DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

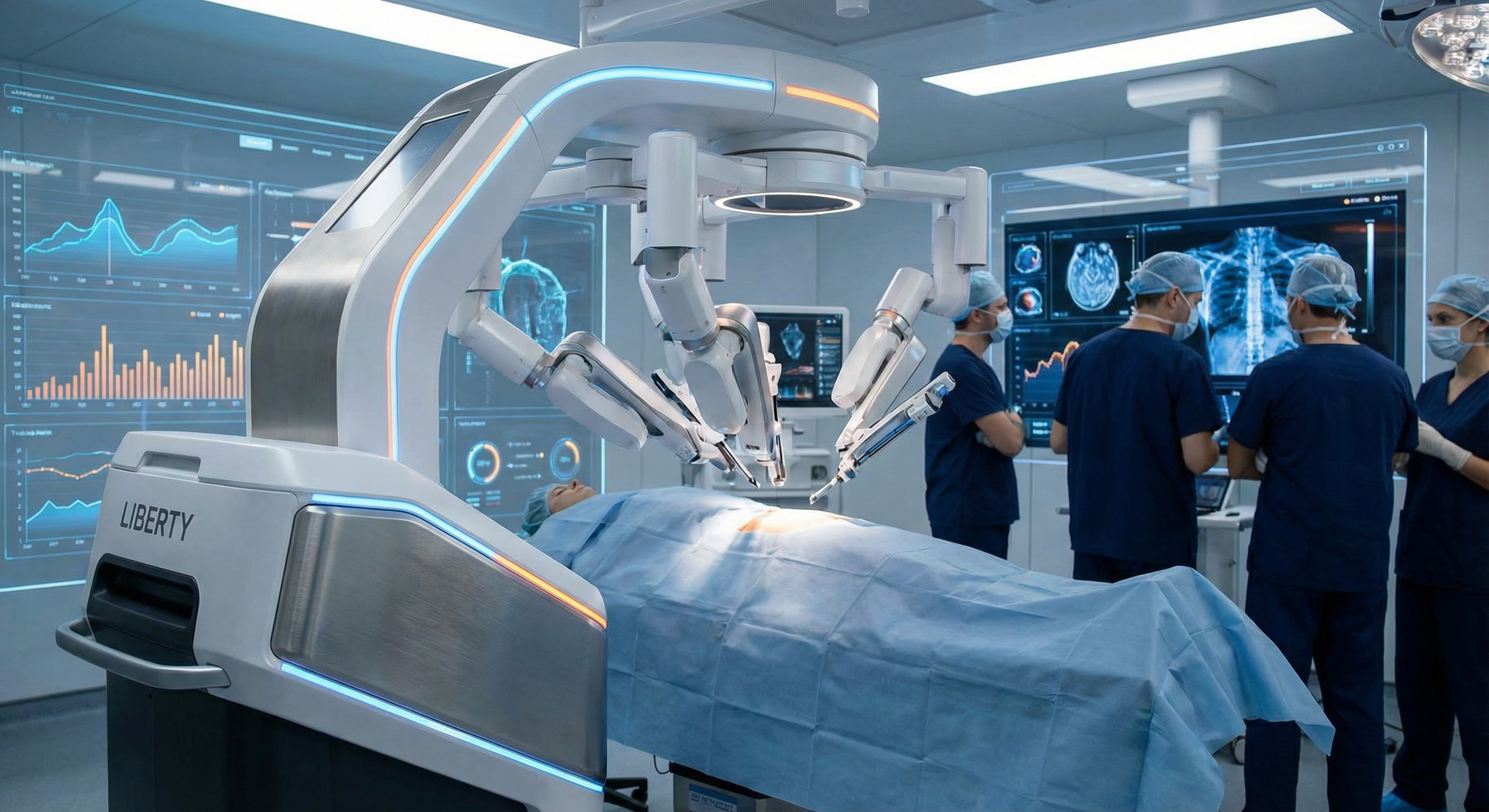
Microbot Medical Inc.
MBOT NASDAQReport Date: November 28, 2025 | Sector: Medical Devices – Surgical Robotics
Analyst Target: $12.00 (HC Wainwright – Buy)
Executive Summary
Microbot Medical is a commercial-stage medical device company specializing in endovascular robotic systems. The company achieved a transformational milestone in September 2025 with FDA 510(k) clearance for its flagship LIBERTY® Endovascular Robotic System – the world’s first FDA-cleared single-use, remotely operated robotic platform for peripheral endovascular procedures.
The company has transitioned from clinical-stage to commercialization phase, with Emory University Hospital becoming the first facility globally to adopt LIBERTY in November 2025. Full market launch is planned for the SIR Conference in April 2026. The company secured up to $92.2 million in financing through preferred investment options in September 2025, providing a strong runway for commercialization activities.
Development Timeline (2024-2025)
June 2024
FDA approval to advance pivotal human clinical trial (IDE expansion). Brigham and Women’s Hospital announced as clinical trial site.
July 2024
Clinical trial partnership with Baptist Hospital of Miami (including Miami Cardiac & Vascular Institute and Miami Cancer Institute). Successful completion of first clinical trial procedures.
August 2024
Phase 2 collaboration agreement signed with Corewell Health for robotic telesurgery development. Partnership with Emory University to develop autonomous capabilities.
October 2024
Successful completion of ACCESS-PVI pivotal human clinical trial enrollment and follow-up. All patients completed the study.
December 2024
FDA 510(k) submission filed for LIBERTY® System as planned. Company concluded year achieving key milestones.
January-February 2025
Raised approximately $30 million through various offerings to fund operations. Settlement agreement related to 2017 financing lawsuit ($1.1M cash + 1,005,965 shares).
February 2025
Corewell Health collaboration reached another meaningful milestone in robotic telesurgery advancement. Dr. Ryan Madder leading simulated cardiovascular procedures evaluation.
April 2025
ACCESS-BVI pivotal trial completed with 100% success rate in robotic navigation to target. Zero device-related adverse events through 3-day follow-up. Data presented at SIR Conference showing 92% reduction in radiation exposure.
September 8, 2025
FDA 510(k) CLEARANCE GRANTED – LIBERTY® becomes the first FDA-cleared single-use, remotely operated robotic system for peripheral endovascular procedures.
September 15-16, 2025
Exercise of outstanding preferred investment options announced, generating up to $92.2 million in financing.
November 26, 2025
Limited market introduction initiated. Emory University Hospital becomes the FIRST hospital in the world to adopt LIBERTY® for patient treatment. Partnership established to develop Endovascular Robotics Program.
Financial Position (SEC Verified)
Cash & Securities (Q2 2025)
~$32.7M
H1 2025 Financing
$32.6M
Q3 2025 Warrant Exercises
Up to $92.2M
Total Equity (Nov 2025)
>$78M
Accumulated Deficit
~$97.0M
FY 2024 Net Loss
$11.44M
H1 2025 Net Loss
$6.1M
Revenue Status
Pre-Revenue
Note: Per SEC 10-Q (Aug 2025), cash position as of June 30, 2025 was ~$32.7M. Following September 2025 preferred warrant exercises (generating up to $92.2M), current cash position is expected to be significantly higher. Management believes current funds are sufficient to operate for more than 12 months. The company has an accumulated deficit of ~$97.0M and continues to incur operating losses.
Pipeline & Technology Assets
| Product | Description | Status | Target Market |
|---|---|---|---|
| LIBERTY® | Single-use, remotely operated endovascular robotic system for peripheral procedures | FDA CLEARED | ~2.5M annual peripheral vascular procedures (US) |
| LIBERTY® Telesurgery | Remote telesurgery functionality for cardiovascular interventional procedures | Phase 2 Dev | Remote/underserved healthcare facilities |
| LIBERTY® Autonomous | Future autonomous navigation capabilities | Early Dev | Enhanced procedural efficiency |
| NovaCross | Guidewire navigation technology platform | Development | Complex vascular navigation |
Leadership – CEO Profile
HG
Harel Gadot
Chairman, CEO & President (Since 2010)
Seasoned healthcare executive and entrepreneur with over 20 years of leadership experience across the United States, Europe, and Israel. Co-founded Microbot Medical in 2011 and led the company through its successful NASDAQ listing in 2016. Founder of MEDX Ventures Group (2010), a unique healthcare investment and management firm. Also serves as Founder and Chairman of XACT Robotics Ltd. and MEDX Xelerator LP, a government-supported medical devices incubator in Israel with partnerships including Boston Scientific Corporation and Sheba Medical Center.
Prior Experience: Regional Marketing Manager at Johnson & Johnson (Ethicon Inc., Paris), leading strategic marketing for Europe, Middle East, and Africa. Education: Marketing and Management degree from Siena College (1997).
Strategic Partnerships & Collaborations
Hospital Partner #1
Emory University Hospital
First global adopter – Nov 2025
Telesurgery Partner
Corewell Health
Phase 2 collaboration
Clinical Trial Site
Brigham and Women’s
IDE pivotal trial site
Clinical Trial Site
Baptist Hospital Miami
Miami Cardiac & Cancer Institute
Upcoming Catalysts
Full Market Launch – SIR Conference
Official commercial launch of LIBERTY® at Society of Interventional Radiology conference
April 2026
Continued Clinical Data Collection
Ongoing real-world data gathering during commercial rollout phase
Q4 2025 – 2026
International Market Expansion
Exploration of international markets following US commercial success
2026+
Telesurgery Development Milestones
Advancement of Corewell Health collaboration for remote surgery capabilities
Ongoing
Analyst Coverage
Analyst Firm
H.C. Wainwright
Rating
BUY
Price Target
$12.00
Last Updated
Nov 28, 2025
Investment Perspectives
? Bull Case
- First-mover advantage with only FDA-cleared single-use endovascular robot
- 100% clinical trial success rate with zero device-related adverse events
- 92% reduction in radiation exposure for medical staff
- $2.5M+ addressable annual US procedures market
- Strong balance sheet with $92.2M financing secured
- Prestigious first adopter (Emory University Hospital)
- Telesurgery potential for underserved markets
- Single-use model eliminates capital equipment barriers
? Bear Case
- Pre-revenue company with continued operating losses
- Commercial execution risk in competitive robotics market
- Reliance on third-party manufacturers
- Operations in Israel subject to geopolitical risks
- Royalty obligations to Israeli Innovation Authority
- Limited analyst coverage (single firm)
- Potential dilution from recent financings
- Adoption timeline and reimbursement uncertainties
Primary Sources
⚠️ Disclaimer
This report is for informational purposes only and does not constitute investment advice, a recommendation, or an offer to buy or sell securities. The information contained herein has been compiled from publicly available sources believed to be reliable, including SEC filings, company press releases, and news wire services. However, no guarantee is made as to its accuracy or completeness. Past performance is not indicative of future results. Investing in stocks involves substantial risk of loss. Always conduct your own due diligence and consult with a qualified financial advisor before making investment decisions. The author may hold positions in securities mentioned. This content is not intended as personalized investment advice.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


