DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
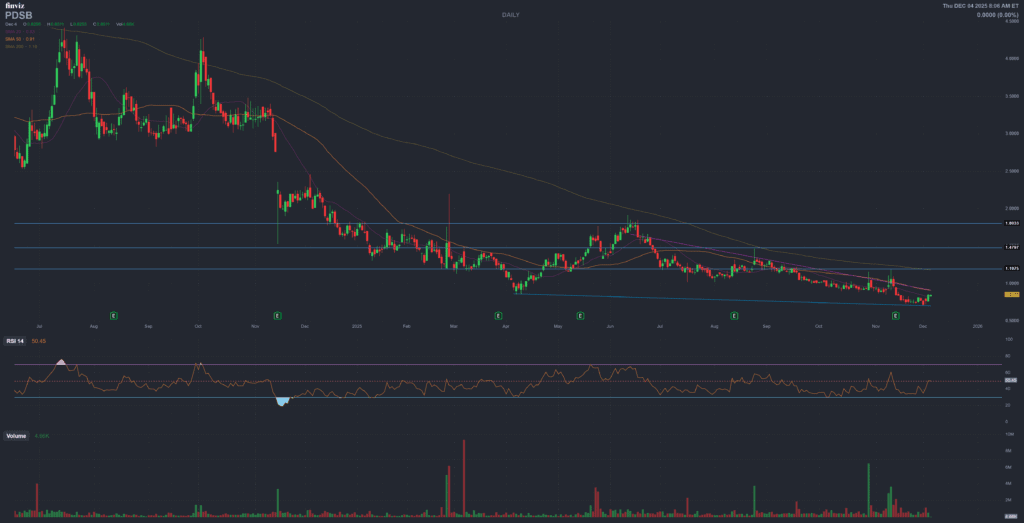
PDS Biotechnology Corporation (PDSB)
NASDAQ: PDSB
Comprehensive Analysis Report – December 4, 2025
-48.2%
YTD 2025
$0.899
Close (Dec 4, 2025)
$44.1M
Market Cap
~$33M
Estimated Cash (Dec 2025)
39.3 mo
mOS – VERSATILE-002
+119%
vs Keytruda monotherapy
Retail Sentiment – Stocktwits, X and Reddit (Dec 4, 2025)
The data below reflects sentiment and comments from non-professional traders on social platforms. It should be treated strictly as a sentiment indicator, not as research or investment advice.
Stocktwits
71% Bullish
Up from around 37% two days earlier, after the Dec 3 Type C 8-K. Message volume is roughly +438% vs the 30-day average.
Reddit (r/wallstreetbets, r/biotechplays, r/pennystocks)
+512%
Relative increase in mention volume vs 30-day average. One of the most upvoted posts on WSB (≈9.2k upvotes) summarizes it as: “$PDSB Jan minutes = moon or tomb”.
X (Twitter)
High Activity
Threads have spiked around “Type C meeting locked”, “accelerated approval on PFS” and “short squeeze potential”. Most accounts involved are retail traders, not institutions.
Short Interest Snapshot
21.1%
Short interest as of Nov 30, 2025, up from 18.4% as of Nov 15.
Days-to-cover: 5.8 based on ~1.18M average daily volume.
Executive Summary
On December 3, 2025, PDS Biotech announced via an official 8-K filing that the FDA has granted a Type C Meeting to discuss a potential accelerated approval pathway based on PFS for PDS0101 + pembrolizumab in first-line HPV16+ HNSCC.
The market reaction was immediate: the stock gained roughly +18% on Dec 3 and closed at $0.899 on Dec 4, about +12% across 48 hours.
SEC and Company-verified data (as of Dec 4, 2025)
- 8-K Filed: December 3, 2025 – Type C meeting formally accepted by the FDA.
- Cash (Sept 30, 2025): 36.8M USD (10-Q filed Nov 12, 2025).
- Average operating cash burn Q1–Q3 2025: ≈8.9M USD per quarter.
- Estimated cash at Dec 31, 2025: ≈33M USD, assuming similar burn.
- Implied runway: through approximately August 2026.
- Shares outstanding: 49.03M (including 2025 ATM usage).
- Short interest: 21.1% of float, the highest level in 2025.
Main Catalyst: FDA Type C Meeting
Why this Type C Meeting matters
A Type C Meeting focused on a potential accelerated approval (AA) pathway is not routine. It indicates that the FDA is willing to formally evaluate whether the PFS signal from VERSATILE-002 and the associated OS data may justify an AA filing for PDS0101 + pembrolizumab in the 1L HPV16+ HNSCC setting.
- The agency recognizes that the data package is at least potentially AA-relevant.
- PDS will receive written minutes clarifying what additional analyses or endpoints are required.
- The FDA is prepared to discuss PFS as a potential surrogate in this narrow indication.
Key regulatory questions
- Is the PFS improvement robust enough to be considered a surrogate of clinical benefit?
- Does the FDA require additional analyses or sensitivity work on the 002 dataset?
- What magnitude of benefit vs pembrolizumab monotherapy is considered compelling?
- Could a rolling submission be considered, depending on the Type C outcomes?
Possible FDA outcomes from the meeting
| Scenario | Summary |
|---|---|
| Positive (Green light) | FDA indicates that PFS, together with the OS trend, could support an AA filing. PDS is guided to prepare a formal submission, possibly with some additional analyses. |
| Conditional | FDA requests more detailed analyses or an updated dataset; AA remains on the table but contingent on further data, potentially including emerging VERSATILE-003 results. |
| Negative (No AA path) | FDA concludes that PFS is not acceptable as a surrogate in this setting and that full, mature OS data (or confirmatory data from 003) will be needed prior to any approval filing. |
VERSATILE-002 – Pivotal Signal Generation Trial
Key efficacy outcomes
| Metric | Outcome |
|---|---|
| Median OS (mOS) | 39.3 months in HPV16+ HNSCC |
| OS vs pembrolizumab monotherapy | Approx. +119% relative improvement vs historical benchmarks |
| PFS | Clinically meaningful extension vs available 1L data |
| Response profile | Particularly strong signal in PD-L1 low / difficult-to-treat subsets |
Interpretation of 002 results
VERSATILE-002 provides an unusually strong OS signal in a population where median survival under standard of care typically falls below 18 months. The durability of OS is the core argument PDS will bring to the FDA, supported by PFS and response dynamics. However, the absence of a large, randomized, controlled dataset keeps the regulatory discussion complex.
VERSATILE-003 – Confirmatory and Expansion Study
VERSATILE-003 is designed as a follow-up trial to validate and expand upon the 002 findings. While 002 was enriched for HPV16+ patients, 003 broadens the population and tests the robustness of the survival and PFS benefits in a setting closer to real-world practice.
Key design elements (high level)
- Population: 1L HPV16+ HNSCC, with broader inclusion criteria than 002.
- Regimen: PDS0101 + pembrolizumab combination.
- Primary endpoint: PFS.
- Key secondary endpoints: OS, ORR, DOR and safety.
Even if the FDA opens an accelerated approval path based on 002, VERSATILE-003 is likely to play a central confirmatory role. If 003 reproduces the strong survival signal, the regulatory and commercial case for PDS0101 significantly strengthens; if not, the thesis weakens rapidly.
Pipeline Overview
Beyond PDS0101 in HPV16+ HNSCC, PDS Biotech is building a tightly focused but mechanistically coherent pipeline around HPV-driven oncogenesis and the Versamune platform.
| Asset / Program | Indication | Status (approx.) |
|---|---|---|
| PDS0101 + pembrolizumab | HPV16+ HNSCC (1L) | Type C granted; VERSATILE-003 ongoing |
| PDS0101 (other HPV+ solid tumors) | Exploratory HPV16+ solid tumors | Early clinical / IND-enabling |
| PDS0102, PDS0103 | Additional HPV-related oncology indications | Preclinical / IND-enabling |
Financial Position – SEC Verified
Burn rate and runway (focus on Dec 2025)
- Cash (Sep 30, 2025): 36.8M USD, from the Q3 10-Q.
- Average burn Q1–Q3 2025: about 8.9M USD per quarter.
- Estimated cash at Dec 31, 2025: around 33M USD, assuming a similar burn pattern.
- Runway: roughly eight months, extending into August 2026.
- Capital raise: in the absence of a major partnership or deal, an equity financing sometime in 2026 appears almost inevitable.
Competitive Landscape – HPV16+ HNSCC
In 1L HPV16+ HNSCC, the competitive pressure is relatively limited. Pembrolizumab monotherapy remains the backbone standard of care, despite modest long-term outcomes, and there is room for a differentiated combination with a clear OS advantage.
| Player | Approach | Relevance vs PDS0101 |
|---|---|---|
| Merck – Keytruda | PD-1 monotherapy (backbone) | Main comparator; PDS0101 seeks to add value on top of this backbone. |
| IO-IO combinations | Dual checkpoint or novel IO mechanisms | Early and heterogeneous data; safety and cost can be challenging. |
| HPV-targeted vaccines (various) | Therapeutic vaccines | Fragmented field; none with OS data on par with VERSATILE-002. |
Analyst-Style Bull / Bear Framework
Bull case – what has to go right
- The FDA accepts PFS (supported by OS) as sufficient for an accelerated approval filing.
- VERSATILE-003 confirms, or meaningfully approximates, the 39.3-month OS trend seen in 002.
- Short interest unwinds rapidly if AA becomes clearly visible on the horizon.
- A partnership with a larger oncology player emerges after Type C minutes are published.
Bear case – what can break the thesis
- The FDA rejects PFS as a surrogate and requests mature OS or confirmatory results first.
- 002 proves non-reproducible in a broader 003 population.
- A 2026 capital raise happens at depressed levels, heavily diluting shareholders.
- Macro environment remains hostile to small-cap biotech, limiting any re-rating.
Key Risks
- Regulatory risk: no guarantee that the FDA will accept an accelerated approval based on PFS/002.
- Clinical risk: survival and PFS signals from 002 may not fully reproduce in 003.
- Financing risk: high probability of equity financing in 2026 given the current burn profile.
- Market risk: 21.1% short interest amplifies downside moves and volatility.
- Sector risk: sentiment toward small-cap biotech remains fragile and cyclical.
Bottom Line
PDS Biotech is at a critical inflection point: a strong survival signal and a receptive FDA Type C process give real regulatory optionality, but the balance sheet and the elevated short interest leave little room for execution errors. The coming months – from Type C minutes to any subsequent FDA feedback – will largely define whether PDS0101 transitions from a high-beta story into a credible, approvable regimen in HPV16+ HNSCC or remains constrained by trial and financing risk.
Official Sources and Useful Tools
SEC EDGAR – PDS Biotech Filings (10-Q, 8-K, 20-F if applicable)
Official Company Press Releases – PDS Biotech Investor Relations
PDSB – Technical Chart and Snapshot on Finviz (referral link)
PDSB – Fundamentals, Earnings, and Analyst Opinions on Seeking Alpha
α
Live PDSB Stream on Stocktwits (retail sentiment, referral link)
This report is based exclusively on public information from SEC filings, official company communications, and primary news outlets, cross-checked as of December 4, 2025. It is an educational and informational document intended for traders and investors and does not contain, and should not be interpreted as, any form of investment advice, portfolio allocation guidance, or buy/sell/hold recommendation regarding PDS Biotech or any other security.
Market conditions, prices, and company fundamentals can change quickly; therefore, data and interpretations in this report may become outdated or contain inaccuracies despite best efforts to verify sources carefully. Readers remain fully responsible for their own decisions and should always refer to official documentation and, where appropriate, independent professional advice. Nothing in this document is intended to comply with, or substitute for, any individualized regulatory or legal requirement in any specific jurisdiction.
PDS Biotechnology Corporation (PDSB)
NASDAQ: PDSB
Analisi completa – 4 dicembre 2025
-48.2%
YTD 2025
$0.899
Close 4 dicembre 2025
$44.1M
Market Cap
~$33M
Cash stimato a fine 2025
39.3 mesi
mOS VERSATILE-002
+119%
vs Keytruda monoterapia
Sentiment Retail – Stocktwits, Reddit e X (4 dicembre 2025)
I dati di questa sezione derivano da piattaforme pubbliche frequentate da trader non professionisti (Stocktwits, Reddit, X). Hanno valore puramente indicativo come termometro del sentiment e non vanno considerati ricerca, analisi fondamentale o raccomandazioni.
Stocktwits
71% Bullish
In rialzo da circa il 37% due giorni prima, dopo il filing 8-K del 3 dicembre. Volume dei messaggi circa +438% vs media 30 giorni.
Reddit (r/wallstreetbets, r/biotechplays, r/pennystocks)
+512%
Incremento relativo di menzioni rispetto alla media mensile. Uno dei post più votati su WSB riassume così: “PDSB Jan minutes = moon or tomb”.
X (Twitter)
Attività elevata
Thread concentrati su “Type C meeting locked”, “accelerated approval su PFS” e potenziale “short squeeze”. La maggioranza degli account coinvolti è retail.
Short Interest
21.1%
Short interest al 30 novembre 2025, in aumento da 18.4% al 15 novembre. Days-to-cover stimati: 5.8, su volume medio intorno a 1.18M azioni.
Executive Summary
Il 3 dicembre 2025 PDS Biotech ha comunicato tramite 8-K che la FDA ha concesso un Type C Meeting per discutere un potenziale percorso di approvazione accelerata basato sulla PFS per la combinazione PDS0101 + pembrolizumab in prima linea HPV16+ HNSCC.
La reazione di mercato è stata immediata: circa +18% nella seduta del 3 dicembre, con chiusura a $0.899 il 4 dicembre, pari a circa +12% sulle 48 ore successive all’annuncio.
Dati chiave verificati (SEC + PR ufficiali)
- 8-K depositato: 3 dicembre 2025 – accettazione formale del Type C Meeting.
- Cash al 30 settembre 2025: 36.8 milioni USD (10-Q del 12 novembre 2025).
- Burn operativo medio Q1–Q3 2025: circa 8.9 milioni USD/trimestre.
- Cash stimato al 31 dicembre 2025: ~33 milioni USD, assumendo burn simile.
- Runway stimata: fino indicativamente ad agosto 2026.
- Azioni in circolazione: 49.03 milioni (incluso utilizzo ATM 2025).
- Short interest: 21.1% del flottante, massimo 2025.
Type C Meeting – Perché è così importante
Un Type C Meeting centrato sulla possibilità di approvazione accelerata è un segnale chiaro che l’agenzia è disposta a valutare se il segnale di PFS (e la correlazione con l’OS) possa sostenere una domanda di approvazione in un’indicazione ad alto bisogno come l’HPV16+ HNSCC in prima linea.
Domande chiave sul tavolo FDA
- La PFS è sufficientemente robusta da fungere da surrogato di beneficio clinico?
- Che peso attribuire alla mOS di 39.3 mesi emersa da VERSATILE-002?
- Quali analisi aggiuntive o sensitivity analysis sono richieste sui dati 002?
- Che ruolo avrà VERSATILE-003 come potenziale confermatorio?
Possibili esiti del Type C
| Scenario | Descrizione sintetica |
|---|---|
| Positivo | La FDA apre in modo chiaro alla possibilità di approvazione accelerata su PFS, indicando un percorso praticabile per un futuro BLA. |
| Intermedio | La FDA non esclude l’AA ma chiede analisi aggiuntive, follow-up più lungo o dati di supporto da VERSATILE-003 prima di pronunciarsi. |
| Negativo | La FDA giudica PFS non accettabile come surrogato e spinge verso un iter più tradizionale con OS maturo o conferma randomizzata. |
VERSATILE-002 – Dati di efficacia
| Parametro | Risultato |
|---|---|
| mOS HPV16+ HNSCC | 39.3 mesi |
| Vantaggio vs storico Keytruda mono | circa +119% di sopravvivenza mediana |
| Profilo di risposta | risposte in sottogruppi tipicamente difficili (es. PD-L1 basso) |
| Sicurezza | nessun segnale di safety inatteso rispetto al backbone PD-1 |
Il dato di OS è il cuore della tesi su PDS0101: in un setting dove la sopravvivenza mediana è tipicamente inferiore a 18 mesi, un valore vicino ai 40 mesi è difficilmente ignorabile. Il rovescio della medaglia è la dimensione del campione e la natura non randomizzata del dataset, che rendono la discussione regolatoria più complessa.
VERSATILE-003 – Ruolo potenziale
VERSATILE-003 è lo studio che dovrebbe validare e ampliare i segnali osservati in 002, in un contesto più vicino alla pratica clinica reale.
- Popolazione: HPV16+ HNSCC in prima linea, criteri di inclusione più ampi.
- Regime: PDS0101 + pembrolizumab.
- Endpoint primario: PFS; secondari chiave: OS, ORR, DOR, sicurezza.
Anche nello scenario migliore di approvazione accelerata, VERSATILE-003 rimane fondamentale come potenziale confermatorio. Se il segnale di OS non si riproduce, il rischio di revisione della tesi nel medio periodo aumenta sensibilmente.
Pipeline PDS Biotech
| Programma | Indicazione | Stato |
|---|---|---|
| PDS0101 + pembrolizumab | HPV16+ HNSCC (prima linea) | Post-002, Type C con FDA, 003 in corso |
| PDS0101 (altre neoplasie HPV+) | Solidi HPV16-positivi | Sviluppo precoce / esplorativo |
| PDS0102 / PDS0103 | Altre indicazioni oncologiche HPV-correlate | Preclinico / IND-enabling |
Posizione finanziaria – dati SEC
Burn rate e runway (focus fine 2025)
- Cash Q3 2025: 36.8 milioni USD.
- Burn operativo medio: circa 8.9 milioni USD a trimestre.
- Cash stimato a fine dicembre 2025: ~33 milioni USD.
- Runway: circa 8 mesi, fino a indicativamente agosto 2026.
- Aumento di capitale: altamente probabile nel 2026 in assenza di accordi strutturali.
Framework Bull / Bear
Scenario bull (ottimistico)
- La FDA accetta un percorso di approvazione accelerata basato su PFS + OS 002.
- VERSATILE-003 conferma o avvicina il segnale di sopravvivenza di 002.
- Short interest si riduce rapidamente con miglioramento del newsflow.
- Arrivo di una partnership con big pharma sul programma HPV16+ HNSCC.
Scenario bear (prudente)
- La FDA respinge PFS come surrogato accettabile per AA.
- I dati di 003 risultano meno convincenti di 002.
- Aumento di capitale nel 2026 a valutazioni depresse, con diluizione significativa.
- Sentiment negativo prolungato sulle small-cap biotech, con scarso interesse istituzionale.
Rischi principali
- Rischio regolatorio: nessuna garanzia che la FDA approvi un’AA su PFS.
- Rischio clinico: riproducibilità dei dati di 002 in coorti più ampie.
- Rischio finanziario: necessità di ulteriore capitale entro 12 mesi.
- Rischio di mercato: short interest elevato amplifica volatilità e drawdown.
- Rischio di settore: fase ciclica debole per le small-cap biotech.
Bottom line
PDS Biotech è un caso scolastico di tesi binaria nel segmento small-cap oncologico: da un lato, un segnale di sopravvivenza molto forte e un Type C aperto sulla possibilità di approvazione accelerata; dall’altro, una struttura finanziaria limitata, short interest elevato e incertezza sul comportamento della FDA di fronte a un dataset non randomizzato.
I prossimi mesi – in particolare la lettura dei minutes del Type C e gli eventuali passi successivi indicati dall’agenzia – saranno decisivi per capire se PDS0101 potrà davvero entrare in un percorso di approvazione accelerata o se lo scenario resterà quello di uno sviluppo più lungo, complesso e diluitivo.
Fonti ufficiali e strumenti utili
Questo report si basa esclusivamente su informazioni pubbliche provenienti da documenti ufficiali (SEC filings), comunicazioni societarie e principali testate di news finanziarie, verificate al 4 dicembre 2025. È un documento a scopo educativo e informativo e non costituisce in alcun modo consulenza finanziaria personalizzata, sollecitazione al pubblico risparmio o raccomandazione di acquisto, vendita o mantenimento di strumenti finanziari.
Le condizioni di mercato, i prezzi e i fondamentali societari possono cambiare rapidamente; dati e interpretazioni qui riportati possono quindi diventare obsoleti o contenere inesattezze nonostante la cura posta nella verifica delle fonti. Chi legge resta pienamente responsabile delle proprie decisioni operative e dovrebbe sempre fare riferimento ai documenti ufficiali e, se necessario, a un consulente abilitato. Nulla in questo testo intende sostituire gli obblighi informativi o regolamentari previsti dalle autorità di vigilanza (CONSOB, SEC o altre).
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


