DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Single-Name Deep Dive – AI Drug Discovery
Recursion Pharmaceuticals (RXRX) – AI-Driven Drug Discovery At An Inflection Point
Speculative biotech with real research, first clinical validation of its AI platform in FAP, and a fresh J.P. Morgan upgrade. Outlook: fundamentally bullish, risk profile high.
News of the day – Analyst upgrade
J.P. Morgan upgrades RXRX to Overweight, raises target to 11 USD
December 17, 2025
J.P. Morgan upgraded Recursion Pharmaceuticals (RXRX) from Neutral to Overweight and increased its price target from 10 to 11 USD after reviewing new Phase 1b/2 data for REC-4881 in familial adenomatous polyposis (FAP). At a spot price around 4.20 USD, the new target implies more than 160 percent upside versus the current quote. The upgrade is based on:
- Strong and durable reductions in polyp burden observed in the TUPELO trial.
- A broader-than-expected addressable population in FAP and related settings.
- Blockbuster-level sales potential for REC-4881 if it reaches the market.
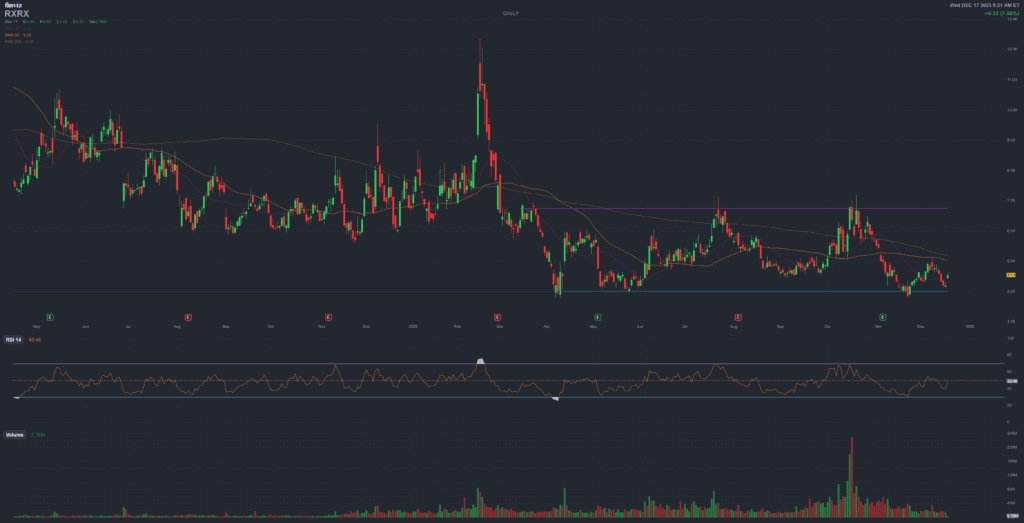
RXRX – Daily chart (Finviz)
As of December 17, 2025, RXRX trades around 4.20 USD, closer to the lower end of its 52-week range than to the prior 12.36 USD high. Technically, the stock sits in a speculative accumulation zone, with significant upside implied by the latest analyst targets but still inside a volatile, high-beta pattern.
Executive summary
Bullish outlook
High risk
Recursion Pharmaceuticals is a clinical-stage “TechBio” company that uses automation, high-throughput biology, and artificial intelligence to discover and optimise drug candidates. Its core platform, the Recursion OS, screens cellular phenotypes at massive scale to map biology and chemistry and propose new therapeutic hypotheses.
The company has just delivered the first clear clinical validation of this approach with REC-4881 in familial adenomatous polyposis (FAP), a rare cancer-predisposition syndrome with no approved pharmacological treatments today. After 12 weeks of treatment, patients on REC-4881 showed a median 43 percent reduction in total polyp burden, and 82 percent maintained reductions 12 weeks after stopping therapy, with a median 53 percent decrease from baseline. Safety was consistent with the MEK inhibitor class, without high-grade unexpected signals.
On top of this data, RXRX keeps a strong cash position (roughly 785 million USD in cash and equivalents after recent financings) and multiple big-pharma collaborations (Roche/Genentech, Sanofi and others) that together could deliver several billions in milestones if the partnered pipelines move forward. The key message for investors is straightforward: this is no longer just a story about AI slides and datasets; it is a story with a first working example in patients, but still early and speculative.
Short version: real research, real trials, real partnerships. No products yet, heavy cash burn, but a clear first proof of concept and an asymmetric upside profile for investors willing to hold speculative biotech risk.
Company overview – From lab automation to “TechBio” platform
Recursion was founded in 2013 by Chris Gibson, Ph.D., during his MD/PhD training at the University of Utah, together with mentors and co-founders at the intersection of biology, data science and engineering. From the start, the idea was to industrialise cell-based experimentation: use automated microscopes, robotics and cloud compute to generate a huge image-based dataset of how cells respond to drugs, genetic edits and other perturbations, then train AI models to read that dataset at scale.
Over roughly a decade, Recursion has:
- Built one of the largest proprietary phenotypic screening datasets in pharma, with millions of microscopy images capturing cellular responses.
- Invested heavily in computing infrastructure, including high-end NVIDIA hardware, to run deep-learning models for representation learning and target discovery.
- Expanded into a multi-site operation in the US, Canada and Europe, hiring a mixed team of biologists, chemists, data scientists and engineers.
- Converted parts of this platform into concrete clinical programs and external collaborations with large pharma players.
The stated mission is to “decode biology to radically improve lives”. For investors, the practical question is whether this infrastructure can consistently translate into drugs that get approved and generate meaningful revenue. REC-4881 in FAP is the first serious datapoint in that direction.
Platform, partnerships and pipeline
Why big pharma and big tech are already at the table
Strategic pharma partnerships
Recursion’s business model is not limited to its own internal assets. The Recursion OS is positioned as a discovery engine that can feed both proprietary and partnered pipelines. Two anchor collaborations stand out:
- Roche / Genentech collaboration (2021–): up to 40 programs in neuroscience and oncology. Roche pays Recursion for mapping disease-relevant biology and nominating candidates, with billions in potential milestones and royalties if drugs reach the market.
- Sanofi collaboration (2022–): up to 15 programs in oncology and immunology, anchored by a 100 million USD upfront payment and up to 5.2 billion USD in milestones across the portfolio.
These deals both validate the technology and provide non-dilutive capital. They also give Recursion access to big-pharma development and commercial infrastructure for some of the resulting drugs.
Internal clinical pipeline (selected)
REC-4881 – MEK1/2 inhibitor for FAP (lead asset)
- In-licensed from Takeda as a shelved oncology compound and repurposed after Recursion’s AI models linked MEK1/2 inhibition to APC loss biology in FAP.
- Phase 1b/2 TUPELO trial in FAP patients has reported positive data (see next section).
- Potential first pharmaco-therapy for FAP, a setting with no approved drugs and a high lifetime CRC risk.
Other programs
- REC-617 (CDK7 inhibitor) – early-stage trial in platinum-resistant ovarian cancer and other solid tumours, with early signs of anti-tumour activity.
- REC-1245, REC-3565 – oncology programs in Phase I/II with data expected 2026–2027.
- REC-102 for hypophosphatasia – oral candidate developed via a joint venture, targeting a rare metabolic bone disease.
Not every early program has worked. In 2025, Recursion discontinued several first-generation assets, including REC-994 (for cerebral cavernous malformation), as part of a pipeline reprioritisation. That pruning is normal in biotech, but also a reminder that the AI layer does not remove biological risk: it tries to improve the odds, not eliminate failure.
Clinical focus – REC-4881 in familial adenomatous polyposis (FAP)
First clear clinical validation of the Recursion OS
Disease background
Familial adenomatous polyposis (FAP) is a rare, inherited syndrome driven by pathogenic variants in the APC gene. Patients develop hundreds to thousands of adenomatous polyps in the gastrointestinal tract and face a near-certain risk of colorectal cancer if left untreated. Current management revolves around surveillance, endoscopic polypectomies and major surgeries (often colectomy), with no approved drug therapy specifically indicated for FAP.
Key TUPELO Phase 1b/2 results
- Design: multicentre, dose-escalation and expansion study of REC-4881 (4 mg once-daily) in adult FAP patients, evaluating polyp burden reductions and safety.
- Efficacy at Week 12: 75 percent of evaluable patients showed reductions in total polyp burden, with a 43 percent median reduction in total polyp count from baseline.
- Durability at Week 25 (12 weeks after stopping therapy): 82 percent of evaluable patients maintained a reduction in polyp burden, with a 53 percent median reduction relative to baseline.
- Upper GI benefit: approximately 40 percent of patients experienced a one-stage or greater improvement in Spigelman stage, suggesting meaningful disease modification in duodenal polyposis.
- Safety: adverse events were consistent with MEK-inhibitor class effects. Most were Grade 1–2; around 16 percent of patients experienced Grade 3 events. No Grade 4 or higher treatment-related events were reported in the presented dataset.
Compared with natural-history cohorts where the vast majority of FAP patients show progressive polyp growth over a year, these reductions and their durability after treatment cessation are clinically significant. For Recursion, the more important point is that REC-4881 is not a random MEK inhibitor tossed into FAP; it is the output of an AI-driven phenotypic insight linking MEK1/2 modulation to APC-loss biology. That closes the loop from platform prediction to human proof.
Regulatory path and next steps
Following these results, Recursion plans to engage the FDA in the first half of 2026 to discuss a potential registration pathway for REC-4881 in FAP. Management has indicated:
- An intention to expand the eligible population by lowering the age threshold from 55 years to 18 years in upcoming cohorts.
- An exploration of alternative dosing schedules and combination approaches, aiming to preserve efficacy while further optimising tolerability.
- Openness to designing a pivotal trial that could support approval in the highest-risk FAP subgroups if the agency agrees on endpoints and design.
J.P. Morgan models more than one billion USD of potential peak annual sales in the US alone for REC-4881 if developed successfully in FAP and related indications. That makes this program one of the most important value drivers in the Recursion story.
Financial profile – Strong cash, heavy burn
Recursion is still pre-revenue from a product perspective, but it does generate collaboration revenue and occasional milestones:
- Q3 2025 revenue: roughly 5 million USD, well below prior analyst expectations around 17 million USD, underlining the volatility of milestone-based income.
- Q3 2025 loss per share: approximately 0.36 USD, slightly better than consensus but still reflecting a large operating loss.
- Operating expenses: R&D hovering around 120 million USD for the quarter, G&A over 40 million USD, illustrating the cost of running both an AI platform and multiple clinical studies.
- Cash and equivalents: about 785 million USD after recent financings and milestone receipts, bolstered by an at-the-market equity program in 2025.
- Runway: management repeatedly guides for a cash runway extending through 2027, assuming current plans and expected collaboration inflows.
The financial picture is typical of an ambitious platform biotech: strong balance sheet today, meaningful but manageable dilution behind, and a clear dependency on future clinical and business execution to avoid further heavy dilution later in the decade.
Sentiment – Retail, social and institutional
What non-professional traders and analysts are actually saying
Retail and social sentiment (Reddit, Stocktwits, X)
- On retail forums (Reddit communities focused on growth and speculative names), RXRX is framed as a classic “AI plus biotech” bet. Enthusiastic posts describe Recursion as a potential “AWS of biotech” or a future platform that could license out its discovery engine across the industry. The tone is often strongly bullish but explicitly speculative.
- There is also a more cautious camp: threads highlight the high price-to-sales multiple, lack of approved products and the fact that this is still essentially “a bet” on management and the platform, not on current numbers.
- On Stocktwits, sentiment oscillated over 2025: periods of bearishness around earnings and dilution, followed by spikes in optimism when AI partnerships or the FAP data hit the tape. Daily sentiment recently shifted more positive after the REC-4881 TUPELO data, but the message stream is still split between optimists and sceptics.
- On X, most discussion centres on the FAP data and the broader theme of AI-enabled drug discovery. Quantitative sentiment tools classify recent news flow as clearly positive, but again, this reflects news headlines, not a guarantee of stock performance.
Note: all these are comments from non-professional traders and investors. They can be useful to gauge attention and crowd mood, but they are not research and should never be treated as a reliable basis for financial decisions.
Analyst and institutional sentiment
- Street coverage remains relatively limited. Consensus 12-month target is in the mid-single digits, with ratings clustered around Hold, a handful of Buy ratings and some cautious equal-weight views.
- The fresh J.P. Morgan upgrade to Overweight with an 11 USD target is one of the clearest institutional votes of confidence so far and could attract incremental fundamental interest if additional positive data arrives.
- Leading AI-thematic investor ARK Invest holds a significant stake (around seven percent of shares outstanding across its ETFs), signalling long-term conviction in the TechBio narrative despite volatility.
- Short interest remains elevated, reflecting a sizeable group of investors betting against Recursion’s ability to convert AI buzz into durable value. This keeps the stock in a high-beta, high-volatility regime.
Bull case vs. bear case
Bull case – Why bulls like RXRX here
- First real proof of concept: REC-4881 in FAP shows that Recursion’s AI-derived biological insights can translate into meaningful clinical benefit in a disease with no approved drugs.
- Lead asset with blockbuster potential: FAP and related indications could support more than one billion USD in peak sales if development stays on track and pricing is favourable.
- Platform leverage: The same Recursion OS that discovered the MEK-FAP link can, in theory, generate multiple new programs, creating a drug “factory” instead of a single-product story.
- Big-pharma and big-tech validation: Long-term partnerships with Roche, Sanofi, NVIDIA and others signal that sophisticated counterparties see value in the platform and are willing to pay for it.
- Cash runway through 2027: With a strong balance sheet today, Recursion can move REC-4881 and other programs through several key milestones without immediate pressure to raise capital.
- Asymmetric setup at current price: Trading near the low end of its 52-week range, the stock offers considerable upside to both Street and J.P. Morgan targets if even part of the bull thesis materialises.
Bear case – Where the story can go wrong
- No approved products: All value is in pipeline potential. FAP data comes from a small study; larger, longer trials could dilute or negate the apparent benefit.
- Heavy cash burn: The model requires sustained high spending. If milestones arrive later than expected or trials expand faster, Recursion may still need new dilutive capital before reaching profitability.
- Execution risk: Moving from early-stage trials to pivotal studies, manufacturing and commercial planning is a leap many biotechs struggle with, especially when led by a relatively new leadership team.
- Competitive and narrative risk: AI drug discovery is a crowded field. If other platforms deliver approvals sooner, or if the market sours on the theme, multiple expansion for RXRX could be limited even with good science.
- Volatility and crowd dynamics: Past trading shows meme-style surges and sharp drawdowns. High short interest and speculative flows can amplify moves in both directions, making timing difficult.
Bottom line from a fundamental perspective: Recursion is not a promotional shell; it is a real R&D-heavy TechBio company with a validated AI platform, at least one clinically meaningful program in FAP and serious partners already on board. At current levels, the stock is firmly in speculative territory but with a credible path to value creation if clinical and regulatory execution on REC-4881 and the broader pipeline does not break.
Key catalysts – 2026 and beyond
- 1H 2026: FDA interaction on REC-4881 FAP registration pathway (study design, endpoints, potential expedited paths).
- 2026: Additional TUPELO data updates, including younger cohorts (down to 18 years) and potentially longer follow-up on durability and safety.
- 2026–2027: Early-stage data from REC-617 and other oncology programs (REC-1245, REC-3565), which will test whether the platform can deliver multiple clinically meaningful assets.
- Ongoing: Milestone announcements under Roche and Sanofi collaborations as new AI-derived programs advance or hit data delivery checkpoints.
- Structural: Leadership transition on January 1, 2026, with Najat Khan stepping in as CEO and Chris Gibson moving to Chair of the Board, and how this translates into execution discipline and capital allocation.
Recursion Pharmaceuticals (RXRX) – Sintesi in italiano
Recursion Pharmaceuticals è una società “TechBio” in fase clinica che unisce biologia ad alto throughput, automazione di laboratorio e intelligenza artificiale per scoprire nuovi farmaci. Il cuore della piattaforma, chiamata Recursion OS, è un enorme sistema di screening fenotipico che osserva come le cellule reagiscono a migliaia di perturbazioni e usa modelli di machine learning per identificare target e molecole promettenti.
Nel dicembre 2025 l’azienda ha comunicato risultati positivi dallo studio TUPELO di REC-4881 nella poliposi adenomatosa familiare (FAP), una malattia genetica rara ad altissimo rischio di tumore colorettale per cui oggi non esiste alcuna terapia farmacologica approvata. Dopo 12 settimane di trattamento, i pazienti hanno mostrato una riduzione mediana del 43 percento del carico di polipi e, a 25 settimane (12 dopo la sospensione), l’82 percento manteneva una riduzione con una mediana del 53 percento rispetto al basale. La sicurezza è risultata in linea con la classe dei MEK-inibitori.
Sulla base di questi dati, J.P. Morgan ha alzato il rating su RXRX da Neutral a Overweight e il target price da 10 a 11 dollari. Con un prezzo in area 4,20 dollari, il potenziale teorico di rialzo è molto ampio, pur all’interno di un profilo decisamente speculativo.
Recursion dispone di:
- Partnership di peso con Roche/Genentech e Sanofi, con miliardi di dollari potenziali in milestone.
- Una pipeline interna che, oltre a REC-4881, include programmi in oncologia (REC-617, REC-1245, REC-3565) e malattie rare (REC-102 per ipofosfatasia).
- Una cassa vicina agli 800 milioni di dollari e una guidance di runway fino al 2027.
Lato sentiment, il titolo è molto amato dai trader focalizzati su AI e biotech ma rimane volatile, con forte componente retail, short interest elevato e forti oscillazioni legate a news flow e narrativa. Per gli investitori, RXRX è chiaramente una scommessa high-beta: non è una truffa, è ricerca vera, ma è ancora lontana dall’avere un farmaco approvato.
In sintesi:
- Tesi positiva: piattaforma AI credibile, primo proof-of-concept clinico forte in FAP, potenziale blockbuster per REC-4881, partnership solide, cassa robusta.
- Rischi: nessun prodotto sul mercato, studi ancora su numeri piccoli, burn rate elevato, concorrenza forte nel settore AI-drug discovery, volatilità di mercato e possibile necessità di ulteriore capitale in futuro.
Outlook del report: bullish ma esplicitamente speculativo, adatto solo a chi è consapevole dei rischi tipici del biotech in fase clinica.
Authors
Research tools and partners
Screening, heatmaps and charts used for technical framing and comparative valuation checks.
ChartsWatcher is a real-time, next-generation scanner for the US stock market, used here to monitor momentum and intraday reaction around catalysts.
Used for earnings call transcripts, news flow aggregation, and cross-checking consensus estimates and rating changes.
Used to monitor real-time retail trader sentiment and message-volume spikes around key headlines.
Professional-grade trading and charting platform used operationally for execution and intraday monitoring.
Independent research hub focused on biotech catalysts, small caps and practical trading workflows.
Some external links above are referral or affiliate links. They do not change the cost for you but may provide a small commission that helps support the site and keep the research free. You are never required to use them.

Support the project
If you find this kind of long-form, sourced research useful and want to help keep it independent and accessible, you can support the site with a one-time PayPal donation.
Disclaimers and legal information
SEC-style disclaimer (EN): This report is for informational and educational purposes only. It is not, and should not be interpreted as, an offer to sell or a solicitation of an offer to buy any security, nor as personalized investment, legal, tax or accounting advice. The author is not a registered investment adviser or a broker-dealer. Any securities mentioned may be volatile and speculative. Investors should conduct their own independent research and, where appropriate, consult a licensed financial professional before making any investment decisions. Past performance is not indicative of future results; you can lose part or all of the capital invested.
Avvertenza CONSOB-style (IT): Il contenuto di questa pagina ha esclusivamente finalità informative e didattiche e non costituisce in alcun modo consulenza finanziaria personalizzata, sollecitazione del pubblico risparmio o raccomandazione a comprare o vendere strumenti finanziari ai sensi della normativa vigente. L’autore non è un consulente finanziario abilitato all’offerta al pubblico. Qualsiasi decisione di investimento resta sotto la piena responsabilità del lettore, che dovrebbe valutare con attenzione la propria situazione patrimoniale e, se necessario, rivolgersi a un professionista abilitato. Gli investimenti in strumenti azionari e, in particolare, nel settore biotech possono comportare la perdita parziale o totale del capitale investito.
For full legal notes and privacy information, please refer to:
Disclaimer –
Terms of use and privacy info.
Biotech Catalyst Calendar
For a broader view of upcoming FDA decisions, clinical readouts and other key biotech events beyond RXRX, you can consult the dedicated Biotech Catalyst Calendar maintained on Merlintrader trading Blog.
Open the Biotech Catalyst Calendar
Open the Biotech Catalyst Calendar
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


