DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
Generated: December 7, 2025
Innoviva Inc (NASDAQ: INVA)
Investment Analysis Report
Executive Summary
Innoviva Inc (NASDAQ: INVA) is a diversified holding company with a core royalty portfolio derived from respiratory assets in partnership with GlaxoSmithKline (GSK) and a growing commercial platform focused on critical care and infectious disease therapies through Innoviva Specialty Therapeutics (IST).
Key Highlights
Strengths
- FY 2024 Revenue Growth: +15.5% YoY, reaching $358.7M
- IST Explosive Sales Growth: Net sales +60.9% YoY, U.S. commercial momentum strong
- Fortress Balance Sheet: $305M cash + $86M receivables = $391M total liquidity
- Pipeline De-risked: ZEVTERA launch mid-2025, zoliflodacin NDA submission Q1 2025
- Forward P/E Attractive: 12.5x vs biotech peer average 20x+
Weaknesses
- Net Income Collapse: Fell from $179.7M (2023) to $23.4M (2024), -87%
- EPS Distortion: $0.36 (2024) vs $2.20 (2023), driven by equity investment losses
- GSK Concentration: 71.2% of revenue from GSK royalties creates dependency risk
- Patent Cliff Risk: BREO/ANORO exclusivity expires 2026-2030, potential $180-200M revenue loss
- Elevated Trailing P/E: 57.6x inflated by depressed EPS
Outlook: Neutral-Positive
Analyst consensus is MODERATE BUY with average price target of $37.60 (+81% upside). However, opinions are polarized (1 Strong Sell vs 5 Buy/Strong Buy), reflecting execution uncertainty and dependency on pipeline catalysts.
1. Company Profile and Business Model
1.1 Business Structure
Innoviva operates through two primary segments:
| Segment | % of Revenue | Key Assets | FY 2024 Revenue |
|---|---|---|---|
| Core Royalty Portfolio | 71.2% | GSK RELVAR/BREO/ANORO | $255.6M |
| IST Commercial Platform | 27.2% | GIAPREZA, XACDURO, XERAVA | $97.5M |
| License Revenue | 1.6% | Milestone payments | $19.5M |
1.2 Core Royalty Portfolio (GSK Partnership)
Three respiratory products approved globally:
- RELVAR®/BREO® ELLIPTA® – Long-acting beta-agonist/Inhaled corticosteroid for asthma and COPD
- ANORO® ELLIPTA® – Dual bronchodilator for COPD
FY 2024 Performance: $255.6M (+1.1% YoY). Q4 2024 was $66.0M vs $69.6M Q4 2023 (-5.2%), reflecting mature product dynamics.
1.3 Innoviva Specialty Therapeutics (IST) Division
Fast-growing platform for critical care and infectious disease:
| Product | Indication | FY 2024 Sales | Growth | Status |
|---|---|---|---|---|
| GIAPREZA® | Vasoconstrictor for shock | $53.4M | +40% | Star performer |
| XACDURO® | Antibacterial (A. baumannii) | $14.7M | New full-year | Key growth driver |
| XERAVA® | Antibiotic (intra-abdominal) | $12.8M | +33% | Moderate growth |
| Ex-U.S. Sales | Global expansion | $16.6M | Expanding | Geographic diversification |
1.4 Pipeline and Future Catalysts
Zoliflodacin (Phase 3 Complete)
- First-in-class oral, single-dose antibiotic for uncomplicated gonorrhea
- NDA submission expected early Q1 2025
- Partnership with GARDP (Global Antibiotic Research and Development Partnership)
- Market opportunity: >$1B in context of rising antimicrobial resistance
ZEVTERA® (Rights Acquisition Dec 2024)
- Cephalosporin (ceftobiprole) for advanced bacterial infections
- U.S. commercial launch expected mid-2025
- Fourth approved product for IST platform
- Acquired exclusive U.S. commercialization rights from Basilea Pharmaceutica
XACDURO® Geographic Expansion
- NMPA approval in China (May 2024)
- Designated preferred agent in IDSA 2024 guidelines for carbapenem-resistant Acinetobacter baumannii
- Expansion to Europe and other markets in progress
2. Financial Analysis (FY 2024)
2.1 Income Statement Overview
| Metric | FY 2024 | FY 2023 | FY 2022 | YoY Growth |
|---|---|---|---|---|
| Total Revenue | $358.7M | $310.5M | $331.3M | +15.5% |
| GSK Royalty Revenue | $255.6M | $252.7M | $269.6M | +1.1% |
| IST Net Product Sales | $97.5M | $60.6M | $53.0M | +60.9% |
| Gross Profit | $322.1M | $267.8M | $317.5M | +20.3% |
| Gross Margin | 89.8% | 86.3% | 95.8% | +3.5pp |
| Operating Income | $166.9M | $113.9M | -$36.9M | +46.5% |
| Operating Margin | 46.5% | 36.7% | -11.2% | +9.8pp |
| Net Income | $23.4M | $179.7M | $213.9M | -87.0% |
| Net Margin | 6.5% | 57.9% | 64.6% | -51.4pp |
| EPS (Diluted) | $0.36 | $2.20 | $2.37 | -83.6% |
2.2 Critical Analysis: Net Income Decline
The dramatic 87% drop in net income from $179.7M (2023) to $23.4M (2024) is primarily attributable to:
- Mark-to-Market Losses on Equity Investments: -$123.4M in FY 2024, mainly from Armata Pharmaceuticals and other portfolio holdings
- Increased Interest Expense: $22.2M (2024) vs $19.2M (2023)
- Impairment Charges and Other Non-Operating Items
Key Point: Excluding non-cash mark-to-market adjustments, operating performance is solid with 46.5% operating margin growth YoY.
2.3 Cash Position and Liquidity
| Item | Amount | Status |
|---|---|---|
| Cash & Cash Equivalents (Dec 31, 2024) | $305.0M | +57.7% YoY |
| Accounts Receivable | $86.0M | Strong collections |
| Total Available Liquidity | $391.0M | Fortress balance sheet |
| Operating Cash Flow (FY 2024) | $188.7M | +33.7% YoY |
| Equity Investments (Fair Value) | $501.5M | Volatility source |
3. Valuation Analysis
3.1 Current Valuation Metrics
Trailing P/E Ratio
57.6x
Elevated due to depressed EPS
Forward P/E (2025E)
12.5x
Consensus EPS $1.19
Forward P/E (2026E)
7.6x
Consensus EPS $2.23
EV/Revenue 2024
3.7x
In-line with peers
3.2 Analyst Price Targets and Consensus
| Metric | Value | Upside from $20.74 |
|---|---|---|
| Average Price Target | $37.60 | +81.3% |
| Median Price Target | $35.00 | +68.8% |
| High Price Target | $55.00 (Scotiabank) | +165.2% |
| Low Price Target | $17.00 (Goldman Sachs) | -18.0% |
3.3 Rating Distribution (December 2025)
| Rating | Count | Percentage |
|---|---|---|
| Strong Buy | 1 | 14% |
| Buy | 4 | 57% |
| Hold | 1 | 14% |
| Sell | 0 | 0% |
| Strong Sell | 1 | 14% |
| CONSENSUS | MODERATE BUY (71% Positive) | |
3.4 Bull vs Bear Case
Bull Case ($45-55 Price Target)
- Zoliflodacin FDA approval in 2025 – first single-dose oral treatment for gonorrhea
- IST platform accelerates with ZEVTERA launch mid-2025 (fourth approved product)
- Projected IST net sales exceed $120M in 2025 vs $97.5M in 2024
- GSK royalties remain stable despite competitive pressures
- M&A optionality with $391M in available liquidity
- Forward P/E only 12.5x on 2025 estimates – significant discount to peers
Bear Case ($17-22 Price Target)
- Zoliflodacin NDA rejection or delayed approval
- ZEVTERA commercial execution disappoints in crowded antibiotic market
- GSK royalty erosion accelerates faster than expected
- BREO/ANORO patent cliff (2026-2030) causes immediate revenue concerns
- Continued mark-to-market losses on equity investments pressure net income
- Competitive dynamics in critical care market intensify
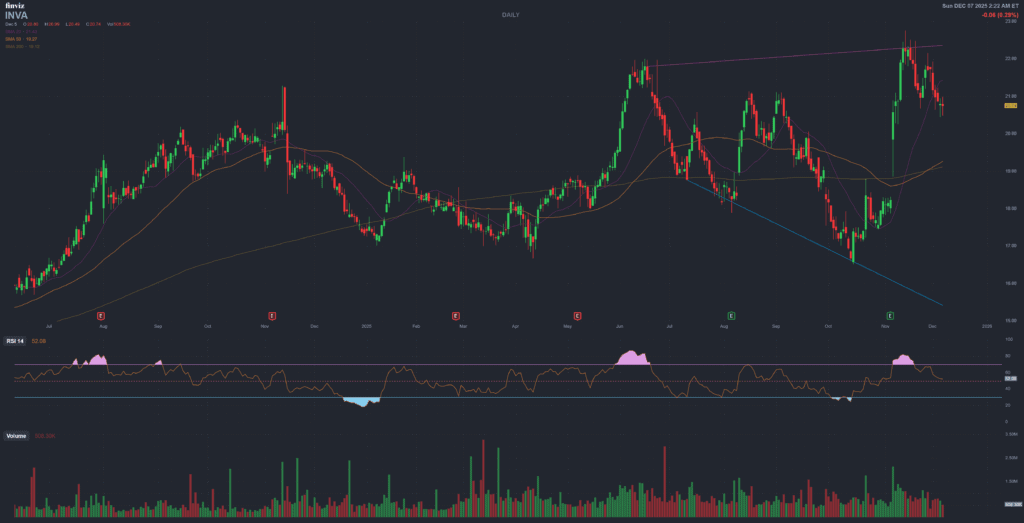
4. Technical Analysis (December 5, 2025)
4.1 Key Technical Indicators
| Indicator | Value | Interpretation | Signal |
|---|---|---|---|
| Current Price | $20.74 | Mid-range 52-week band | Neutral |
| 52-Week Range | $16.67 – $22.28 | Current near 52-week high | Neutral |
| MA 20-Day | $21.43 | Price below MA20 | Short-term weakness |
| MA 50-Day | $19.27 | Price above MA50 | Medium-term strength |
| RSI (14) | 25.41 | Oversold zone (<30) | Potential rebound signal |
| Volatility (20D) | 26.9% annualized | Average for biotech | Normal |
| Beta | 0.47 | Low market correlation | Lower downside in bear market |
4.2 Price Support/Resistance Levels
- Strong Resistance: $21.50 – $22.00 (MA20, 52-week high area)
- Key Support: $19.00 – $19.50 (MA50 zone)
- Weak Support: $18.00 (psychological level)
4.3 Price Performance
| Period | Return | Status |
|---|---|---|
| 1 Month | -0.58% | Flat |
| 3 Months | +7.07% | Positive |
| 6 Months | +2.98% | Slightly positive |
| YTD 2025 | +14.87% | Outperforming S&P |
5. Risk Assessment
5.1 Company-Specific Risks
High risk factors
1. Revenue Concentration Risk
- GSK royalties represent 71.2% of total revenue
- BREO/ANORO patent cliff 2026-2030 creates cliff risk
- Potential revenue loss: $180-200M annually post-genericization
- Mitigation: IST growth targets >$120M sales in 2025
2. Equity Investment Volatility
- $501.5M in fair value equity investments subject to mark-to-market
- -$123.4M unrealized losses in FY 2024 from Armata and others
- Significant impact on reported net income and P/E ratios
- Mitigation: Strong operational cash flow ($188.7M) supports balance sheet
3. Pipeline Execution Risk
- Zoliflodacin: FDA approval not guaranteed; NDA submission Q1 2025
- ZEVTERA: Commercial execution uncertain in competitive market
- Failure could delay IST growth targets
Medium risk factors
4. IST Product Competition
- GIAPREZA: Vasoconstrictor market growing but competitive
- XACDURO/XERAVA: Crowded antibiotic space with pricing pressure
- New entrants may erode market share
5. Regulatory and Reimbursement Risk
- Potential Medicare/Medicaid policy changes
- Hospital antibiotic pricing pressure
- Inflation Reduction Act (IRA) pricing mechanisms
6. M&A Integration Risk
- Track record: Entasis acquisition (2022), ZEVTERA rights (2024)
- Commercial execution complexity on new products
- Integration costs could pressure margins
Low risk factors
7. Balance Sheet Risk
- $305M cash + $86M receivables = $391M liquidity
- No debt maturity concerns in near term
- Strong operating cash flow ($188.7M FY 2024)
6. Catalysts and Key Events (2025)
Q1 2025
Zoliflodacin NDA Submission (Expected)
First-in-class oral single-dose antibiotic for uncomplicated gonorrhea. FDA review could extend into Q2-Q3. Approval could unlock significant commercial potential.
First-in-class oral single-dose antibiotic for uncomplicated gonorrhea. FDA review could extend into Q2-Q3. Approval could unlock significant commercial potential.
Q2 2025
ZEVTERA® U.S. Commercial Launch (Expected Mid-2025)
Cephalosporin for advanced bacterial infections. Test of IST commercial execution. Expected to add $20-30M revenue in 2025.
Cephalosporin for advanced bacterial infections. Test of IST commercial execution. Expected to add $20-30M revenue in 2025.
Q2 2025
Q1 2025 Earnings Release
Confirmation of IST momentum. Guidance update on FY 2025 target: IST net sales >$100M (+25% YoY).
Confirmation of IST momentum. Guidance update on FY 2025 target: IST net sales >$100M (+25% YoY).
H2 2025
FDA Review of Zoliflodacin (If Submitted Q1)
Potential Complete Response Letter (CRL) or approval. Major news driver for stock.
Potential Complete Response Letter (CRL) or approval. Major news driver for stock.
H2 2025
XACDURO® Geographic Expansion
New market approvals (Europe, other Asia markets). Expansion of addressable market.
New market approvals (Europe, other Asia markets). Expansion of addressable market.
2025-2026
M&A Potential
$391M available liquidity provides capacity for strategic acquisitions to address BREO/ANORO patent cliff.
$391M available liquidity provides capacity for strategic acquisitions to address BREO/ANORO patent cliff.
7. Investment Recommendation
Overall Rating
Price Targets
| Target | Value | Upside | Probability |
|---|---|---|---|
| 12-Month Base Case | $32 – $35 | +54% to +69% | 50% |
| Bull Case | $45 – $55 | +117% to +165% | 25% |
| Bear Case | $17 – $22 | -18% to +6% | 25% |
Entry Strategy by Investor Type
For Existing Shareholders
– Maintain position. Wait for catalysts (zoliflodacin NDA, ZEVTERA launch). Potential 12-month upside to $32-35.
For New Investors (Opportunistic)
– Consider entry on pullback toward $18-19 (MA50 support zone). RSI oversold at 25.41 suggests near-term bounce potential.
Risk Management
- Target Entry Price: $18.50 – $19.50
- Stop Loss: $17.00 (-15% from current)
- Position Size: 2-3% of portfolio (typical biotech risk sizing)
- Time Horizon: 12-18 months for catalysts to play out
8. Investment Thesis Summary
Why INVA is Attractive
- Forward Valuation Compelling: Forward P/E 12.5x vs biotech peer average 20x+. 2026 P/E only 7.6x on consensus EPS.
- IST Growth Acceleration: Net sales +60.9% YoY. U.S. commercial momentum strong. Target >$100M in 2025.
- Operating Performance Strong: 46.5% operating margin (+9.8pp YoY). Operating income up 46.5%.
- Pipeline De-risked: Two near-term catalysts: zoliflodacin NDA Q1 2025, ZEVTERA launch mid-2025.
- Balance Sheet Fortress: $391M liquidity provides M&A optionality and downside protection.
- Low Market Correlation: Beta 0.47 means lower volatility in broader market downturns.
Why Caution is Warranted
- GSK Dependency: 71.2% revenue concentration on single partner creates execution risk.
- Patent Cliff: BREO/ANORO exclusivity expires 2026-2030. Potential $180-200M revenue cliff.
- Net Income Volatility: Equity investment mark-to-market (-$123.4M FY 2024) distorts reported earnings.
- Pipeline Uncertainty: Zoliflodacin FDA approval not guaranteed. ZEVTERA commercial success not assured.
- Polarized Analyst Opinion: 1 Strong Sell vs 5 Buy ratings reflects genuine execution uncertainty.
9. Conclusion
Innoviva Inc represents a moderately attractive investment opportunity in the specialty pharma/biotech space with a balanced risk-reward profile.
The Bull Case: Forward-looking valuation is compelling (12.5x P/E). IST platform is inflecting higher with 60%+ sales growth. Two catalysts (zoliflodacin NDA, ZEVTERA launch) could unlock significant upside to $32-38 within 12 months. $391M cash provides acquisition capacity to mitigate BREO/ANORO patent cliff risk.
The Bear Case: Heavy GSK dependency (71% of revenue) with patent cliff 2026-2030. Net income distorted by equity investment volatility. Pipeline execution uncertain. Current elevated trading valuation (57.6x trailing P/E) reflects earnings depression that may not resolve quickly.
Best for: Value-oriented investors with 12-18 month horizon, biotech sector exposure, tolerance for 25-30% volatility, and interest in the intersection of specialty pharma and critical care markets.
Not suitable for: Dividend seekers (0% yield), growth-at-any-price investors requiring immediate momentum, or those with very low risk tolerance.
Action Items:
- Monitor Q1 2025 earnings for IST guidance confirmation
- Watch for zoliflodacin NDA submission announcement (expected early Q1)
- Track ZEVTERA launch progress (mid-2025 commercial update)
- Set price alerts at $18.50 (buy zone) and $22.50 (profit-taking area)
10. Market Sentiment Snapshot
This section summarizes public trader sentiment from major retail communities. These are opinions from non-professional traders and should be treated as anecdotal, not as research or advice.
Reddit (biotech and stock subreddits)
Overall bias: Mildly positive
Discussions focus on the pipeline optionality (zoliflodacin and ZEVTERA) and the royalty cash flow as a defensive anchor. Concerns frequently mention the patent cliff on BREO/ANORO and headline volatility around small-cap holdings.
Stocktwits
Overall bias: Swing-trading oriented
Many posts frame INVA as a technical swing between support at 19 USD and resistance near 22 USD, with traders closely watching catalysts and RSI oversold levels. Time horizon is typically days to weeks, not long-term fundamentals.
X (Twitter)
Overall bias: Mixed / event-driven
Commentary is split between event-driven accounts following FDA timelines and more cautious voices highlighting execution risk and equity portfolio volatility. Sentiment often shifts quickly around earnings and regulatory headlines.
Official Partners & Referral Resources
Sources and Data Attribution
- Directors Talk Interviews – Innoviva stock analysis (December 1, 2025)
- Biotechhealthx – Q3 2024 analysis (December 25, 2024)
- MarketBeat – Institutional activity (December 6, 2025)
- Yahoo Finance – Q4 & FY 2024 earnings report (February 26, 2025)
- MarketBeat – Analyst consensus forecasts (December 2025)
- Business Wire – Q4 2024 financial results (February 25, 2025)
- Stock Analysis – Price targets and analyst ratings
- SEC EDGAR – Official company filings (10-K, 10-Q)
- Company investor relations – Press releases and presentations
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.






