DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
? ACRS – ACLARIS THERAPEUTICS
Fundamental & Technical Analysis | SEC-Verified Q3 2025

? Executive Summary
Aclaris Therapeutics [finance:Aclaris Therapeutics, Inc.] (NASDAQ: ACRS) is a clinical-stage dermatology-focused biopharmaceutical company developing innovative immunological therapies for inflammatory and immune-mediated skin diseases. The company transitioned from commercial-stage to clinical-stage organization, building a pipeline with three programs in Phase 1b-2 stage.
KEY HIGHLIGHTS (Q3 2025 – SEC VERIFIED):
- ✓ Total Liquidity: $167.2M (Q3 2025) — runway extending through 2027-2028
- ✓ Minimal Debt: $2.24M total debt vs. $167.2M liquidity = strong net cash
- ✓ Burn Rate: ~$4M/month (accelerating in 2025); runway ~42 months
- ✓ Pipeline: ATI-2138 (Phase 2a+ positive), ATI-052 (Phase 1b cleared), ATI-045 (Phase 2 active)
- ✓ Recent Catalysts: ATI-2138 Phase 2a positive (Jul 2025), ATI-052 IND cleared (Apr 2025)
- ⚠ Risks: 53% share dilution since 2023, accelerated R&D burn, clinical execution risk
? Financial Position (Q3 2025 – SEC Verified)
Update: Q3 2025 10-Q filed November 6, 2025 shows updated financial position with $167.2M total liquidity (down from $203.9M in Dec 2024). Burn rate accelerating due to expanded R&D programs.
Balance Sheet Q3 2025
| Metric | Q3 2025 | Dec 2024 | Change |
|---|---|---|---|
| Cash & Equivalents | $25.3M | $24.6M | +$0.7M |
| Short-term Securities | $70.7M | $89.0M | -$18.3M |
| Long-term Securities | $71.3M | $90.3M | -$19.0M |
| TOTAL LIQUIDITY | $167.2M | $203.9M | -$36.7M (-18%) |
| Total Debt | $2.24M | $0.0M | Minimal (leases) |
| Stockholders’ Equity | $120.1M | $155.6M | -23% |
CRITICAL UPDATES:
- Burn Rate Accelerating: $36.7M consumed in 9 months (2025) = ~$4M/month
- R&D Spending Up 35%: $36.1M YTD 2025 vs. $24.6M prior year (Phase 1b-2 programs expanded)
- Equity Dilution: 108.3M shares (Oct 2025) vs. 70.9M (Dec 2023) = 53% dilution from $80M private placement
- Estimated Runway: $167.2M ÷ $4M/month = ~42 months (late 2028)
? Clinical Pipeline & Upcoming Catalysts
Lead Program: ATI-2138 (Oral ITK/JAK3 Inhibitor)
? MOST ADVANCED – Atopic Dermatitis
Status: Phase 2a (POSITIVE DATA – July 29, 2025)
Mechanism: Selective oral covalent ITK/JAK3 inhibitor
Data: 14-patient open-label Phase 2a showed favorable tolerability, NO serious adverse events, mild transient side effects at 10mg BID
Next Catalyst: Phase 2b interim/top-line data Q2-Q3 2026 (±6 months estimate)
Secondary: ATI-052 (Bispecific Antibody)
? IND CLEARED – Immune-mediated Conditions
Status: Phase 1a/1b (IND Cleared April 22, 2025)
Mechanism: Bispecific anti-TSLP/IL-4R monoclonal antibody (dual-pathway)
Design: Randomized, double-blind, placebo-controlled; SAD/MAD + PoC phase
Next Catalyst: Phase 1a/1b PoC data Q4 2025 or Q1 2026 (±3 months estimate)
Tertiary: ATI-045 (Bosakitug)
? PHASE 2 ONGOING – Atopic Dermatitis
Status: Phase 2 initiated June 2, 2025
Mechanism: TSLP monoclonal antibody (licensed from Biosion)
Design: Randomized, double-blind, placebo-controlled Phase 2 in moderate-to-severe AD
Next Catalyst: Phase 2a top-line data Q4 2025 or Q1 2026 (±3 months estimate)
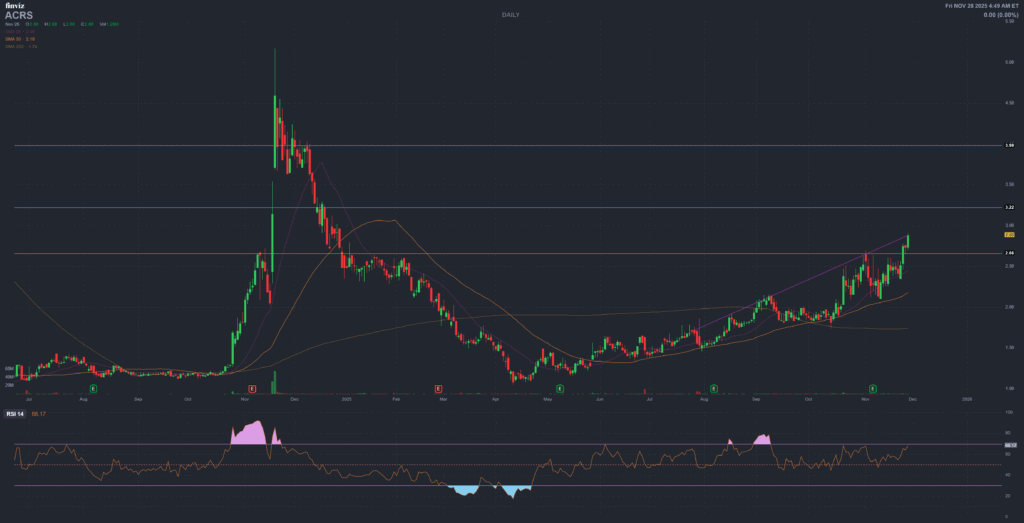
? Technical Analysis – Critical Levels
Support Zones (Historical Paused Points)
? FIRST SUPPORT: $2.00-2.25
Observation: Historical support zone where selling exhausted. Volume historically increases at this level. Next support if broken: $1.50-1.75
? SECOND SUPPORT: $1.50-1.75
Observation: 52-week historical low zone. Strong volume cluster. Critical threshold—breaches suggest liquidation or severe negative catalyst
Resistance Zones (Historical Selling Points)
? FIRST RESISTANCE: $4.50-5.00
Observation: Historical resistance where selling emerged. Multiple touches without breakout. Breakout above $5.00 would signal bullish shift
? SECOND RESISTANCE: $6.50-7.50
Observation: Upper band from 2024 action. Heavy historical volume. Breakout would require significant bullish catalyst (positive Phase 2 data)
Volatility & Volume Observations
- Intraday Swings: 5-15% common on neutral days; 20-40% on catalyst days
- Average Volume: 1-2M shares daily; spikes to 5-10M+ on catalysts
- Volume Pattern: Spikes 2-3x average on clinical announcements, FDA decisions, earnings
- Spread Widening: Bid-ask spreads widen significantly during high-volatility periods
- Gap Risk: Large overnight gaps common on pre-market catalyst news
⚡ Expected Catalyst Timeline (12-18 Months)
| Expected Period | Catalyst Event | Typical Price Impact | Details |
|---|---|---|---|
| Q4 2025 / Q1 2026 | ATI-052 Phase 1 PoC | ±15-25% | Safety, dosing, early efficacy signal |
| Q4 2025 / Q1 2026 | ATI-045 Phase 2a Data | ±20-35% | Efficacy/safety readout in AD patients |
| Q2-Q3 2026 | ATI-2138 Phase 2b Data | ±25-40% | Critical for lead program; Phase 3 pathway |
| Q4 2025 | Q3 Earnings / 10-Q | ±5-10% | Financial update, burn rate confirmation |
*IMPORTANT DISCLAIMER: All catalyst dates are ESTIMATES based on standard clinical timelines. Actual readout dates may differ significantly (±3-6 months or more). These are NOT confirmed dates. Always verify with official company announcements and SEC filings.
? Market Opportunity & Competitive Landscape
Atopic Dermatitis Market Size: $10B+ global market with high unmet need for oral alternatives to biologics. Relapse rates post-biologic discontinuation create demand for other treatment options.
Key Competitors
- Dupilumab (Sanofi [finance:Sanofi S.A.]) — JAK/STAT pathway, biologic
- Abrocitinib (Pfizer [finance:Pfizer Inc.]) — JAK inhibitor, oral
- Ruxolitinib Cream (Incyte [finance:Incyte Corporation]) — JAK topical
- Tralokinumab (LEO Pharma) — IL-13 antibody, biologic
- Baricitinib (Eli Lilly [finance:Eli Lilly and Company]) — JAK1/JAK2, oral
Aclaris Positioning
- ITK/JAK3 Selectivity: Potentially lower systemic JAK inhibition vs. pan-JAK competitors
- Oral Formulation: Patient preference vs. injectable biologics
- Bispecific Approach: Dual-pathway blockade (TSLP + IL-4R) for differentiation
⚠️ Risk Assessment
CLINICAL EXECUTION: Multiple Phase 1b-2 programs carry inherent failure risk. Negative efficacy/safety data would trigger stock re-valuation downward.
COMPETITIVE CROWDING: Large pharma competitors with approved AD products; market likely to consolidate around 1-2 winners. Late entrants face disadvantage.
ACCELERATED BURN: $4M/month burn rate (2025) is rising. Capital raise may be required before Phase 3 = further dilution to shareholders.
SHARE DILUTION: 53% dilution 2023-2025 from $80M placement. Additional dilution likely if funding needed before clinical approvals.
CLINICAL TIMELINE RISK: Estimated catalyst dates may slip 6+ months; Phase 3 initiation depends on Phase 2 data strength.
? Sommario Esecutivo
Aclaris Therapeutics (NASDAQ: ACRS) è azienda biofarmaceutica fase clinica focalizzata dermatologia sviluppando terapie immunologiche per malattie pelle infiammatorie. Società ha transizionato da fase commerciale a fase clinica, costruendo pipeline con tre programmi in stage Phase 1b-2.
HIGHLIGHTS CHIAVE (Q3 2025 – SEC VERIFIED):
- ✓ Total Liquidity: $167.2M (Q3 2025) — runway fino a 2027-2028
- ✓ Debito Minimo: $2.24M debito totale vs. $167.2M liquidità = net cash forte
- ✓ Burn Rate: ~$4M/mese (accelerating 2025); runway ~42 mesi
- ✓ Pipeline: ATI-2138 (Phase 2a+ positivo), ATI-052 (Phase 1b cleared), ATI-045 (Phase 2 active)
- ✓ Catalyst Recenti: ATI-2138 Phase 2a positivo (Lug 2025), ATI-052 IND cleared (Apr 2025)
- ⚠ Rischi: 53% diluizione share dal 2023, R&D burn accelerato, rischio esecuzione clinica
? Posizione Finanziaria (Q3 2025 – SEC Verificata)
Update: Q3 2025 10-Q depositato 6 Novembre 2025 mostra posizione aggiornata con $167.2M liquidity totale (down da $203.9M dic 2024). Burn rate accelerando dovuto a programmi R&D espansi.
Bilancio Q3 2025
| Metrica | Q3 2025 | Dic 2024 | Cambio |
|---|---|---|---|
| Cassa & Equivalenti | $25.3M | $24.6M | +$0.7M |
| Titoli Short-term | $70.7M | $89.0M | -$18.3M |
| Titoli Long-term | $71.3M | $90.3M | -$19.0M |
| TOTAL LIQUIDITY | $167.2M | $203.9M | -$36.7M (-18%) |
| Debito Totale | $2.24M | $0.0M | Minimo (leases) |
| Patrimonio Azionisti | $120.1M | $155.6M | -23% |
UPDATES CRITICI:
- Burn Rate Accelerando: $36.7M consumati in 9 mesi (2025) = ~$4M/mese
- Spesa R&D Up 35%: $36.1M YTD 2025 vs. $24.6M anno precedente
- Diluizione Equity: 108.3M share (ott 2025) vs. 70.9M (dic 2023) = 53% diluizione
- Runway Stimato: $167.2M ÷ $4M/mese = ~42 mesi (fine 2028)
? Pipeline Clinica & Catalyst Prossimi
Program Lead: ATI-2138 (Inibitore Orale ITK/JAK3)
? PIÙ AVANZATO – Dermatite Atopica
Stato: Phase 2a (DATI POSITIVI – 29 Luglio 2025)
Meccanismo: Selettivo inibitore orale covalente ITK/JAK3
Dati: 14-pazienti Phase 2a open-label tollerabilità favorevole, NESSUN evento avverso serio
Catalyst Prossimo: Phase 2b interim/top-line Q2-Q3 2026 (±6 mesi stima)
Secondario: ATI-052 (Anticorpo Bispecifico)
? IND CLEARED – Condizioni Immune-mediate
Stato: Phase 1a/1b (IND Cleared 22 Aprile 2025)
Meccanismo: Anticorpo bispecifico anti-TSLP/IL-4R (dual-pathway)
Design: Randomizzato, doppio-cieco, placebo-controllato; SAD/MAD + PoC
Catalyst Prossimo: Phase 1a/1b PoC Q4 2025 o Q1 2026 (±3 mesi stima)
Terziario: ATI-045 (Bosakitug)
? PHASE 2 ONGOING – Dermatite Atopica
Stato: Phase 2 iniziato 2 Giugno 2025
Meccanismo: Anticorpo monoclonale TSLP (licensed da Biosion)
Design: Randomizzato, doppio-cieco, placebo-controllato Phase 2
Catalyst Prossimo: Phase 2a top-line Q4 2025 o Q1 2026 (±3 mesi stima)
? Analisi Tecnica – Livelli Critici
Zone Support (Punti Pausa Storici)
? PRIMO SUPPORT: $2.00-2.25
Osservazione: Zona support storica dove vendite esaurite. Volume storicamente aumenta. Prossimo support se rotto: $1.50-1.75
? SECONDO SUPPORT: $1.50-1.75
Osservazione: Zona low 52-settimane. Strong volume cluster. Soglia critica—rotture suggeriscono liquidazione o catalyst negativo
Zone Resistance (Punti Vendita Storici)
? PRIMO RESISTANCE: $4.50-5.00
Osservazione: Resistance storica dove vendite emergevano. Tocchi multipli senza breakout. Breakout sopra $5.00 segnalerebbe shift bullish
? SECONDO RESISTANCE: $6.50-7.50
Osservazione: Upper band da 2024. Heavy volume storico. Breakout richiederebbe catalyst bullish significativo (Phase 2 positivo)
Volatilità & Osservazioni Volume
- Swing Intraday: 5-15% comune su giorni neutral; 20-40% su catalyst days
- Volume Media: 1-2M share daily; spike 5-10M+ su catalyst
- Pattern Volume: Spike 2-3x average su annunci clinici, decisioni FDA
- Spread Widening: Bid-ask spread si allargano significativamente durante alta-volatilità
- Gap Risk: Gap overnight grandi su pre-market catalyst news
⚠️ Valutazione Rischi
ESECUZIONE CLINICA: Multipli programmi Phase 1b-2 comportano rischio fallimento intrinseco. Dati negativi triggereranno stock re-valuation downward.
COMPETIZIONE AFFOLLATA: Competitori big pharma con prodotti AD approvati; mercato probabilmente consolidarsi attorno 1-2 vincitori.
BURN ACCELERATO: $4M/mese burn rate (2025) in aumento. Capital raise probabile prima Phase 3 = ulteriore diluizione shareholders.
DILUIZIONE SHARE: 53% diluizione 2023-2025 da placement $80M. Ulteriore diluizione probabile se finanziamento necessario.
TIMING CLINICO: Date catalyst stimate possono slittare 6+ mesi; Phase 3 dipende da forza dati Phase 2.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


