DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
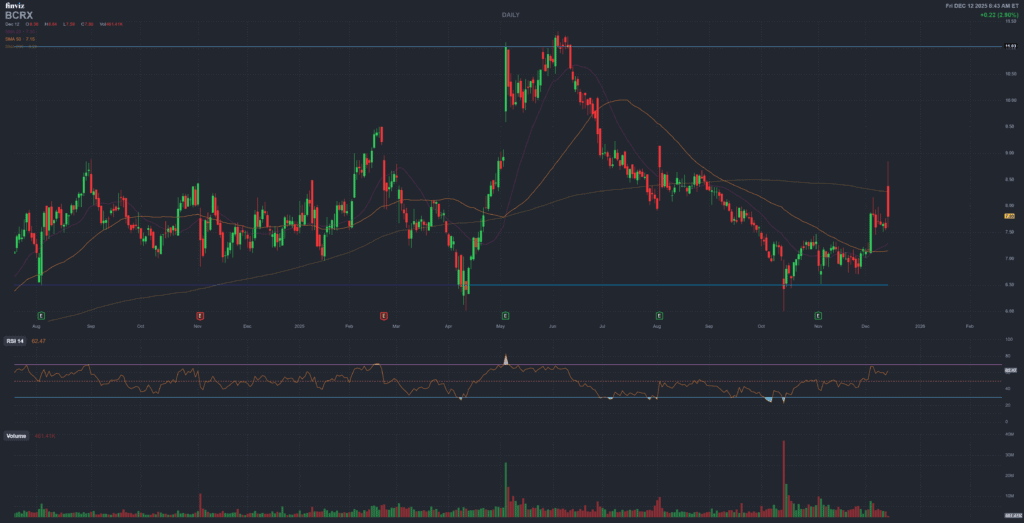
BCRX • BioCryst Pharmaceuticals
FDA decision • Positive
Published: Dec 12, 2025
BioCryst (BCRX) Update: FDA Approves ORLADEYO Oral Pellets for Younger Children
The FDA expanded ORLADEYO (berotralstat) prophylaxis use to children aged 2 to under 12 with hereditary angioedema (HAE), via a new “sprinkle-like” oral pellet formulation designed for pediatric administration. :contentReference[oaicite:4]{index=4}
Executive Summary
What happened (Dec 12, 2025): FDA approved ORLADEYO oral pellets for prophylaxis to prevent HAE attacks in children aged 2 to under 12, expanding use beyond the capsule label for ages 12+. :contentReference[oaicite:5]{index=5}
Why this matters (beyond a headline)
- Access + usability: the pellet formulation can be mixed with soft food or taken with liquids, addressing the “cannot swallow capsules” problem in younger children. :contentReference[oaicite:6]{index=6}
- Positioning: Reuters describes it as the first oral prophylactic option for younger children with HAE in the U.S. :contentReference[oaicite:7]{index=7}
- Commercial logic: pediatric starts can increase long-duration adherence (a “lifespan” therapy pathway), potentially improving long-term patient retention.
- De-risking: a binary regulatory event is resolved positively, shifting the narrative back to execution (uptake, access, international approvals, and pipeline integration).
What the FDA decision actually changed
| Before | Now |
|---|---|
| ORLADEYO once-daily capsule approved for ages 12+ (U.S.) :contentReference[oaicite:8]{index=8} | ORLADEYO oral pellets approved for ages 2 to under 12 (U.S.) :contentReference[oaicite:9]{index=9} |
| Younger children relied on targeted treatments via IV infusions or injections (as described by Reuters) :contentReference[oaicite:10]{index=10} | Oral pellet option designed for easier pediatric administration (mix with soft food / liquids) :contentReference[oaicite:11]{index=11} |
Regulatory timeline (what we can verify)
- May 14, 2025: BioCryst announced FDA acceptance of the pediatric NDA for ORLADEYO oral granules (2–11 years) and Priority Review (initial target action date noted in the release). :contentReference[oaicite:12]{index=12}
- Dec 12, 2025: FDA approval announced for pediatric use in children aged 2 to under 12 with the pellet formulation. :contentReference[oaicite:13]{index=13}
Market reaction: Reuters reported BioCryst shares up about 5% premarket on the approval news. :contentReference[oaicite:14]{index=14}
Commercial outlook: what to watch next
- Launch mechanics: early pediatric prescribing trends and payer coverage decisions (the practical “speed limit” for uptake).
- Switch dynamics: whether caregivers and physicians move from injectable prophylaxis options toward daily oral prophylaxis in younger patients.
- International filings: Reuters notes BioCryst has sought approval in Europe and Japan for pediatric use. :contentReference[oaicite:15]{index=15}
- HAE visibility: BioCryst has highlighted the pediatric burden of HAE (quality-of-life, caregiver impact) in company communications, which often supports awareness and adoption campaigns. :contentReference[oaicite:16]{index=16}
Company strategy context: Astria acquisition (HAE pipeline angle)
Reuters also linked the newsflow to BioCryst’s announced plan (Oct 2025) to acquire Astria Therapeutics in a deal valued around $700M to gain access to navenibart, a long-acting injectable HAE candidate. This keeps BioCryst’s HAE franchise narrative centered on both oral convenience (ORLADEYO) and next-gen long-acting options. :contentReference[oaicite:17]{index=17}
Financial / execution context (primary-company disclosures)
BioCryst’s Q3 2025 report references the pediatric ORLADEYO regulatory timeline and provides financial performance context for the ongoing franchise. (Use this section as the anchor for “verified numbers” when you refresh the article with the latest quarterly filing.) :contentReference[oaicite:18]{index=18}
Risks to keep on the radar (post-approval reality)
- Access & reimbursement: pediatric coverage decisions can be slower and more complex than adult coverage.
- Competitive HAE landscape: prophylaxis is a competitive category (different modalities and dosing schedules).
- Post-marketing surveillance: pediatric safety monitoring remains a continuing obligation even after approval.
- Execution risk: integrating pipeline/strategy moves (including M&A) can distract from commercial focus.
Sentiment snapshot (retail channels)
Retail chatter typically frames this as a “convenience + addressable market expansion” headline rather than a brand-new molecule story. A Stocktwits-linked news recap emphasized the approval and highlighted heightened retail attention around the event (sentiment is not a substitute for fundamentals). :contentReference[oaicite:19]{index=19}
Quick Takeaways
| Item | What we can verify |
|---|---|
| Approval | ORLADEYO oral pellets approved for HAE prophylaxis in children aged 2 to under 12 :contentReference[oaicite:20]{index=20} |
| Formulation | Sprinkle-like pellets; mix with soft food or swallow with water/milk :contentReference[oaicite:21]{index=21} |
| Prior label | Once-daily capsule approved for ages 12+ (U.S.) :contentReference[oaicite:22]{index=22} |
| Share move | ~5% premarket (Reuters) :contentReference[oaicite:23]{index=23} |
| International | Applications for pediatric formulation noted for Europe and Japan (Reuters) :contentReference[oaicite:24]{index=24} |
Primary Sources
- Reuters (Dec 12, 2025) — FDA expands approval for BioCryst’s ORLADEYO to younger children :contentReference[oaicite:25]{index=25}
- BioCryst IR (Dec 12, 2025) — FDA approval press release :contentReference[oaicite:26]{index=26}
- BioCryst IR (May 14, 2025) — FDA acceptance of pediatric NDA + Priority Review :contentReference[oaicite:27]{index=27}
- BioCryst IR (Nov 3, 2025) — Q3 2025 financial results :contentReference[oaicite:28]{index=28}
- BioCryst IR (Nov 6, 2025) — Pediatric HAE burden communications :contentReference[oaicite:29]{index=29}
Reader Note
This update focuses on verifiable facts (FDA decision + primary company communications + Reuters). Any forward-looking comments are framed as “what to watch” rather than predictions.
Related BCRX Reading (internal)
FDA Pillar + BCRX Articles
PDUFA – December 2025 (Global Calendar)
Published: 2025-12-04
-
BCRX BioCryst Pharmaceuticals – Main Report (Nov 21)
Published: 2025-11-21
-
BCRX – Deep Dive / Expanded Notes
Published: 2025-11 (site timestamp)
-
BCRX – Overview Page
Published: 2025-11-04
-
Catalyst FDA Report (Nov 11, 2025) – Includes BCRX ORLADEYO Pediatric NDA Timeline
Published: 2025-11-11 (site)
Tools & Partners
- Finviz
- ChartsWatcher — ChartsWatcher is a real-time, next-generation scanner for the US stock market.
- Seeking Alpha
- Stocktwits
- Medved Trader
- Merlintrader trading Blog
Affiliate note: Some links above may be affiliate/referral links. If you use them, it may support the site at no extra cost to you.

Support the site
If you find these reports useful and want to help keep the site free, you can donate here:
PayPal Donation Link
Legal:
Disclaimer
•
Terms & Privacy
Disclaimer (SEC/CONSOB style): This content is for informational and educational purposes only and does not constitute investment advice, an offer, or a solicitation. Biotech equities are volatile and catalyst-driven. Always verify data in primary sources and consider professional advice where appropriate.
Biotech Catalyst Calendar
Use the calendar to track upcoming biotech catalysts and key regulatory dates in one place.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


