DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
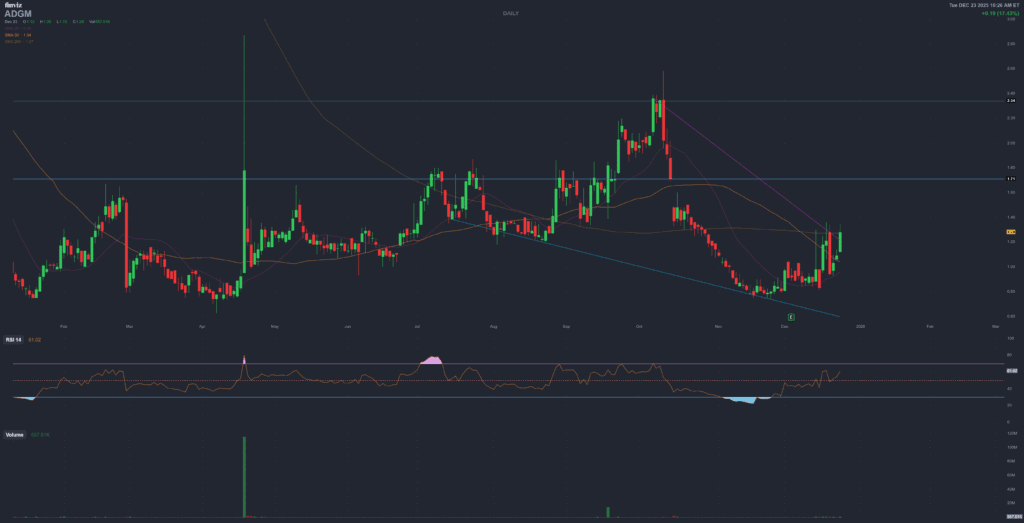
Adagio Medical Holdings (ADGM) – From CE Marks and Breakthrough Status to a 90% Drawdown
Deep dive on Adagio Medical Holdings, Inc. (NASDAQ: ADGM), a medtech company focused on ultra-low temperature cryoablation for cardiac arrhythmias. We walk through its CE Mark approvals, FDA Breakthrough Device designation, pivotal FULCRUM-VT trial, balance sheet stress and why the stock has been crushed despite apparently “good news”. Data checked against SEC filings, company PRs and major data providers as of December 23, 2025.
Ticker ADGM (NASDAQ)
Sector Medtech – Cardiac Ablation
Stage Commercial / Pivotal IDE (VT, AF)
Market status Post-SPAC, high leverage, high volatility
Key 2024–2025 milestones vs stock price collapse
In the last two years Adagio has ticked several boxes that, in teoria, should have been bullish:
- CE Mark approvals for its ultra-low temperature cryoablation systems: iCLAS for atrial fibrillation/atrial flutter (2020) and vCLAS VT system for monomorphic ventricular tachycardia (March 2024).
- FDA IDE and Breakthrough Device designation for the vCLAS VT system (IDE in 2024, Breakthrough Device in April 2025).
- FULCRUM-VT pivotal IDE trial fully enrolled (206 VT patients) as of October 1, 2025, with results intended to support a US PMA submission.
- Private placement up to $50M in October 2025, with $19M upfront and up to $31M more upon warrant exercises, to fund FDA submission and next-gen catheters.
Yet the stock has gone from around $11 in mid-2024 to roughly $1 today – a drawdown of over 90%. After a brief squeeze on the Breakthrough Device news (one day +130% intraday), the broader trend has been down, driven by heavy losses, negative equity, large convertible notes and repeated dilutions. This report focuses exactly on that disconnect: solid technical milestones vs brutal market reaction.
1. Strategic investment thesis – what ADGM really is
Adagio Medical is not a biotech but a medical device company developing catheter-based ultra-low temperature cryoablation (ULTC) systems for cardiac arrhythmias: atrial fibrillation (AF), atrial flutter and ventricular tachycardia (VT). Its technology uses near-critical nitrogen to reach temperatures as low as −196°C in a linear catheter system, aiming to create rapid, continuous, transmural lesions.
The core products and programs:
- iCLAS: ultra-low-temperature atrial catheter system for AF and atrial flutter. It has had CE Mark approval since 2020 and has generated European study data suggesting good acute success and one-year arrhythmia-free rates in persistent AF cohorts.
- vCLAS ULTC VT system: ventricular catheter and console for monomorphic VT in structural heart disease. CE Mark approved March 2024, with the FULCRUM-VT pivotal trial in the US and Canada designed to support a PMA. In April 2025 vCLAS received FDA Breakthrough Device designation.
- FULCRUM-VT pivotal IDE trial: 206 VT patients enrolled across ~20 sites in North America, with the goal of demonstrating safety and efficacy versus historical standards and supporting a PMA filing. Enrollment was completed on October 1, 2025.
On paper, this positions Adagio as a pure play on a large and fast-growing market: catheter ablation for arrhythmias, where big players (Medtronic, J&J/Biosense Webster, Boston Scientific) are investing heavily in cryoballoon and pulsed field ablation (PFA).
The problem – and the reason the stock has been “sent down” despite CE marks and Breakthrough status – is that Adagio came public via SPAC with a heavy capital structure, minimal revenue (annual sales well below $1M), high operating losses, significant convertible notes and the need for repeated equity financings in a hostile small-cap medtech market. The fundamental risk is not whether the catheters work at all – they clearly do something – but whether the company can reach US approval and meaningful sales before the balance sheet runs out of road.
3. Technology – ultra-low temperature cryoablation (ULTC) and vCLAS/iCLAS
Classic cryoballoon systems for AF (for example Medtronic and Boston Scientific) freeze tissue around −80°C. Adagio’s ultra-low temperature system takes this much further: by using near-critical nitrogen as the refrigerant, its catheters can deliver temperatures down to roughly −196°C along a 9F linear catheter. The idea is to:
- create continuous, transmural lesions in fewer applications;
- shorten procedure times and reduce the need for multiple passes;
- offer precise lesion sets for both atrial and ventricular arrhythmias.
The platform consists essentially of:
- a console controlling near-critical nitrogen flow and monitoring catheter temperature and system parameters;
- the iCLAS atrial catheters (linear, circular, One Shot+ variants) for AF and atrial flutter;
- the vCLAS ventricular catheter and VT console, designed to deliver linear lesions in the ventricle (endocardial and potentially epicardial) for monomorphic VT.
Clinical data presented in peer-reviewed journals and at EP congresses suggest that ULTC lesions are durable and can be created quickly, with acute success rates and 12-month arrhythmia-free survival in the same ballpark – and in some series arguably better – than existing technologies in difficult AF cohorts. For VT, Adagio has shown promising lesion durability and arrhythmia-free survival in CE-Mark studies, though data are still early compared with larger competitors.
In Europe, Adagio’s iCLAS system received CE Mark in 2020 for AF and atrial flutter, and in March 2024 the company obtained CE Mark for its ULTC system to treat monomorphic VT. The VT approval allows use of the updated console and vCLAS catheter and was followed by initial commercial use in select centres.
In the US, Adagio has:
- an IDE-approved pivotal VT trial (FULCRUM-VT), now fully enrolled, for the vCLAS system;
- FDA Breakthrough Device designation for vCLAS (April 2025), granting priority review and more intensive interaction during the PMA process;
- a history of iCLAS IDE work in AF, with earlier generations of the system evaluated in US trials.
Short version: the technology is real, differentiated and has already cleared meaningful regulatory hurdles (CE Marks, IDE, Breakthrough). What it does not have yet is an approved US product generating material revenue.
4. Regulatory status, clinical timelines and what’s actually “approved”
A key point for investors and traders: not all “approvals” are created equal.
- CE Mark (EU) is a marketing authorization for Europe. Adagio’s atrial (iCLAS) and VT ULTC systems are approved for sale and use in the EU for AF/AFL and monomorphic VT, respectively.
- FDA Breakthrough Device designation for vCLAS is not an approval; it is a program that grants priority review and closer interaction during the US premarket process.
- IDE approval allows Adagio to run the pivotal FULCRUM-VT trial in the US, but does not allow commercial sales yet. The FULCRUM-VT results are intended to support a future PMA (Premarket Approval) submission.
The current status:
- iCLAS and VT ULTC systems are approved and CE-marked in Europe, but the company has paused its broader EU commercial rollout and reduced headcount in early 2025 to conserve cash and focus on the US pivotal trial.
- In the US, no product is yet fully approved for commercial VT or AF ablation. FULCRUM-VT has finished enrollment; patients still need follow-up and the company needs to analyse safety and efficacy, then submit a PMA dossier and get through FDA review.
This is the heart of your original observation: “even though approved, it was sent down.” The nuance is: Adagio does indeed have CE-Mark approvals and a Breakthrough Device designation, but it does not yet have a US PMA, and EU commercialization was deliberately slowed to avoid burning cash without a fully-scaled commercial engine. The market has therefore treated these approvals as necessary but far from sufficient to justify the valuation that the stock briefly enjoyed post-SPAC.
5. Financial deep dive – why the balance sheet scares the market
The Q3 2025 10-Q (period ended September 30, 2025) and the FY 2024 10-K show a familiar medtech pattern, but at a scale that is aggressive for a micro-cap:
- Cash and equivalents: $4.7M as of Sept 30, 2025, down from $20.6M at Dec 31, 2024. This reflects operating burn and partial repayment of liabilities, before the October private placement.
- Total assets: $31.5M, including ~$7M of intangibles and ~$14M of goodwill from the SPAC/business-combination accounting.
-
Total liabilities: $32.9M, including:
- ~$21.2M of convertible notes payable (net);
- lease liabilities;
- warrant liabilities and deferred tax liabilities.
- Stockholders’ equity: negative (~$1.3M deficit), i.e. liabilities exceed assets.
- 9M 2025 net loss: about $21.8M, on minimal revenue (low six figures). The loss includes non-cash fair-value swings on convertible notes and warrants, but the underlying cash burn is still high.
The October 2025 private placement, led by Perceptive and other healthcare-focused investors, provides roughly $19M upfront plus the potential for another ~$31M if the attached warrants are fully exercised. That is a meaningful lifeline: it can fund completion of FULCRUM-VT, preparation of the PMA package and further development of next-gen catheters. However:
- it comes with substantial dilution (new shares and warrants on top of the SPAC structure);
- the company still carries a large block of convertible debt, which brings covenant risk if performance or cash levels deteriorate;
- at recent burn rates, even a $19M injection does not guarantee years of runway – especially if the company needs to scale a commercial organisation around a future US approval.
The 10-K is explicit: Adagio will likely need additional capital in the future to support commercialization, manufacturing scale-up and expansion into new markets. The Board notes that failure to access capital on acceptable terms could force it to slow development, cut back operations or explore strategic alternatives (M&A, asset sale, restructuring).
6. Stock performance – from SPAC euphoria to post-approval hangover
ADGM’s price action makes more sense if you track both the medtech story and the SPAC / post-SPAC context:
- August 2024: the business combination with ARYA Sciences Acquisition Corp IV closes, and Adagio Medical Holdings starts trading as ADGM on Nasdaq. The stock briefly trades around $9–11 as investors bet on ULTC cryoablation as a differentiated arrhythmia platform.
- 2024–early 2025: the company announces CE-Mark approval for the VT ULTC system, launches initial EU commercial activity and begins FULCRUM-VT. At the same time, macro conditions for small medtech deteriorate, rates are high, and investors grow wary of loss-making SPAC names. Despite positive technical news, ADGM slowly bleeds lower.
- February–March 2025: Adagio announces a restructuring and workforce reduction, pausing a broader European launch of its cryo catheters to focus on the US pivotal VT trial and to conserve cash. The market reads this as both prudent and as confirmation that near-term revenue traction in Europe is modest at best. The stock continues to trend down.
- April 17, 2025: FDA Breakthrough Device designation for vCLAS sparks a violent short-squeeze: ADGM roughly doubles intraday and retail attention explodes. Within days, though, the stock gives back a large part of the spike as reality sets in: Breakthrough status is positive but not a PMA approval, and the underlying financials remain fragile.
- Mid-2025: volatility remains extreme. On some days the stock gains 100%+, on others it drops 30%+ as traders rotate in and out. Meanwhile, the share price drifts steadily lower from the post-Breakthrough highs, reflecting dilution from additional equity/warrant deals and concerns around convertible debt.
- Late 2025: FULCRUM-VT enrollment completes; the October private placement is announced. Despite being fundamentally positive (cash in the door, clinical milestone), both events are followed by renewed pressure on the stock as investors focus on the cost of capital and the remaining execution risk. By December 2025, ADGM trades roughly 90% below its mid-2024 high.
In other words, the price collapse is not because “approvals are bad”, but because:
- approvals so far are primarily CE Marks and program designations, not US PMA approvals with meaningful revenue attached;
- the capital structure is heavy: large convertible notes, warrant overhang, negative equity, repeated financings below previous levels;
- the investor base is dominated by short-term and event-driven traders, not long-only medtech funds willing to sit through years of execution risk.
7. Analyst coverage, sentiment and how the Street sees ADGM
Publicly available data show:
- Limited formal coverage: one or a few specialist firms cover ADGM. Some services list an average 12-month target around $4 per share, with a mix of ratings that historically have included Buy calls but also at least one Sell.
- TTM numbers: trailing twelve-month revenue well under $1M, net loss upwards of $70M, and about 80 employees. On typical valuation metrics, the company screens as “very early stage” and “very cash-hungry”.
- Retail sentiment: Stocktwits and Reddit threads on ADGM often frame it as a “90%-off medtech lottery ticket” with references to the VT and AF opportunity and the $19M financing as reasons the story might survive. Many comments, però, are clearly speculative and focused on squeezes rather than deep due diligence.
The net result is that ADGM sits in an awkward corner of the market:
- too illiquid and early to attract broad long-only institutional ownership;
- too complex (devices, CE Marks, IDE, PMA, PFA competition) for casual traders to fully understand;
- attractive to event-driven and sentiment-driven accounts who amplify volatility around each press release.
8. Risk map and prospective scenarios
As of late 2025, ADGM is clearly high-risk, event-driven medtech. A non-exhaustive risk map:
Upside scenario – what would need to happen
- FULCRUM-VT generates strong VT outcomes versus historical comparators (reduction in VT episodes, acceptable safety), convincing FDA that ULTC adds meaningful value over existing options.
- Adagio secures either PMA approval for vCLAS and/or a strategically interesting partnership (for example, regional distribution or co-development) with a larger electro-physiology player.
- The October private placement and potential warrant exercises prove sufficient to bridge to major catalysts, reducing immediate going-concern concerns and shifting the narrative from “will they survive?” to “how fast can they grow?”.
- PFA and competing technologies expand the overall ablation market rather than completely displacing ULTC, leaving room for a niche but profitable VT/AF franchise for Adagio.
Downside scenario – how it can break
- FULCRUM-VT results are mixed or underwhelming (e.g., not clearly superior to existing VT ablation outcomes, or with a safety signal that worries regulators), reducing the probability of a clean PMA.
- The company struggles to access capital on acceptable terms once the October raise is consumed, leading to more aggressive dilution, covenant issues on the convertible notes, or in the worst case, forced restructuring or sale of assets.
- PFA and competing technologies become dominant in VT and AF faster than expected, making it harder for ULTC to find a differentiated, sustainable niche.
- Execution risk – on manufacturing scale-up, quality systems, reimbursement and sales-force build-out – proves too high for a lean organisation, even if regulatory hurdles are cleared.
Given these dynamics, the market is not “wrong” to discount the CE Marks and Breakthrough Device designation. Those milestones are real, but they exist alongside a balance sheet that has little room for error.
Adagio Medical is a good example of how regulatory wins and stock performance can diverge completely.
The ULTC platform is differentiated, the VT and AF opportunities are large, and the company has cleared a
surprisingly long list of regulatory and clinical hurdles for its size. At the same time, the capital
structure is heavy, losses are large relative to revenue, and even after a $19M private placement the company
still faces hard questions on runway and execution. This is not a “safe approval story” but a classic
event-driven micro-cap where outcomes can range from turnaround (if data and capital align) to further
dilution or worse if they do not.
Biotech / Medtech Catalyst Calendar
Stories like ADGM live and die on catalysts: interim analyses, pivotal trial readouts, PMA submissions, FDA panel dates and financing windows. To keep an overview of the main upcoming FDA decisions, clinical milestones and other binary events in the biotech/medtech space, you can use the dedicated Merlintrader page:
Biotech Catalyst Calendar – Merlintrader
Questo box è pensato come ultimo blocco del report; eventuali blocchi autore, partner o donazioni sono gestiti direttamente dal tema del sito.
Adagio Medical Holdings (ADGM) – CE Mark, Breakthrough FDA e crollo del 90%
Deep dive su Adagio Medical Holdings, Inc. (NASDAQ: ADGM), società medtech focalizzata sulla crioablazione a ultra-bassa temperatura per le aritmie cardiache. Vediamo CE Mark, Breakthrough Device Designation, studio pivotale FULCRUM-VT, numeri di bilancio e perché il titolo è stato affondato dal mercato nonostante le “buone notizie”. Dati verificati su filing SEC, comunicati ufficiali e principali provider al 23 dicembre 2025.
Ticker ADGM (NASDAQ)
Settore Medtech – Ablazione cardiaca
Stadio Commerciale EU / pivotale IDE USA
Stato di mercato Post-SPAC, leva elevata, alta volatilità
Milestone 2024–2025 vs crollo in borsa
Negli ultimi due anni Adagio ha centrato una serie di obiettivi che, sulla carta, sono tutti “green flag”:
- CE Mark per i sistemi di crioablazione ULTC: iCLAS per fibrillazione atriale/flutter (2020) e sistema vCLAS per tachicardia ventricolare monomorfa (marzo 2024).
- IDE FDA e Breakthrough Device designation per il sistema vCLAS (IDE nel 2024, Breakthrough ad aprile 2025).
- Studio pivotale FULCRUM-VT completamente arruolato (206 pazienti con VT) al 1° ottobre 2025, con risultati destinati a supportare la PMA negli USA.
- Private placement fino a 50M USD nell’ottobre 2025: circa 19M upfront e fino a ~31M aggiuntivi se i warrant verranno esercitati, per finanziare submission FDA e cateteri di nuova generazione.
Eppure il titolo è passato da circa 11 USD a metà 2024 a intorno a 1 USD oggi – drawdown oltre il 90%. Dopo un breve squeeze sulla notizia del Breakthrough Device (+130% intraday in una seduta), il trend di fondo è rimasto al ribasso, schiacciato da perdite pesanti, equity negativa, note convertibili importanti e diluizioni ripetute. Qui andiamo a vedere proprio questo mismatch: milestone regolatorie vs reazione durissima del mercato.
1. Tesi di investimento – che cos’è davvero ADGM
Adagio Medical è una società di dispositivi medici, non una biotech classica. Sviluppa sistemi di crioablazione a ultra-bassa temperatura (ULTC) per aritmie cardiache: fibrillazione atriale (AF), flutter atriale e tachicardia ventricolare (VT). La tecnologia utilizza azoto in stato “near-critical” per raggiungere temperature fino a circa −196°C su un catetere lineare 9F, con l’obiettivo di creare lesioni continue e transmural in modo rapido.
I prodotti e programmi principali:
- iCLAS: sistema atriale ULTC per AF e flutter atriale. Ha CE Mark dal 2020 e dati europei con buoni tassi di successo acuto e free-from-AF a 12 mesi in coorti di PsAF.
- vCLAS ULTC VT system: catetere e console ventricolare per VT monomorfa in cardiopatie strutturali. CE Mark approvato a marzo 2024, con studio pivotale FULCRUM-VT in USA/Canada pensato per la PMA. Ad aprile 2025 vCLAS ottiene la FDA Breakthrough Device designation.
- FULCRUM-VT: studio IDE pivotale VT, 206 pazienti arruolati in ~20 centri, i cui risultati serviranno per la richiesta di approvazione PMA negli Stati Uniti.
Sulla carta, Adagio è un’esposizione relativamente pura al mercato in crescita dell’ablazione per aritmie, in cui colossi come Medtronic, J&J/Biosense Webster e Boston Scientific stanno spingendo forte su cryoballoon e PFA (pulsed field ablation).
Il problema – e il motivo per cui il titolo è stato “mandato giù” nonostante le approvazioni – è che Adagio è arrivata in borsa via SPAC con una struttura di capitale pesante, ricavi praticamente inesistenti (meno di 1M USD annuo), perdite operative elevate, note convertibili importanti e bisogno ricorrente di aumenti di capitale in un contesto molto difficile per le small-cap medtech. Il rischio principale non è tanto “funziona il catetere?”, ma “arriva a monetizzarlo prima di finire benzina?”.
3. Tecnologia – crioablazione ULTC e sistemi vCLAS/iCLAS
I cryoballoon classici per AF lavorano intorno a −80°C. L’ULTC di Adagio spinge molto più in basso: usando azoto near-critical ottiene temperature intorno a −196°C su un catetere lineare 9F, con l’obiettivo di creare lesioni continue e transmural in meno applicazioni.
Il sistema comprende:
- una console che gestisce il flusso di azoto e monitora temperature e parametri;
- i cateteri atriali iCLAS per AF e flutter (varianti lineari, circolari, One Shot+);
- il catetere ventricolare vCLAS e la console VT per lesioni nel ventricolo (VT monomorfa).
I dati clinici presentati in letteratura e ai congressi mostrano che le lesioni ULTC sono durature e possono essere create rapidamente, con tassi di successo acuto e outcome a 12 mesi in linea – e in alcuni lavori potenzialmente migliori – rispetto alle piattaforme tradizionali, soprattutto in AF persistente. Per la VT, i dati CE Mark suggeriscono risultati promettenti, anche se ancora su numeri e follow-up limitati rispetto ai big del settore.
In Europa:
- iCLAS ha CE Mark dal 2020 per AF/AFL;
- il sistema ULTC per VT ha CE Mark da marzo 2024, con lancio iniziale in alcuni centri.
Negli Stati Uniti:
- Adagio ha un studio IDE pivotale VT (FULCRUM-VT) in corso – ora completamente arruolato;
- il sistema vCLAS ha Breakthrough Device designation dalla FDA (aprile 2025);
- c’è una storia di studi IDE su iCLAS in AF, con generazioni precedenti del sistema valutate in trial USA.
In sintesi: tecnologia reale, differenziata e già passata attraverso CE Mark, IDE e Breakthrough; ma manca ancora una vera approvazione FDA PMA e un flusso di ricavi significativo.
4. Stato regolatorio e cosa significa “approvata”
Vale la pena chiarire la parola “approvata”:
- CE Mark in UE è un’autorizzazione commerciale: i sistemi iCLAS e ULTC VT sono approvati per la vendita e l’uso in Europa per AF/AFL e VT monomorfa.
- La Breakthrough Device designation FDA per vCLAS non è una approvazione, ma un canale priorizzato per la revisione PMA e il dialogo con l’agenzia.
- L’IDE consente lo studio clinico (FULCRUM-VT), ma finché non si arriva alla PMA accettata non si può vendere il dispositivo sul mercato USA.
La situazione attuale:
- iCLAS e ULTC VT sono approvati in UE ma il management ha messo in pausa l’espansione commerciale europea e ha ridotto il personale per concentrare le risorse sullo studio pivotale USA;
- negli Stati Uniti non c’è ancora un prodotto approvato per la vendita; FULCRUM-VT è arruolato, ma servono follow-up, analisi dei dati, PMA e revisione FDA.
Quindi sì, Adagio ha sistemi “approvati” (CE Mark) e un vCLAS “promosso” a Breakthrough dalla FDA, ma il mercato guarda a dove arrivano ricavi e cash flow, non solo alle etichette regolatorie. Finché non c’è una PMA USA o una rampa commerciale europea visibile, le CE Mark restano milestone importanti ma non decisive per la sopravvivenza finanziaria.
5. Approfondimento numeri – perché il bilancio spaventa
Dal 10-Q Q3 2025 e dal 10-K 2024:
- Cassa ed equivalenti: 4,7M USD a fine settembre 2025 (contro 20,6M a fine 2024), prima del private placement di ottobre.
- Attività totali: 31,5M USD, inclusi ~7M di intangibili e ~14M di goodwill derivante dal deal SPAC.
-
Passività totali: 32,9M USD, con:
- ~21,2M USD di note convertibili;
- passività su lease, warrant liabilities e debiti fiscali differiti.
- Equity: negativa per circa 1,3M USD – le passività superano le attività.
- Perdita netta 9M 2025: ~21,8M USD, su ricavi ancora molto limitati (bassa sei cifre).
Il private placement di ottobre 2025 porta ~19M USD di cassa fresca e potenzialmente altri ~31M se i warrant verranno esercitati. È una boccata di ossigeno che:
- può finanziare completamento FULCRUM-VT, preparazione della PMA e sviluppo dei cateteri next-gen;
- però aggiunge ulteriore diluizione su una base già gonfia post-SPAC;
- non elimina il problema delle note convertibili né il rischio che serva ancora capitale in caso di ritardi o studio meno brillante del previsto.
Nel 10-K la società è chiara: servirà probabilmente altro capitale per sostenere lo scale-up commerciale, la produzione e l’espansione. In mancanza, il board non esclude di dover ridurre i piani, vendere asset o valutare opzioni strategiche più pesanti.
6. Andamento del titolo – dallo SPAC al post-approvazione “amaro”
Riassunta la cronologia:
- Estate 2024: chiude la business combination con ARYA Sciences Acquisition IV, Adagio sbarca al Nasdaq come ADGM. Il titolo gira intorno a 9–11 USD, sospinto dal tema ULTC e dalla narrativa “best in class VT/AF”.
- Fine 2024: CE Mark VT, lancio iniziale in Europa, avvio studio FULCRUM-VT. Ma il sentiment su SPAC/medtech è pessimo, i tassi alti e il mercato tratta molti nomi simili come “over-promised, under-funded”: ADGM inizia a scendere.
- Febbraio 2025: annuncio di ristrutturazione e taglio staff, pausa del rollout europeo per focalizzarsi su FULCRUM-VT e preservare cassa. Il mercato apprezza il focus, ma legge anche la mossa come segnale di ricavi UE modesti nel breve e struttura fragile.
- Aprile 2025: la Breakthrough Device designation della FDA fa esplodere il titolo (+100%/+130% intraday), con flusso retail e short squeeze. Nei giorni successivi però il grafico si sgonfia: la notizia è positiva ma non cambia il fatto che non c’è ancora PMA e la cassa scende.
- Estate–autunno 2025: continua la volatilità estrema. Giorni +80%, giorni −30%; in parallelo, il prezzo medio scende, complice la percezione di “equity ATM” per finanziare i trial e la presenza delle note convertibili.
- Ottobre 2025: completato l’arruolamento di FULCRUM-VT e annunciato il private placement fino a 50M USD. Fundamentalmente sono step positivi, ma il mercato guarda immediatamente a diluizione e leva, e il titolo resta in zona 1 USD, distante anni luce dai massimi post-SPAC.
La morale: il crollo di ADGM non arriva “nonostante le approvazioni”, ma perché:
- le approvazioni sono per ora CE Mark e designazioni, non una PMA USA “che stampa fatturato”;
- la struttura finanziaria è pesante (note convertibili, warrant, equity negativa, round ripetuti);
- il flottante è in gran parte in mano a trader event-driven, non a fondi long-only disposti ad aspettare anni.
7. Copertura analisti e sentiment
A livello di “Wall Street classica”:
- la copertura è limitata a poche case specializzate small-cap; alcuni service riportano un target medio a 12 mesi intorno a 4 USD per azione, con mix di rating che in passato includevano Buy ma anche Sell/Underperform;
- i numeri TTM indicano ricavi ben sotto 1M USD e perdite complessive oltre 70M USD, con circa 80 dipendenti: tipico profilo di medtech molto early-stage;
- su Stocktwits/Reddit ADGM viene spesso descritto come “lottery ticket” medtech post-crash, con molta enfasi su short interest, ULTC “innovativa” e possibilità di squeeze, meno su bilanci e covenants.
In sintesi, ADGM è:
- troppo piccola/volatile per entrare in modo significativo nei portafogli istituzionali generalisti;
- troppo complessa dal punto di vista tecnico (PFA vs ULTC, VT vs AF, IDE, PMA, CE) per il retail medio;
- perfetta, purtroppo, per chi cerca solo eventi binari e volatilità, non business visibili e lineari.
8. Mappa dei rischi e scenari prospettici
ADGM oggi è chiaramente un titolo event-driven, rischio molto alto. Alcuni assi principali:
Scenario positivo – cosa dovrebbe andare bene
- FULCRUM-VT mostra un segnale forte di riduzione VT e outcome migliori rispetto a dati storici, con sicurezza accettabile, convincendo la FDA per una PMA “smooth”.
- Adagio riesce a trasformare questa base in approvazione USA per vCLAS e/o in un accordo strategico con un grande player EP, ottenendo capitale e supporto commerciale.
- Il private placement da 19M + eventuali warrant bastano per arrivare ai prossimi step senza dover ricorrere a ulteriori aumenti devastanti.
- ULTC trova uno spazio preciso accanto a PFA, anziché essere completamente cannibalizzata, e Adagio riesce a ritagliarsi una nicchia profittevole in VT/AF complessa.
Scenario negativo – come può andare storto
- FULCRUM-VT esce con dati solo tiepidi (non inferiori, ma neppure chiaramente migliori) o con segnali di sicurezza che rendono la mia storia molto meno appetibile per FDA e per eventuali partner.
- Le finestre di mercato per nuovi finanziamenti si chiudono; la società è costretta a fare round a valutazioni ancora più basse, con forte diluizione, o a considerare ristrutturazioni / vendite asset.
- PFA prende il sopravvento anche in VT, riducendo drasticamente la percezione di valore del portafoglio ULTC.
- I rischi operativi tipici (produzione, qualità, rimborso, rete commerciale) si rivelano troppo pesanti per una struttura ridotta e molto indebitata.
In questo quadro, il mercato sconta pesantemente le CE Mark e il Breakthrough: le vede come condizioni necessarie, ma non sufficienti, finché non vede cash flow e bilancio stabilizzato.
Adagio Medical è un caso classico in cui approvazioni e performance in borsa vanno in direzioni opposte.
La tecnologia ULTC è reale e ben posizionata, le opportunità in VT/AF sono grandi e la società ha superato
già molti step regolatori per le sue dimensioni. Ma la struttura finanziaria è pesante, i ricavi ancora
trascurabili e anche dopo 19M USD di raccolta la domanda “come arrivate alla vera fase commerciale?” resta aperta.
Non è una storia “FDA approvata, quindi al sicuro”, ma un titolo micro-cap puramente event-driven, dove la forchetta
va da turnaround serio a ulteriore azzeramento in caso di mix sfavorevole fra dati e capitale.
Biotech / Medtech Catalyst Calendar
Storie come ADGM vivono e muoiono sulle date: analisi ad interim, dati finali, submission PMA, advisory committee, round di finanziamento. Per avere sempre una panoramica dei principali catalyst biotech/medtech in arrivo puoi usare la pagina dedicata di Merlintrader:
Biotech Catalyst Calendar – Merlintrader
Anche qui questo box è pensato come ultimo blocco del report; eventuali blocchi autore, partner o donazioni sono gestiti dal tema del sito.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


