DISCLAIMER — Not financial advice. Educational content only, not an offer or solicitation to buy or sell any security. Biotech and small/mid-cap stocks are highly speculative and volatile and can result in a partial or total loss of capital. Do your own research and consult a licensed advisor where appropriate. / Contenuti a solo scopo informativo e didattico, non costituiscono consulenza finanziaria né offerta o sollecitazione al pubblico risparmio ai sensi delle normative CONSOB e SEC. Le azioni biotech e le small/mid cap sono strumenti altamente speculativi e volatili e possono comportare la perdita parziale o totale del capitale investito. Si raccomanda di effettuare sempre le proprie ricerche e, se necessario, di rivolgersi a un consulente abilitato.
Investment ThesisBull vs Bear
Bull thesis (what can go right)
- FDA approval secured in August 2024 for relapsed/refractory Stage I–III CTCL after at least one prior systemic therapy, with orphan designation and potential multi-year exclusivity protections.
- Approximately 8.27 million dollars of LYMPHIR inventory on the balance sheet, ready to be monetized as commercialization ramps.
- Compelling clinical efficacy: around 36% objective response rate with roughly 84% of evaluable patients showing meaningful tumor burden reduction in the pivotal study.
- Relatively rapid median time to response (about 1.4 months), which helps both patients and prescribers quickly evaluate benefit.
- Limited direct competition in an initial CTCL market estimated above 400 million dollars, with management guiding to a market that could expand toward the one-billion range over the next decade if broader indications materialize.
- Capillary leak syndrome (CLS) risk is meaningful but supported by boxed warnings, monitoring guidance and growing real-world experience.
- Majority ownership by Citius Pharmaceuticals provides strategic and financial backing, including funding support and corporate infrastructure.
- Q4 2025 U.S. commercial launch with a dedicated specialty distribution network and early steps toward international access agreements.
Bear thesis (what can go wrong)
- As of the last audited annual report, cash and cash equivalents at the CTOR level were essentially zero, with an annual net loss above 21 million dollars and operating expenses exceeding 20 million dollars.
- Commercialization is starting from a standing start: no revenue at the time of that filing, but full commercial costs beginning to appear.
- The core U.S. CTCL opportunity is small in absolute patient numbers (rough order of magnitude: a few thousand eligible patients per year), so revenue potential is naturally capped unless label expansion succeeds.
- CLS, including rare fatal events in the pivotal trial, obliges tight monitoring and risk-management programs that can limit uptake or keep some prescribers cautious.
- Highly concentrated prescriber base: a few hundred hematology/oncology specialists drive most of the addressable volume, and 60% of targets are clustered in about ten states, creating execution risk.
- Significant dilution risk from equity financing and warrant exercises as the company raises capital for launch and post-launch activities.
- Execution risk typical of ultra-orphan commercial launches: reimbursement, access, physician education and patient identification all need to work in sync.
- If real-world usage remains modest or competitors improve their positioning, CTOR could struggle to support its current valuation.
Key catalysts and timeline
Q4 2025 – U.S. commercial launch
LYMPHIR enters the U.S. market through a specialty distribution network, with initial focus on a defined universe of CTCL-treating hematologists and oncologists. Early prescription trends and access feedback will set the tone for 2026.
Q1 2026 – First post-launch numbers
Initial revenue contribution from LYMPHIR and basic key performance indicators (new patient starts, reorder patterns, gross-to-net dynamics) are expected in early financial updates. This is often where the market recalibrates expectations.
2026 – Financing and balance-sheet events
The group has already announced capital raises to support the launch. Additional funding steps, warrant exercises or partnership-related cash flows could shift the risk profile, positively or negatively, depending on terms and timing.
2026 – 2027 – Market adoption curve
Over the next 4–6 quarters, the market will likely focus on sustained prescription growth, broadening prescriber base, inclusion in guidelines and real-world safety/efficacy consistency versus pivotal data.
2027 and beyond – Expansion optionality
Longer-term upside scenarios include broader T-cell malignancies, potential combination strategies with checkpoint inhibitors, and international markets beyond the initial agreements. All of these remain optionality, not base case.
Pipeline overview
| Asset | Indication | Stage | Status |
|---|---|---|---|
| LYMPHIR | Relapsed/Refractory CTCL (adult Stage I–III) | Commercial | FDA APPROVED |
| LYMPHIR | First-line CTCL (potential) | Early research / strategy | EXPLORATORY |
| LYMPHIR | Other T-cell malignancies / IO combinations | Investigator-initiated / early stage | EXPLORATORY |
LYMPHIR is currently the only revenue-generating asset for Citius Oncology. The company and academic partners are exploring broader use in T-cell malignancies and combinations, but at this stage those programs should be treated as long-dated options rather than near-term drivers.
Financial profile and runway
Balance sheet highlights (last annual 10-K)
Cash and cash equivalents
Approximately $0
LYMPHIR inventory
Approximately $8.27M
Stockholders’ equity
$46.14M
Operating metrics (FY 2024)
Net loss
About -$21.1M
Operating expenses
About $20.6M
Estimated cash runway at filing
Very limited, pre-financing
The standalone oncology entity had virtually no cash at the time of the last annual 10-K and relies on support from the parent and capital markets. Subsequent press releases detail additional fund-raises designed to cover launch costs and working capital. From a trading perspective, this means that balance-sheet risk and dilution are structurally part of the CTOR story and should be treated as core risk factors, not exceptions.
Retail sentiment and narrative
On retail-oriented platforms (Stocktwits, Reddit, X), the discussion around CTOR after LYMPHIR’s launch is split between two clear camps. One group focuses on the rarity of CTCL, the fact that LYMPHIR is now an approved, boxed-warning immunotherapy in a previously underserved niche, and the prospect of broader indications or combinations over time. This camp tends to emphasize the upside ranges highlighted by analysts and management.
The other camp is more cautious and concentrates on funding risk, repeated equity offerings, warrant overhang and the practical limits of a small orphan market. The most recent 18 million dollar raise has reinforced the view that dilution will likely accompany each step of the launch phase, especially if ramp-up is slower than bulls expect.
Note: sentiment observations are based on public discussions among non-professional traders. These comments are often emotional, incomplete and sometimes inaccurate. They should never be treated as research or advice, but only as an indicator of how retail participants are framing the story at a given moment.
Price action and scenario framework
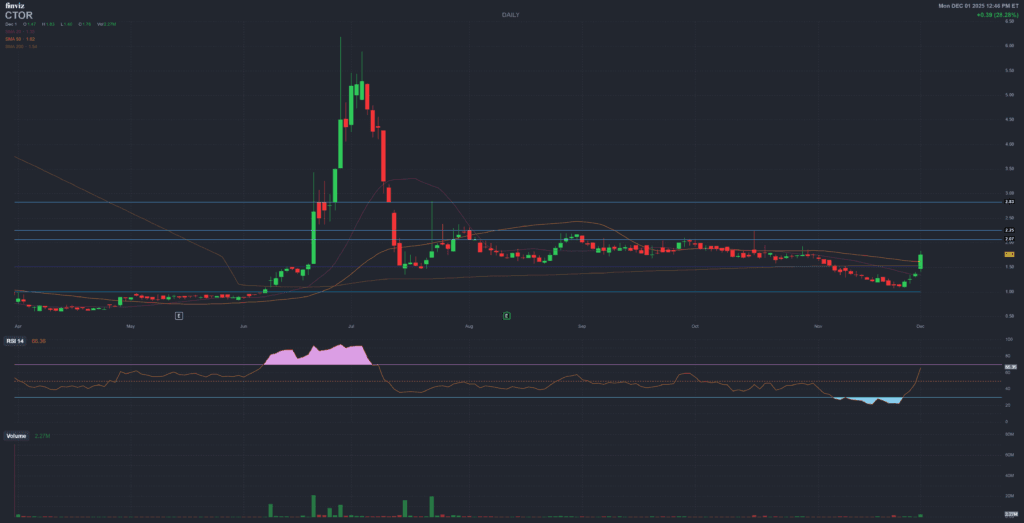
CTOR has already moved sharply from its lows near the fifty-cent range to recent levels around one and a half dollars, with spikes above six dollars earlier in the year. This type of path is typical of small-cap biotech names around major catalysts and financing events: wide swings, thin liquidity at times and heavy sensitivity to news.
The following ranges are illustrative only and are meant to frame possible paths, not to suggest entries, exits or position sizes:
- Support band: the 0.55–0.95 dollar zone has historically acted as a deep wash-out area where selling pressure previously exhausted itself during risk-off phases.
- Intermediate band: prices between roughly 1.20 and 2.80 dollars have hosted most of the recent trading after the approval narrative became “real” but before robust, recurring revenues were visible.
- Upper band: the 4.00–6.00 dollar region corresponds to prior spikes where expectations ran ahead of fundamentals, often around key headlines or speculative flows.
For many traders, the practical takeaway is not a mechanical buy/sell signal but an understanding that CTOR is structurally volatile, with potential for both large percentage gains and equally large drawdowns. Any use of these levels should be done within a personal risk-management plan, independently of this report.