DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker

Merlintrader Trading Pub
Biotech catalyst news and analysis. FDA PDUFA tracker
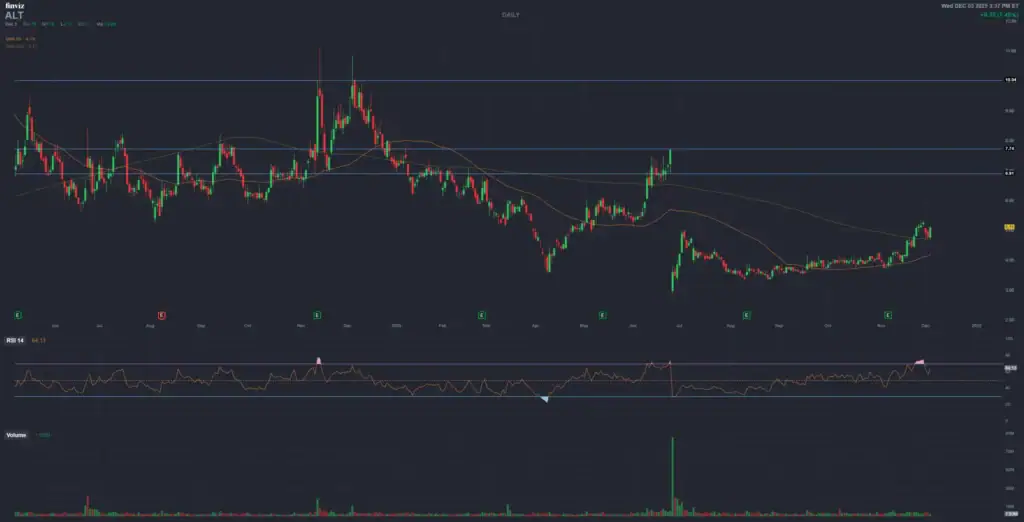
Altimmune (ALT)
Pemvidutide MASH Breakthrough and CEO Catalyst: Multi-Year Inflection Point
Positive momentum — updated December 3, 2025
Strong and active bullish discussions on StockTwits
Today: +7.5 percent — Trading around 5.12 USD
NASDAQ: ALT | Pemvidutide (Dual GLP-1/Glucagon Agonist) | Street targets generally in the mid-teens to high-twenties range
Executive Summary — A Positive Turning Point
Altimmune is entering a pivotal phase with several bullish drivers converging through Q4 2025 and into 2026:
- CEO Transition (January 1, 2026): Jerry Durso (former Intercept CEO, long career at Sanofi) becomes CEO. His background aligns strongly with commercialization, reimbursement strategy, and liver/metabolic drug launches.
- IMPACT Phase 2b MASH: At 24 weeks, MASH resolution without fibrosis worsening reached 59.1 percent (1.2 mg) and 52.1 percent (1.8 mg) vs 19.1 percent placebo; weight loss of 5.0–6.2 percent; liver fat reduction of 58–62.8 percent and meaningful fibrosis signals using AI-assisted analysis.
- Key upcoming milestones: 48-week data (Q4 2025), End-of-Phase 2 FDA meeting (Q4 2025), and final Phase 3 trial design for MASH.
- Financial position: Approximately 210.8 million USD in cash (Q3 2025), burn of 20–22 million per quarter, plus a 125 million USD Hercules credit line. Cash runway estimated at more than two years.
- Addressable market: MASH alone represents a multibillion-dollar market. The dual GLP-1/glucagon mechanism positions pemvidutide also for weight management and cardiometabolic indications.
1. The CEO Catalyst: Jerry Durso
Why This CEO Change Matters
Community Sentiment on the New CEO
Most active traders interpret the leadership change as follows:
Durso is seen as a serious, execution-focused leader with significant experience in liver disease and commercial strategy.
His background at Intercept and network built at Sanofi fuel speculation about potential partnerships.
Jerry Durso — Background (Verified Information)
| Item | Details |
|---|---|
| Industry experience | More than 30 years in life sciences, focused on commercial strategy, metabolic franchises and rare diseases. |
| Most recent role | CEO of Intercept Pharmaceuticals until its acquisition by Alfasigma. |
| Liver disease track record | Hands-on experience with PBC, regulatory complexities, pricing and market access. |
| Career at Sanofi | Over 20 years in senior commercial roles, including Head of U.S. Commercial Operations. |
| Entry into Altimmune | Joined Board in 2025, became Chairman and later CEO effective January 1, 2026. |
Verified data: Information sourced from Altimmune’s official CEO Transition announcement and public Intercept/Sanofi biographies.
2. Pemvidutide MASH Program — IMPACT Phase 2b Results
2.1 Summary of the 24-Week Results
MASH Resolution (24w)
59 percent (1.2 mg)
Liver Fat Reduction
58–63 percent
Average Weight Loss
5.0–6.2 percent
Fibrosis Reduction (AI, ≥60 percent)
30.6 percent (1.8 mg)
| Endpoint (24 weeks) | Pemvidutide 1.2 mg | Pemvidutide 1.8 mg | Placebo |
|---|---|---|---|
| MASH Resolution without Fibrosis Worsening | 59.1 percent | 52.1 percent | 19.1 percent |
| Fibrosis Improvement without MASH Worsening | 31.8 percent | 34.5 percent | 25.9 percent |
| Liver Fat Reduction (MRI-PDFF) | 58.0 percent | 62.8 percent | 16.2 percent |
| Weight Loss | -5.0 percent | -6.2 percent | -1.0 percent |
| Participants ≥60 percent Fibrosis Reduction (AI) | 11.9 percent | 30.6 percent | 8.2 percent |
Data sourced from Altimmune’s official IMPACT Phase 2b topline release (June 2025).
2.2 Clinical Relevance vs Competitors
Key Observations
- High MASH resolution: 52–59 percent places pemvidutide at the upper end of industry outcomes for 24 weeks.
- Fibrosis signals: Primary fibrosis endpoint not statistically significant, but AI-based analysis shows strong differentiation (30.6 vs 8.2 percent).
- Dual liver + metabolic profile: Strong liver fat reduction, weight loss and NIT improvements.
- Safety and tolerability: Very low discontinuation rates, no drug-related SAEs.
2.3 Upcoming MASH Catalysts
| Catalyst | Estimated Timing | Impact |
|---|---|---|
| 48-Week IMPACT Data | Q4 2025 | Durability of MASH resolution; extended fibrosis data; expanded safety window. |
| FDA End-of-Phase 2 Meeting | Q4 2025 | Alignment on Phase 3 structure; key to reduce regulatory uncertainty. |
| Phase 3 Initiation | 2026 | Potentially major sentiment driver depending on design and timelines. |
3. Financial Position: Cash & Runway
Cash & Equivalents (Q3 2025)
210.8M USD
Estimated Quarterly Burn
20–22M
Hercules Credit Facility
125M USD
Estimated Runway
Over 2 Years
| Metric | Q3 2025 | Approx. Q3 2024 |
|---|---|---|
| Cash & Equivalents | 210.8M USD | Approx. 132M USD |
| Net Loss (Quarterly) | Approx. -19M USD | -15M USD |
| R&D Spend | Approx. 15M USD | 12M USD |
| Total Assets | Approx. 265M USD | Approx. 150M USD |
Financial Strength Consideration
- Cash + credit facility provides enough buffer for Phase 3 initiation and major clinical milestones.
- A more aggressive strategy may require future financing, depending on clinical expansion.
Verified data: Figures sourced from Altimmune’s Q3 2025 earnings release and Form 10-Q.
4. Market Performance and Analyst Consensus
4.1 Market Snapshot (December 3, 2025)
| Metric | Indicative Value |
|---|---|
| Current Price | Approx. 5.12 USD (+7–8 percent on the day) |
| 52-Week Range | 2.90 – 10.88 USD |
| Market Cap | Approx. 530–540M USD |
| Volume Today | 1.8–1.9M shares |
| YTD Performance | Weak, with significant drawdown from yearly highs |
4.2 Analyst Consensus and Price Targets
| Firm | Rating | Target | Upside vs 5.12 USD |
|---|---|---|---|
| Piper Sandler | Overweight / Buy | Approx. 25 USD | Approx. +380 percent |
| Jefferies | Buy | High-twenties | Approx. +400 percent |
| H.C. Wainwright | Buy | Approx. 12 USD | Approx. +130 percent |
| Stifel Nicolaus | Buy | Approx. 18 USD | Approx. +250 percent |
| Other brokers | Buy / Outperform | Mid-teens to low-twenties | Significant re-rating potential |
| Consensus Range | Mid-teens / low-twenties | Large theoretical upside | |
Takeaway from Sell-Side Analysts
- Analysts remain broadly bullish, but the stock trades at a discount due to execution concerns.
- If Phase 3 confirms the Phase 2b profile, valuation could move sharply higher.
5. Trader Sentiment & Online Discussion
Common Themes Among Active Traders
Most frequent interpretations include:
Many see CEO Durso + IMPACT results + FDA meeting as aligned catalysts indicating confidence from the Board.
Partnership speculation remains elevated due to Durso’s network at Sanofi.
Pemvidutide is increasingly viewed as a dual-use asset (MASH + weight), expanding market potential.
In short: sentiment is constructive but aware of clinical and regulatory risks.
6. Valuation Framework: Bull, Base and Bear
Bull Case
20 – 28 USD
Assumptions: Strong Phase 3 confirming MASH resolution and fibrosis improvement; accelerated approval potential; partnership or acquisition; billion-plus sales potential.
Base Case
12 – 18 USD
Assumptions: Phase 3 positive but with mixed signals; approval achieved; moderate sales ramp.
Bear Case
2 – 6 USD
Assumptions: Phase 3 fails; safety issues; FDA requests further studies; dilution likely.
7. Key Catalysts Ahead (Q4 2025 – 2026)
Summary of Catalysts
- Q4 2025: IMPACT 48-week data (durability, fibrosis, safety).
- Q4 2025: FDA End-of-Phase 2 meeting.
- January 1, 2026: Durso becomes CEO.
- 2026: Phase 3 initiation and pipeline updates.
8. Disclaimer & Key Risk Factors
Important Disclaimer
This report is for educational and informational purposes only. It is not investment advice and does not constitute a recommendation to buy or sell any security.
- Biotech risk: Clinical-stage biotech companies carry very high inherent risk.
- Clinical and regulatory risk: Positive Phase 2b results do not guarantee Phase 3 success.
- Competitive risk: MASH is a highly competitive space.
- Dilution risk: Additional financing remains possible.
- Market data variability: Price, volumes and market cap refer to December 3, 2025.
9. Sources & Useful Links
- Altimmune — IMPACT Phase 2b topline results (June 2025).
- Altimmune — Q3 2025 financial results and SEC filings.
- Altimmune — CEO Transition announcement.
- ALT on Finviz
- ALT page on Seeking Alpha
- ALT on StockTwits
- Altimmune official website
Support the Merlintrader trading Blog
If this report is helpful and you want to support the site remaining free for everyone, you can contribute via PayPal.
Scanner for active traders

Try ChartsWatcher free, then unlock 10% OFF with SAVE10
ChartsWatcher is a real-time scanner for momentum traders: fast movers, unusual volume and rotations — so you can focus on the few tickers that matter right now, instead of watching hundreds of charts.
Start with the free version. When you upgrade, use SAVE10 for 10% OFF your first paid period.
Start free – then use SAVE10
No credit card required to start. Apply SAVE10 when upgrading.
Recommended platform

One platform. All your brokers.
Medved Trader connects multiple brokers in one workspace, with pro charts, hotkeys and fast execution — without changing your broker accounts.
A single cockpit for positions, Level II and multi-broker order routing, built for active day & swing traders.
Get 1 Month Free ➔
Multi-broker workflow + customizable layouts in one platform.


