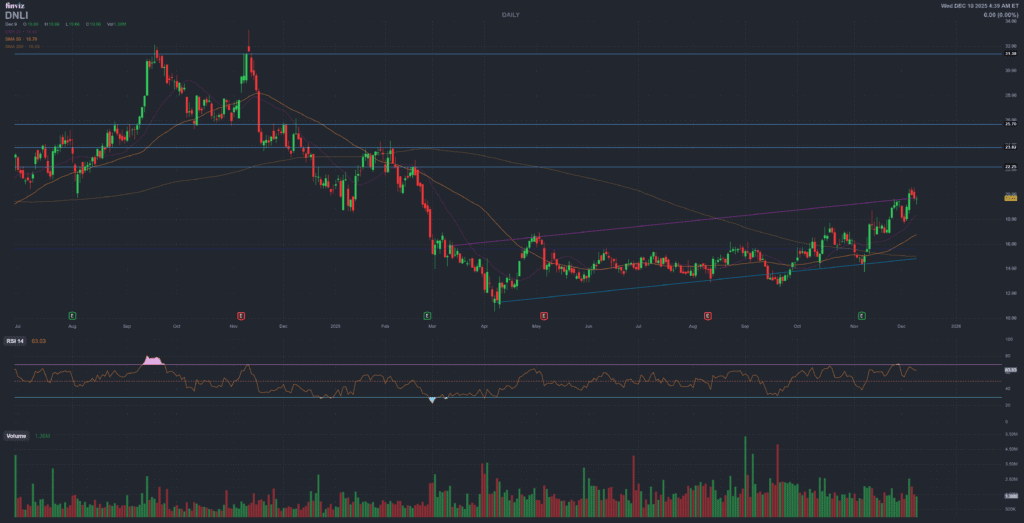
Price action
Recent performance into the financing
Before the latest offering, DNLI had already staged a strong recovery from the 52-week low, with notable momentum in the last one to three months. The post-offering after-hours drop shows how sensitive the stock remains to financing events.
| Period | Performance | Comment |
|---|---|---|
| 1 week | +3.5% approx. | Short-term trend positive into news flow |
| 1 month | +26.0% approx. | Strong rally, typical of pre-catalyst positioning |
| 3 months | +32.7% approx. | Robust medium-term momentum |
| 6 months | +29.4% approx. | Uptrend from mid-2025 lows |
| 1 year | Around -21% | Still below prior peaks despite recent strength |
With the price recently close to the 52-week high, technical indicators such as RSI were in a stretched zone before the offering. The new shares and warrants reset the near-term technical picture and can change where active traders look for support (for example, around prior private placement levels).
Financial snapshot – Q3 2025
SEC-filed data, summarised
Income statement – last four quarters
| Quarter | Revenue | R&D | Operating expenses | Net loss | Diluted EPS |
|---|---|---|---|---|---|
| Q3 2025 | $0 | $102.0M | $137.4M | -$126.9M | -$0.74 |
| Q2 2025 | $0 | $102.7M | $131.9M | -$124.1M | -$0.72 |
| Q1 2025 | $0 | $116.2M | $145.6M | -$133.0M | -$0.78 |
| Q4 2024 | $0 | $99.8M | $129.8M | -$114.8M | -$0.67 |
Balance sheet highlights (Q3 2025)
Cash & marketable securities
$872.9M
As of 30 Sep 2025
Total assets
$1.06B approx.
Asset base built around R&D
Stockholders’ equity
$926M approx.
Healthy equity cushion
Accumulated deficit
-$1.9B approx.
Typical for long R&D history
A simple way to think about the funding profile is:
- Quarterly burn around 125M USD.
- Annualised burn roughly 500M USD, before any commercial build-out.
- Runway around 1.7 years from Q3 2025 cash alone, extended by the 200M USD offering and potential 200M USD from Royalty Pharma upon FDA approval.
These numbers are rounded and based on SEC-filed financials; they are not forecasts.
Pipeline and key catalysts
TV platform across rare disease and neurodegeneration
1. DNL310 (tividenofusp alfa) – Hunter syndrome (MPS II)
- Status: BLA under U.S. FDA Priority Review for accelerated approval.
- PDUFA date: 5 April 2026 (extended from an earlier 2026 date).
- Indication: Mucopolysaccharidosis type II (Hunter syndrome).
- Mechanism: TV-enabled enzyme replacement therapy designed to cross the blood–brain barrier and address both somatic and CNS manifestations.
- Regulatory designations: Fast Track, Breakthrough Therapy, Rare Pediatric Disease, with additional support programmes in the EU.
- Clinical data: Deep and sustained reduction of CSF heparan sulfate with durable responses beyond 100 weeks in extension data, plus improvement in key clinical measures for many patients.
From a purely data-driven perspective, DNL310 looks like a high-impact, high-probability catalyst, but the outcome still depends on the final FDA view on the totality of data, including safety and the surrogate endpoint strategy.
2. DNL126 (ETV:SGSH) – Sanfilippo syndrome type A
- Status: Phase 1/2 ongoing, with alignment on an accelerated-approval pathway and participation in FDA’s START programme.
- Designations: Orphan Drug, Fast Track, Rare Pediatric Disease.
- Data: Strong reduction in CSF heparan sulfate, including normalisation in many cases, suggesting a similar mechanistic story to DNL310.
- Timeline: A BLA around 2026–2027 is plausible if data and regulatory feedback remain favourable.
3. BIIB122 / DNL151 – LRRK2 inhibitor for Parkinson’s disease
- Status: Phase 2b LUMA study with Biogen; enrolment completed in 2025.
- Positioning: Genetically validated target, but Parkinson’s is a highly competitive and complex field with many development efforts underway.
- Risk profile: The readout expected around 2026 could expand the story beyond rare disease or highlight limits of the approach in common neurodegeneration.
4. DNL593, DNL628, DNL952 and other TV assets
- DNL593 (PTV:PGRN) for FTD-GRN in partnership with Takeda.
- DNL628 (OTV:MAPT) for tau-related disease; CTA submitted in 2025.
- DNL952 (ETV:GAA) for Pompe disease; IND submitted with some additional preclinical requests from FDA.
- DNL343 in ALS was discontinued after missing endpoints, a reminder that the platform is promising but not risk-free.
Overall, Denali’s pipeline offers multiple shots on goal, but over the next 12–18 months the market will mainly focus on DNL310, DNL126 progression, and the Parkinson and neurodegeneration readouts.
Deals and funding
Royalty, equity, and past placements
1. 275M USD royalty funding with Royalty Pharma
- Date: Announced 4 December 2025.
- Structure: 200M USD upfront at FDA accelerated approval of DNL310 in Hunter syndrome; 75M USD upon EMA approval within a specified time window.
- Royalty terms: 9.25% royalty on worldwide net sales of tividenofusp alfa, with a cap of approximately 3.0x on invested capital (reduced to 2.5x if reached by early 2039).
- Control: Denali retains full development and commercial rights.
This deal improves the quality of future funding (royalty-based rather than purely equity) but is contingent on regulatory success.
2. 200M USD underwritten public offering
- Date: Announced 9 December 2025, shortly after the Royalty Pharma agreement.
- Structure: Common shares plus pre-funded warrants, with an additional 30M USD greenshoe option.
- Underwriters: Major investment banks including Goldman Sachs, J.P. Morgan, Morgan Stanley, and Jefferies.
- Market reaction: Around -9.7% in after-hours trading, consistent with investor concern about dilution after a strong run-up.
3. 2024 private placement (~500M USD)
- Size: Roughly 500M USD net proceeds from a February 2024 transaction.
- Terms: Combination of common shares and a large block of pre-funded warrants.
The sequence of financings shows a deliberate strategy to secure a robust war chest ahead of a potential 2026 launch. The price for this is cumulative dilution for existing shareholders.
Analyst view and sentiment
Wall Street, market tape, and retail mood
Sell-side consensus
- Coverage: Roughly mid-teens number of analysts, a relatively deep coverage universe for a company of this size.
- Rating mix: All currently in the Buy / Strong Buy area, with no active Sell ratings reported at the time of this review.
- 12-month price targets: Clustered broadly in the 25–35 USD range, with outliers on both sides; this implies upside versus the 19–20 USD spot level used in this report, but those are external forecasts, not guarantees.
- Recent actions: A mix of target raises and modest trims, typical of a name that has re-rated but still has a pending binary event.
Market and retail sentiment
- Price action: the strong move off the lows, plus the rapid reaction to the offering, suggests an investor base that is actively trading catalysts and balance-sheet news.
- Social / retail tone: overall cautious-positive, with interest centred on the DNL310 opportunity and the Royalty Pharma validation, but recurring concern about dilution and cash burn.
Scenario analysis
Illustrative views, not price targets or advice
The ranges below translate the qualitative discussion into three simplified scenarios. They are not recommendations; they simply illustrate how the same facts can lead to different outcomes depending on how the key variables play out.
Bull case – platform success
Illustrative probability: around 40%
- DNL310 receives timely approval in Hunter syndrome with a usable label.
- DNL126 progresses smoothly towards registration with additional data in 2026–2027.
- Launch metrics, payer feedback, and early sales support a credible path to several hundred million USD in peak sales for DNL310.
- Pipeline news in Parkinson and other TV assets reinforces the broader platform.
- In this setup, it is possible to justify valuations that, in rough terms, map to the mid-30s or higher per share over time, depending on assumptions.
Base case – approval with execution questions
Illustrative probability: around 45%
- DNL310 is approved but with some label or commercial constraints that moderate the peak-sales opportunity.
- Launch is steady but not explosive; the company still needs to manage costs tightly in the first commercial years.
- The market gradually shifts to focus on DNL126 and neurodegeneration readouts, with mixed but broadly supportive data.
- In this situation, a valuation corridor in roughly the mid-20s to low-30s appears reasonable as a working assumption rather than a target.
Bear case – regulatory setback
Illustrative probability: around 15%
- DNL310 experiences a delay, a restrictive label, or a Complete Response Letter that pushes meaningful revenue beyond 2026.
- Additional capital is required sooner than expected, at less favourable terms.
- Confidence in the broader TV pipeline is reduced until more data arrive.
- In this downside case, it is plausible to see price levels revisiting a low- to mid-teens area, at least temporarily, before the market can re-underwrite the story on new information.
These probabilities and ranges are purely illustrative and based on public information and typical biotech patterns. They are not personalised investment advice and are not a forecast in the formal sense.
Timeline – events to watch (2025–2027)
Dates and periods are approximate and subject to change
December 2025
Closing of the 200M USD public offering
Short-term focus on pricing, final size, and resulting float.
Q1 2026
Execution updates and preparation for DNL310 launch
Investors typically watch for commentary on salesforce build-out, payer work, and
manufacturing readiness.
5 April 2026
PDUFA action date – DNL310 in Hunter syndrome (MPS II)
Binary catalyst with potential double-digit percentage moves in either direction over
a short period.
H1 2026 – 2027
LUMA Parkinson study readout and DNL126 progress
These events will influence whether the TV platform is seen as a focused rare-disease
story or as a broader CNS franchise.
Sources and data quality
Primary documents only, cross-checked
This report is based exclusively on primary filings and high-quality financial news outlets, including:
- Denali Therapeutics Q3 2025 financial results and business highlights (press release and 10-Q).
- Denali Therapeutics and Royalty Pharma 275M USD royalty funding agreement press release.
- Recent Denali investor presentations and publicly available pipeline updates.
- Regulatory information from FDA and EMA communications where available.
- Price, volume, and 52-week range data from reputable market data providers.
All figures are as of the dates indicated and can change as new filings and press releases are published. Readers are encouraged to verify the latest data directly from the original sources.
Important legal and risk disclaimer
This report is provided solely for informational and educational purposes and does not constitute, and must not be interpreted as, personalised investment advice, a solicitation, or a recommendation to buy, sell, or hold any security or financial instrument. The analysis is based on public information from company filings, official press releases, regulatory documents and reputable data providers available at the time of writing. Data, estimates and examples may become inaccurate as new information is released.
The type of securities discussed here can be highly volatile and speculative. It is possible to lose a substantial portion or the entirety of the capital invested. Readers remain solely responsible for their own decisions. Before making any investment decision, consult a qualified financial advisor and carefully evaluate whether such decisions are appropriate in light of your objectives, financial situation and risk tolerance.
This text is written with attention to general principles of transparency and fairness consistent with the spirit of regulations applied by authorities such as CONSOB in Italy and the SEC in the United States, but it is not a legal document and does not replace the official rules of any regulator.
For full legal notices and privacy information, please refer to the official pages: Disclaimer and Terms of use & privacy info.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.



