DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Janux Therapeutics Inc
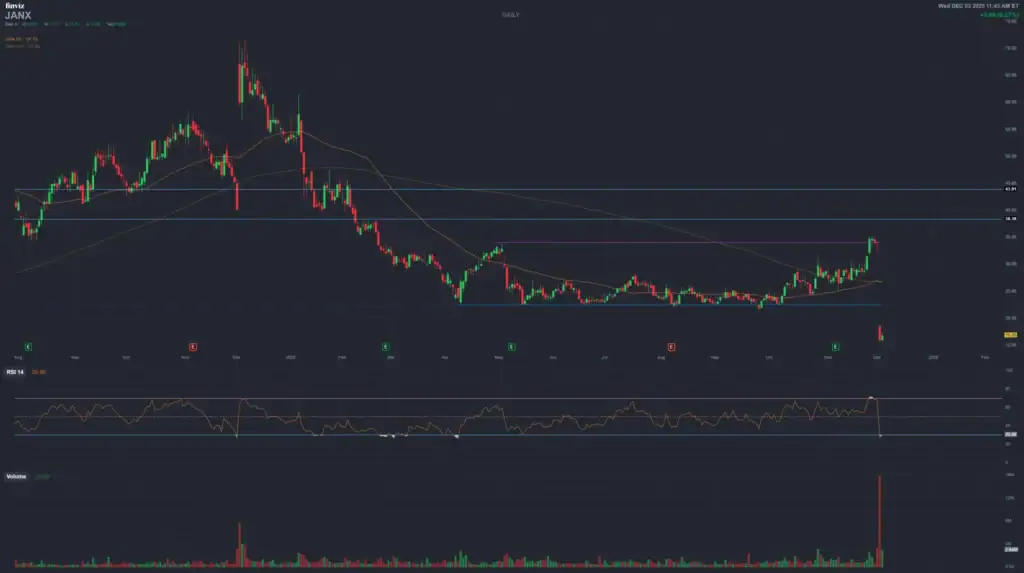
Janux Therapeutics (JANX)
Comprehensive 12-Month Deep Dive: From Peak Euphoria to December Crash & What Comes Next
December 3, 2025 — Caution: massive drawdown from 2025 highs
NASDAQ: JANX | TRACTr Platform (JANX007, JANX008) | mCRPC & Solid Tumors | Market Cap: ~US$1.0B
? THE SITUATION AT A GLANCE
Janux Therapeutics has gone from market darling to high-risk story stock in 2025:
- 52-Week High (early 2025): around US$71–72 per share (after strong early JANX007 data and sector momentum).
- Price after December 2–3, 2025 update: mid-teens (≈US$16–17), following a two-day collapse of ~50% after the latest Phase 1 JANX007 update.
- Key Trigger: interim JANX007 Phase 1 update on December 2, 2025 — data interpreted by the market as good but not clearly best-in-class, with limited transparency on expansion cohorts and future regulatory path.
- Core Debate: can JANX007 carve out a differentiated profile versus Amgen’s xaluritamig and J&J’s pasritamig in metastatic castration-resistant prostate cancer (mCRPC), or is it a “me-too” program with rich valuation and big execution risk?
- Cash Position (Q2 2025): US$996.0M in cash, cash equivalents and short-term investments — one of the strongest balance sheets in small/mid-cap biotech.
Table of Contents
- 1. 12-Month Timeline (Peaks & Failures)
- 2. Company Overview & CEO
- 3. Lead Program: JANX007 & Clinical Status
- 4. Financial Analysis (SEC-Verified Q2 2025)
- 5. December 2025 Crash Explained
- 6. Analyst Consensus & Price Targets
- 7. Trader Sentiment & Whispers
- 8. Upcoming Catalysts (Next 6–12 Months)
- 9. Key Risks & Red Flags
- 10. Valuation Framework
- 11. Sources, Tools & Support
- 12. Disclaimer & Disclosure
1. 12-Month Timeline: Peaks & Failures (2024–2025)
? December 2024
JANX007 Phase 1a data look very encouraging in heavily pre-treated mCRPC (median 4 prior lines). PSA responses and radiographic PFS spark enthusiasm that Janux may have a competitive TRACTr platform in prostate cancer.
? Q1 2025 — Euphoria
JANX rallies into the US$70s as investors price in “best-in-class” potential for JANX007, Merck’s historical interest in the platform, and broader appetite for oncology bispecifics. Liquidity is high and option activity aggressive.
✅ May 5, 2025
Phase 1b expansion launched: Janux opens taxane-naïve mCRPC cohorts in the ongoing Phase 1 study of JANX007, aiming to move the drug earlier in the treatment sequence and explore multiple dosing regimens.
? Q2 2025 Earnings (Aug 6, 2025)
Balance sheet impresses: Janux reports cash, cash equivalents and short-term investments of about US$996.0M, no debt, and quarterly R&D spend in the mid-30s (US$M). Runway >2 years at current burn; the company calls out progress with JANX007 and JANX008.
⏳ October 15, 2025 — Data Cut-off
Cut-off date for the latest JANX007 analysis used in the December 2, 2025 update. The company prepares a larger dataset including Phase 1b patients, but details remain under wraps until the call.
⚠️ December 2, 2025 — Inflection
Interim Phase 1 data update for JANX007 drops. Janux highlights median rPFS roughly in the 7–9 month range and a ~30% confirmed partial response rate in RECIST-evaluable patients. Safety (CRS) looks manageable. However, cross-trial comparisons versus Amgen’s xaluritamig and J&J’s pasritamig suggest no clear efficacy lead, and details on expansion cohorts are limited.
? December 2–3, 2025 — Market Reaction
Shares are cut roughly in half over two sessions. The market re-prices JANX from “best-in-class” hopes to “show-me” mode. Retail and momentum players capitulate; high-conviction biotech specialists debate whether this is overreaction or the start of a longer de-rating.
✓ Timeline verification: dates and cash numbers are based on Janux’s Q2 2025 10-Q and earnings release, the December 2, 2025 JANX007 press release, and mainstream financial news coverage of the subsequent stock move. Exact intraday price prints may differ depending on data provider.
2. Company Overview & Leadership
2.1 Company Profile
Janux Therapeutics, Inc. is a clinical-stage biopharmaceutical company founded in 2017 and headquartered in San Diego, California. The company develops tumor-activated immunotherapies using its proprietary TRACTr (Tumor Activated T Cell Engager) and TRACIr (Tumor Activated Immunomodulator) platforms, with the goal of improving the therapeutic window versus conventional T-cell engagers.
| Metric | Value |
|---|---|
| Founded | 2017 |
| Headquarters | San Diego, California (USA) |
| Public Listing | NASDAQ: JANX (IPO 2021) |
| Shares Outstanding (mid-2025) | ≈59M basic shares (per Q2 2025 filings) |
| Lead Program | JANX007 — PSMA-targeted TRACTr for metastatic castration-resistant prostate cancer (mCRPC) |
| Secondary Program | JANX008 — EGFR-targeted TRACTr for solid tumors (preclinical / early clinical development) |
2.2 CEO & Management
David Campbell, Ph.D. — Founder, President & CEO
- Background: >25 years of experience in biotech drug discovery, company building and partnerships.
- Education: Ph.D. in Organic Chemistry (Cornell University); B.S. in Chemistry (Harvey Mudd College).
- Prior Roles:
- Leadership roles at Sitari Pharma, Afraxis, RQx Pharmaceuticals and Phenomix, with multiple partnering deals with large pharma (e.g., GSK, Genentech).
- Earlier career at Bayer in discovery research.
- Insider alignment: beneficial owner of a meaningful equity stake in Janux according to the latest proxy statement, aligning his incentives with long-term shareholders.
- Assessment: experienced oncology entrepreneur with a history of moving assets into larger-pharma hands; execution on JANX007 / JANX008 will be the real test of this platform.
3. Lead Program: JANX007 & Clinical Status
3.1 JANX007 Mechanism & Rationale
JANX007 is a PSMA-targeted, tumor-activated T-cell engager (TRACTr) designed for metastatic castration-resistant prostate cancer. It combines a PSMA-binding arm with a masked CD3 arm; the CD3 epitope is unmasked by proteases in the tumor micro-environment, with the goal of concentrating T-cell activation in tumors while limiting systemic cytokine release.
3.2 Current Clinical Status (December 2025)
| Metric | Value / Status |
|---|---|
| Trial ID | ENGAGER-PSMA-01 – Phase 1 (NCT05519449) |
| Phase | Phase 1 (dose escalation completed; multiple Phase 1b expansions ongoing) |
| Population | Adults with metastatic castration-resistant prostate cancer (mCRPC), including heavily pre-treated and taxane-naïve cohorts. |
| Dose Escalation (Phase 1a) | Heavily pre-treated patients; early data showed deep PSA declines and objective responses at target doses. |
| Phase 1b Taxane-Naïve Expansion | Ongoing; evaluates JANX007 at multiple dose levels and schedules earlier in the treatment paradigm. |
| Primary Endpoints | Safety/tolerability, dose-limiting toxicities, PK/PD; key efficacy readouts include radiographic PFS and objective response rate. |
| rPFS (headline range, Dec 2025 update) | Roughly 7–9 months in heavily pre-treated patients, depending on dose and schedule (company commentary). |
| Response Rate | Around 30% confirmed partial responses among RECIST-evaluable patients in the analyzable set, with higher PSA response rates. |
| Safety Profile | Cytokine release syndrome predominantly Grade 1–2 and manageable with mitigation strategies; no unexpected safety signals reported so far. |
| Competitive Comparison | Amgen’s xaluritamig and J&J’s pasritamig have shown rPFS in the high-7-month range in similar mCRPC settings; cross-trial comparisons are imperfect but set the “bar” in investors’ minds. |
Key Clinical Questions After the December 2025 Update
- Is JANX007 clearly differentiated? Current data suggest competitive activity, but not a slam-dunk superiority versus xaluritamig/pasritamig. Investors were hoping for a clean “best-in-class” story; instead, they got nuance.
- How strong is the taxane-naïve signal? Limited disclosure so far on Phase 1b expansion cohorts — the market wants to see more granularity by line of therapy, dose, schedule and PSMA expression.
- Durability and depth: long-term follow-up will be needed to understand how durable responses are and whether the rPFS curve bends favourably over time.
- Future combinations: planned combinations with androgen-receptor-targeted agents (e.g., darolutamide) could enhance efficacy but also add safety complexity.
3.3 Secondary Program: JANX008 (EGFR-TRACTr)
JANX008 targets EGFR-expressing solid tumors (such as subsets of NSCLC and other epithelial cancers). Preclinical work has shown promising activity, and Janux is positioning this molecule as a second pillar of the TRACTr platform. As of December 2025, JANX008 remains earlier-stage and is not yet the main driver of the stock, but positive safety/efficacy signals here could diversify the story beyond prostate cancer.
4. Financial Analysis (SEC-Verified Q2 2025)
4.1 Balance Sheet Snapshot (June 30, 2025)
Cash & Equivalents
$52.4M
Short-Term Investments
$943.6M
Total Liquid Assets
$996.0M
Total Assets
$1,032.2M
Stockholders’ Equity
$990.5M
Total Liabilities
$41.7M
| Item | Q2 2025 | Q2 2024 | 6M 2025 | 6M 2024 |
|---|---|---|---|---|
| Collaboration Revenue | $0.0M | $8.9M | $0.0M | $10.1M |
| R&D Expense | $34.7M | $14.9M | $59.7M | $29.0M |
| G&A Expense | $10.5M | $7.8M | $20.3M | $15.2M |
| Total Operating Expenses | $45.1M | $22.7M | $80.0M | $44.1M |
| Net Loss | ($33.9M) | ($6.0M) | ($57.4M) | ($20.7M) |
| Diluted EPS | ($0.55) | ($0.11) | ($0.93) | ($0.40) |
- Cash Runway: roughly US$996M of liquid assets vs. a quarterly burn around US$40M implies >2 years of runway at the current spend level — no immediate financing pressure, even after the stock drop.
- End of Merck Collaboration: the Merck collaboration has wound down, so collaboration revenue has dropped to zero; Janux is now fully dependent on internal R&D progress and any future deals.
- R&D Acceleration: R&D more than doubled year-on-year as JANX007 moves through Phase 1b and platform programs scale; this is where most of the cash is going.
- Equity Dilution: share count has increased meaningfully since IPO, reflecting stock-based comp and capital raises typical for a high-burn clinical-stage company.
- Binary Profile: with no product revenue and a single lead clinical asset, Janux’s valuation is highly sensitive to every data update on JANX007.
✓ SEC verified: all figures in this section come directly from Janux’s Form 10-Q for the quarter ended June 30, 2025 (filed August 6, 2025) and the accompanying earnings press release.
5. December 2025 Crash Explained: What Went Wrong?
5.1 The Data
On December 2, 2025, Janux released an interim update from the ongoing Phase 1 JANX007 trial. The company framed the data as “encouraging,” but investors clearly wanted more.
Key elements of the update (high-level):
- Heavily pre-treated mCRPC population with multiple prior lines of therapy.
- Median radiographic PFS in the general range of 7–9 months, depending on dose and schedule.
- Confirmed partial response rate around 30% among RECIST-evaluable patients; PSA response rates higher.
- Cytokine release syndrome largely manageable (mostly Grade 1–2) with mitigation measures.
Why the market was disappointed:
- No obvious best-in-class edge: cross-trial comparisons to xaluritamig and pasritamig suggested similar efficacy, not a clear win, even acknowledging the limitations of comparing different trials.
- Limited transparency on Phase 1b: investors got high-level numbers but little granularity on taxane-naïve cohorts, by-dose breakdown or deeper durability analyses.
- Path to Phase 2 and registration left vague: no detailed Phase 2 design or timing, fuelling concern that the program may need more time and data before a clear regulatory strategy is locked in.
- Expectations were elevated: after the run into the US$70s, sentiment was skewed bullish; anything short of a home-run was likely to trigger a sharp re-rating.
5.2 Market Reaction
| Date | Approx. Close | Move vs. Pre-Update | Context |
|---|---|---|---|
| Dec 1, 2025 (pre-announcement) | Low-US$30s | — | Stock discounts strong Phase 1b read-through and a clear regulatory path. |
| Dec 2, 2025 | High-US$10s / low-US$20s | ≈-40–50% | Interim data perceived as “good but not spectacular”; aggressive de-rating. |
| Dec 3, 2025 | Mid-teens | Further pressure intraday | Follow-through selling, position clean-up, and early analyst reactions. |
Analyst & Market Commentary (high-level tone)
Several banks acknowledged that the absolute efficacy of JANX007 looks competitive, but highlighted gaps in disclosure and the lack of a clear edge versus emerging PSMA T-cell engagers. The common theme: good data in a tough competitive context, with valuation and expectations previously ahead of themselves.
6. Analyst Consensus & Price Targets
6.1 Before the Crash
| Metric | Indicative Pre-Crash Levels |
|---|---|
| Number of covering analysts | High-teens (specialist biotech coverage) |
| Consensus rating | Overwhelmingly “Buy/Outperform”, with very few Neutral ratings. |
| Average 12-month target | In the US$70–80 range. |
| Target range | Wide, from mid-US$20s on the low end to >US$200 on the high end. |
6.2 After the Crash (What to Expect)
Formal post-crash updates are still rolling in, but several dynamics are likely:
- Multiple target cuts as analysts re-run models with more conservative peak-sales and probability-of-success assumptions.
- At least some downgrades from Buy to Hold/Neutral for houses focused on 12-month risk/reward rather than long-term optionality.
- More emphasis on “show-me” new data from Phase 1b and any emerging combinations, rather than extrapolating early data out to blockbuster scenarios.
7. Trader Sentiment & Whispers (“What They’re Saying”)
?️ TRADER SENTIMENT BOX — Unverified Social-Media Commentary
⚠️ NOTE: the comments below are paraphrased from public forums (StockTwits, Reddit, Twitter/X, etc.) to illustrate market mood. They are not fact-checked and must not be used as the basis for any investment decision.
“JANX has turned into a binary show-me story. If Phase 1b in taxane-naïve patients looks clearly better, this bounces; if not, it stays a teens stock.”
“The data aren’t bad, they’re just not clearly best-in-class. The bar was set way too high. I’ll wait for the dust to settle.”
“Huge cash pile, so no near-term raise. That’s the only reason I’m even considering a position here after a -50% move.”
“Feels like classic biotech over-reaction, but I’d rather see one more clean update before stepping in. There are easier trades.”
Overall sentiment (0–10, where 10 = wildly bullish): roughly 3–4/10 — pessimistic and bruised, with a minority arguing the sell-off is an opportunity if JANX007 can close the perceived gap versus competitors.
8. Upcoming Catalysts & Regulatory Timeline (6–12 Months)
Key Milestones to Watch
- More detailed Phase 1b data (2026): fuller cut of JANX007 in taxane-naïve and other expansion cohorts, ideally with more granular breakdowns and durability curves.
- Formal Phase 2 design and timing: clarity on endpoints, comparator arms (if any) and geographies will shape the regulatory narrative.
- Combination-strategy updates: news on JANX007 combinations (e.g., with darolutamide or PARP inhibitors) could change the perceived risk/benefit profile.
- JANX008 progress: a clean safety signal and early efficacy hints in EGFR-driven tumors would help diversify the story.
- Partnerships or collaborations: any new strategic deal with big pharma would be taken as external validation of the platform and could de-risk funding needs.
9. Key Risks & Red Flags
Clinical & Regulatory Risks
- Competitive pressure: if xaluritamig and pasritamig move faster through development or show a clearer efficacy edge, JANX007 could be relegated to a smaller slice of the market.
- Uncertain registrational path: Phase 1/1b data alone are unlikely to be enough for approval; requirements for Phase 2/3 design, control arms and follow-up could be demanding.
- Safety / immunogenicity over time: longer follow-up might reveal late toxicities or immunogenicity that are not yet fully visible in the current dataset.
Financial & Market Risks
- Burn vs. pipeline breadth: aggressive investment into multiple TRACTr programs could force Janux to increase spend just as the market becomes less willing to fund early-stage stories.
- Dilution risk later in the decade: even with a strong current cash balance, additional capital could be required for Phase 3 programs or commercial build-out if trials are expanded.
- Biotech sector volatility: macro risk-off episodes can drag JANX down regardless of company-specific news.
10. Valuation Framework & Scenario Thinking
? Bull Scenario (Long Term)
$50 – $75+
Assumptions: JANX007 shows clear, reproducible differentiation in Phase 1b/2 (e.g., stronger rPFS and clean safety); combinations work; regulators accept a relatively efficient path; JANX008 or other TRACTr assets add a second leg to the story. In this world, JANX could justify a multi-billion-dollar enterprise value.
? Base Scenario
$25 – $40
Assumptions: JANX007 remains competitive but not obviously best-in-class; the program moves into Phase 2 on a reasonable timeline; cash runway is used efficiently; and the sector backdrop is neutral/slightly positive. Stock revisits pre-crash levels over time but with higher volatility.
⚠️ Bear Scenario
$5 – $15
Assumptions: further updates fail to impress; competitors capture most of the PSMA-TCE narrative; timelines slip; and the market grows sceptical about long-term platform value. Dilution later in the decade becomes painful and the stock trades as a binary option on a crowded space.
Important: the ranges above are illustrative scenario bands, not price targets or recommendations. They are meant to help frame risk/reward under different combinations of clinical, competitive and market outcomes.
11. Sources, Tools & Support
Primary Sources & Tools
- SEC Filings: Janux 10-Q and 10-K filings — EDGAR company page: SEC – Janux Therapeutics
- Q2 2025 Earnings Release: detailed financials and cash breakdown.
- Clinical Trials: Phase 1 JANX007 trial ENGAGER-PSMA-01 — clinicaltrials.gov search
- Official Company Data: investor presentations and JANX007 clinical updates on the Janux IR site.
- Market Data: real-time and historical quotes from your preferred broker/data provider.
Affiliate Tools (for further research)
Support the project
If this type of deep-dive helps you navigate the biotech jungle and you’d like to support the work behind Merlintrader trading Blog, you can contribute via PayPal:
12. Disclaimer & Disclosure
⚠️ IMPORTANT DISCLAIMER
This report is for informational and educational purposes only. It is not investment advice, and it does not constitute an offer or solicitation to buy or sell any security.
- High-risk profile: clinical-stage biotech companies like Janux can be extremely volatile and carry a material risk of permanent capital loss. A single trial setback or financing event can significantly impact the share price.
- Data sources: financial figures draw from SEC filings and official company communications; clinical information is based on press releases, trial registries and reputable news outlets as of the report date. Market data (prices, ranges) depend on third-party providers and may differ slightly across platforms.
- No personalised advice: I do not know your personal financial situation, objectives or risk tolerance. Nothing here should be interpreted as a personalised recommendation. Always consult a qualified financial adviser before making investment decisions.
- Forward-looking uncertainty: any discussion of “scenarios,” “runway,” or potential future valuations is inherently uncertain and may turn out to be wrong. Biotech development is subject to regulatory, clinical, competitive and macroeconomic risks.
- Regulatory note (EU / US): this content is not produced by a licensed investment firm or a registered investment adviser. It should be viewed as independent commentary and educational analysis, not as research prepared under MiFID II or SEC/FINRA research rules.
Bottom line: treat JANX as a speculative biotech position that depends heavily on future clinical outcomes. Never invest more than you can afford to lose, and always cross-check key numbers directly in the primary sources linked above.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


